div class=”ChapterContextInformation”>
69. Urologic Complications of Gender-Affirming Surgery
Keywords
TransgenderGender dysphoriaGender confirming surgeryMetoidioplastyPhalloplastyVaginoplastyFistulaUrethral stricturePersistent vaginal cavity69.1 Introduction
A recent meta-analysis of multinational data estimates the prevalence of individuals identifying as transgender to be approximately 355 in 100,000, and 9.2 in 100,000 will seek gender affirming surgery or hormonal therapy . [1] Canner et al. report 37,827 encounters with the ICD-9 code of transsexualism or gender identity disorder from 2000 to 2014 from the National Inpatient Sample, a representative pool of inpatient visits across the United States, of which 10.9% involved gender-confirming surgery. The investigators showed that the number of patients seeking genital surgery more than doubled from 698 in 2000 to 2005 to 1584 in 2006 to 2011. Furthermore, the percentage of individuals seeking gender-affirming procedures and who are covered by Medicare and Medicaid increased from 25% to 70% between 2012 to 2013 and 2014, respectively [2].
Survey data shows that the prevalence of attempted suicide amongst unoperated transