div class=”ChapterContextInformation”>
34. Impact of Urethral Reconstruction on Sexual Function
Keywords
Sexual functionUrethral strictureUrethroplasty34.1 Introduction
Established believes concerning outcomes in anterior urethral reconstruction are changing, with regard both to the impact on sexual activity and genital cosmetic appearance [1]. Following repair of anterior urethral strictures, successful outcome is generally defined as normal voiding without further invasive procedures. However, the urethra is also involved in sexual and genital functions and most patients undergoing urethroplasty worry about the impact of surgery on sexual life. Therefore, the aim of stricture repair is not only to restore urinary function but also to safeguard sexual activity and guarantee genital cosmesis.
A complete evaluation of anterior urethroplasty should include the sexual point of view, which seems to play an important role in overall postoperative patient satisfaction. Despite the importance of the sexual outcome following penile or bulbar urethroplasty techniques, very few studies have focused on these aspects so far. The most commonly reported sexual problems following anterior urethroplasty include: erectile and ejaculatory dysfunctions, penile curvature or shortening, dissatisfactions with genital cosmetic appearance, sensorial impairment of genitalia and overall dissatisfaction about sexual life. The prevalence of specific postoperative sexual problems may be related to the site of urethral reconstruction (penile or bulbar) and to the technique of urethroplasty employed (graft versus flap and transecting versus non-transecting techniques). In penile urethral reconstruction, the increasing use of buccal mucosa seems to provide better results than the use of skin flaps which easily distorts the cosmesis and elasticity of the penis, thus increasing the risk of complications [1]. In bulbar reconstructions, graft augmentation techniques seem to have less impact on sexual outcome than transecting anastomotic techniques, which has been considered the gold standard for a long time.
Aim of this chapter is to analyse various anterior urethroplasty techniques for penile and bulbar reconstructions and related postoperative sexual complaints, providing also some technical hints on the surgical procedures in order to improve sexual outcomes.
34.1.1 Neuroanatomy
The understanding of the neuroanatomy of the urethra is a prerequisite for prevention or reduction of the neural injuries during the surgical urethral repairs. Two sets of nerves control erectile function. They are designated as autonomic (motor) and somatic (sensory) innervation.
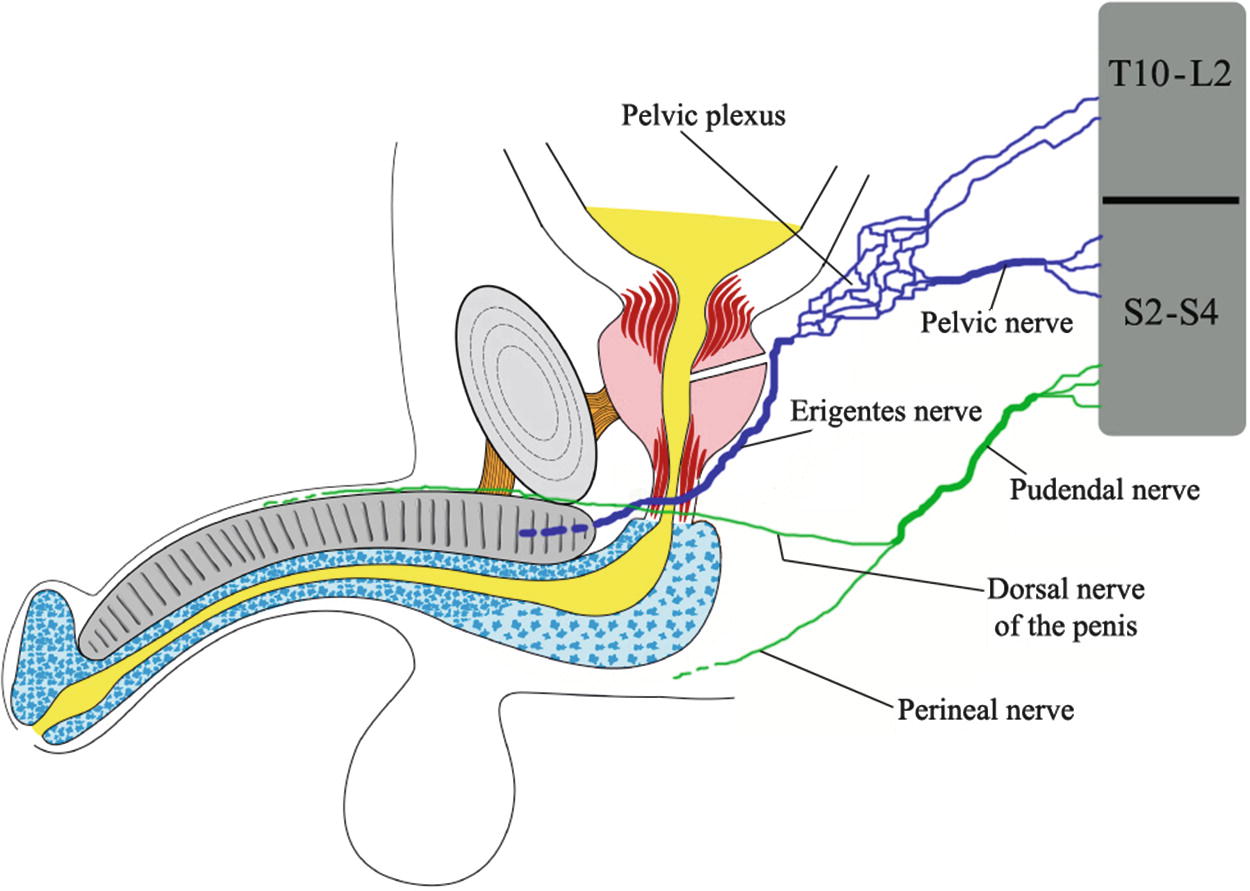
Relationship of the nerves to the urethra
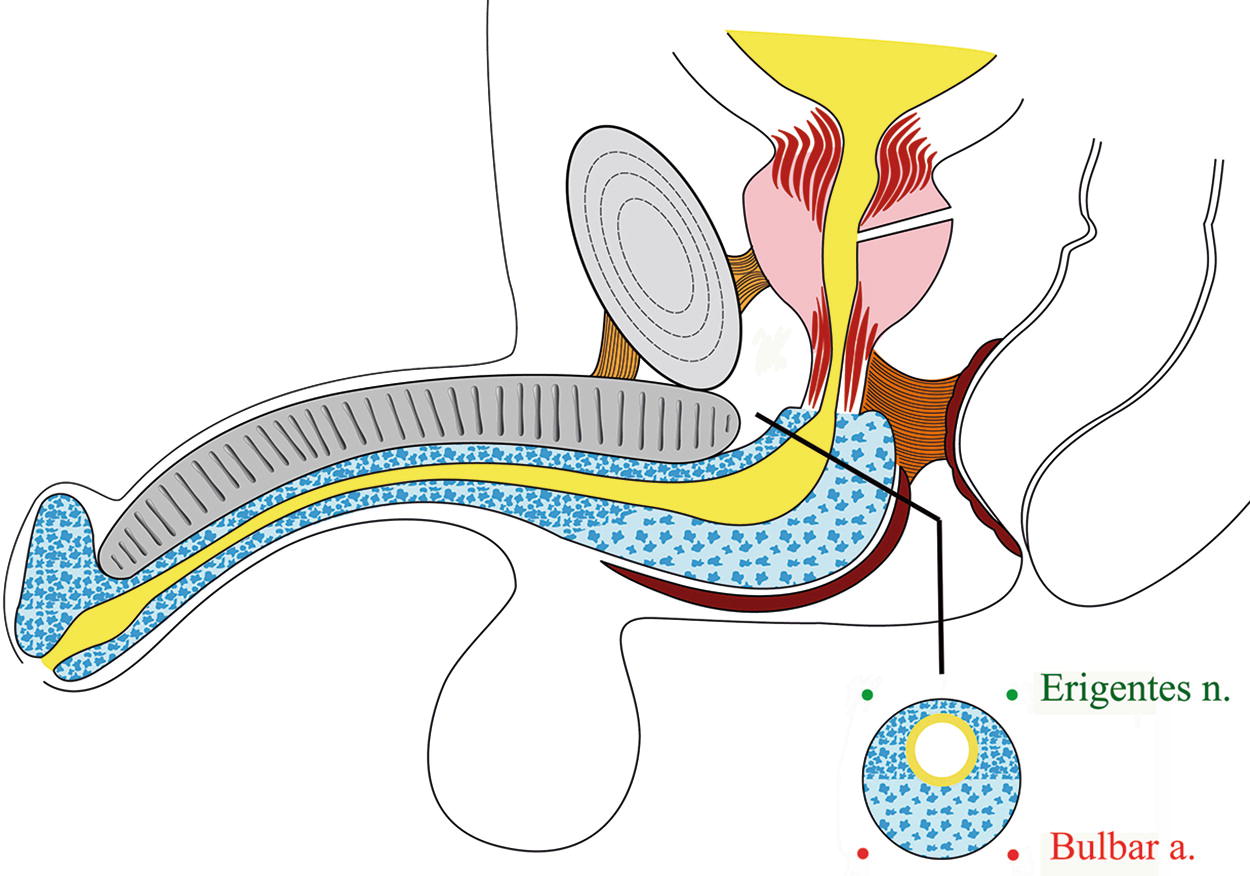
The proximal bulbar corpus spongiosum reaches its greatest thickness in the “bulb” and then it disappears in the membraneous urethra: in this area there are the two bulbar arteries (at 5 and 7 o’clock) and the two erigentes nerves (at 11 and 1 o’clock)
The somatic system consists of the paired pudendal nerves which receive contributions from S2-S4 and innervate the pelvis andala perineum. They accompany the internal pudendal vessels through Alcock’s canal to innervate the striated muscle of the pelvic floor (levator ani) and the penis (ischiocavernosus and bulbocavernosus muscles). The pudendal nerves also supply sensory perception to the pelvis and the perineum and terminate as the dorsal nerve of the penis.
The dorsal nerve of the penis is an important branch of the pudendal nerve : it travels underneath the levator ani and continues distally along the dorsolateral surface of the penis lateral to the dorsal artery, gives multiple branches, and terminates in the glans penis. Anterior urethral innervation is by the urethrobulbar nerve, a branch of the perineal nerve : this latter is a branch of the pudendal nerve.
34.1.2 Anatomical and Surgical Considerations
The bulbar urethra is emptied, following micturition, by its closure mechanism created by the elasticity of the spongy tissue, combined with the voluntary contraction of the bulbospongious muscle ; in a similar way, an efficient urethral emptying, during ejaculation, is achieved by the combination of the bulbo-penile urethral closure-pressure with the intermittent reflex contractions of the spongious muscle. In particular, erection of the spongy tissue not only considerably increases the passive urethral closure-pressure but it also increases the bulk of the bulb which, in turn, increases the effect of the reflex ejaculatory contractions of the surrounding bulbo-spongious muscle. Thus, organic emission is a function of the posterior urethra resulting from a simultaneous contraction of the bladder neck, sustained contraction of the seminal vesicles and opening of the distal sphincter. Once the emission has passed the distal sphincter, the continuous flow of the semen is converted into a forceful intermittent ejaculation by the anterior bulbo-penile urethral structure.
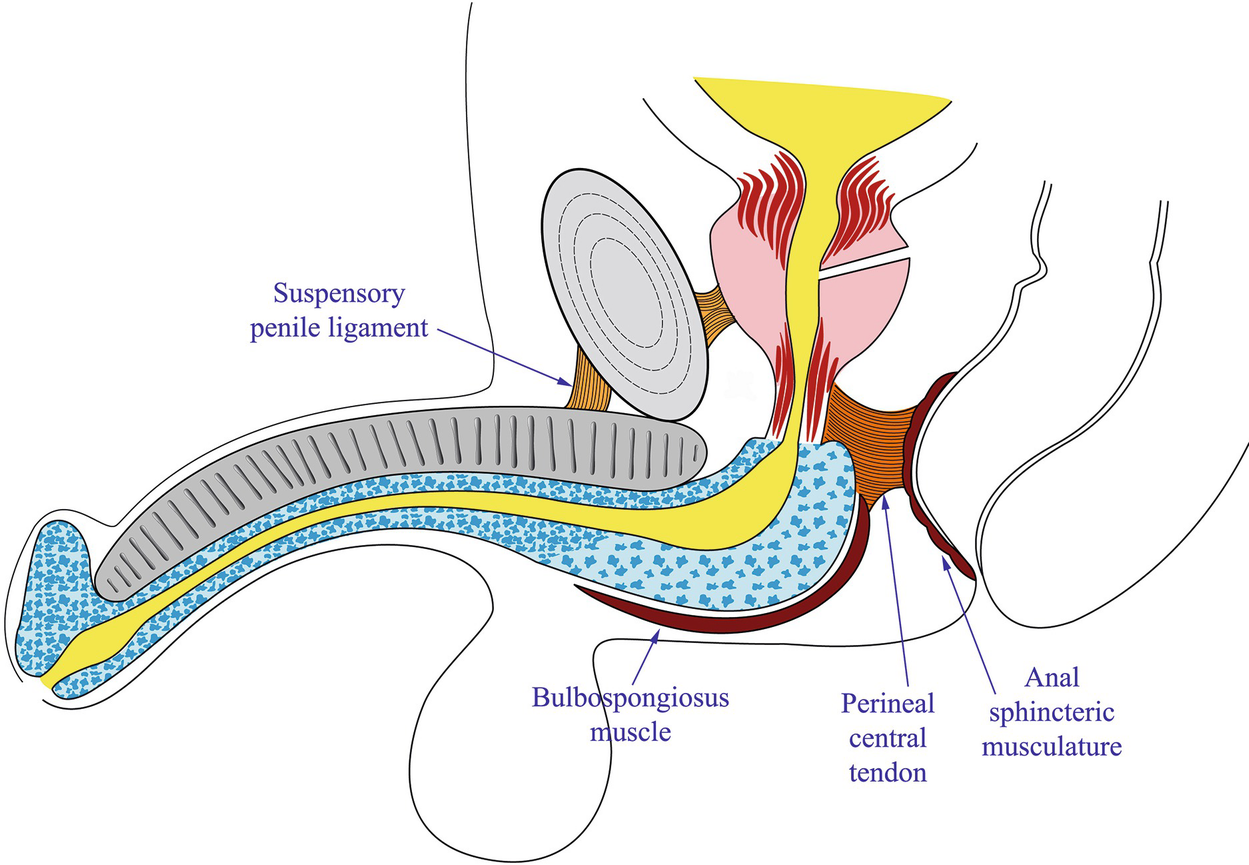
Externally the bulbar corpus spongiosum is enveloped in the bulbospongiosus muscle. The proximal bulbar urethra is anchored deep in the perineum, especially in the midline posteriorly by a fibrous septum: the perineal central tendon
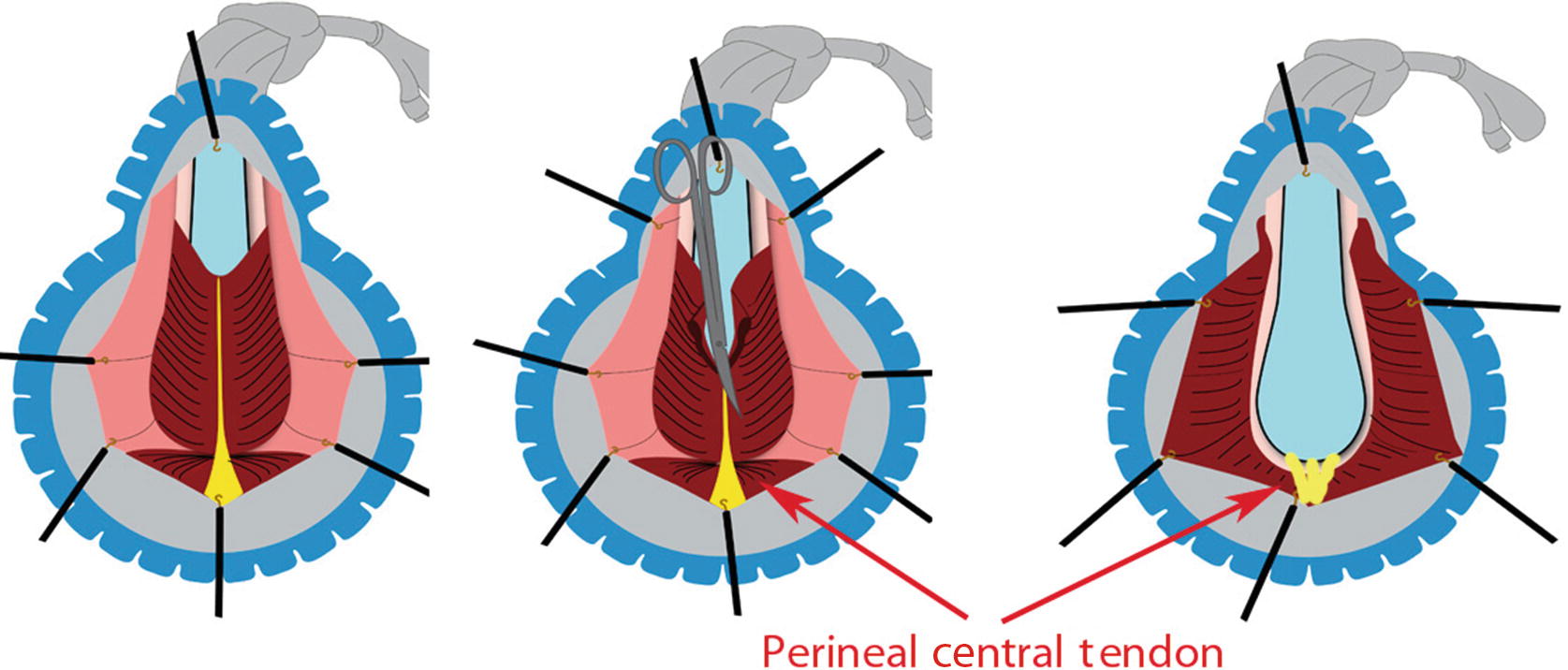
During urethroplasty and opening of the bulbospongiosus muscle, it is advisable to spare the Perineal Central Tendon
With the aim to reduce semen sequestration Barbagli described a new muscle- and nerve-sparing bulbar avoiding dissection of the bulbospongiosum muscle from the corpus spongiosum and leaving the central tendon of the perineum intact [10].
However, it must be said that urethral reconstructive procedures inevitably impair the natural emptying mechanism of the spongy tissue, since they interrupt the structural muscle-elastic circularity of the urethral tube which may stop the rhythmic urethral voiding contractions. Furthermore, urethral elasticity is often already impaired, to some extent, by the spongio-fibrotic disease [11].
34.2 Sexual Complaints Following Bulbar Urethroplasty
Anastomotic procedures (%) | BM graft (%) | |
|---|---|---|
Penile curvature | 36 | 8 |
Penile shortening | 38 | 11 |
Impaired erection | 79 | 15 |
Complete loss of erection | 9 | 0 |
Overall dissatisfaction in erection | 26 | 19 |
Temporary impotence | 53 | 33 |
Impaired sexual life | 57 | 19 |
Overall satisfaction regarding sexual life | 74 | 97 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree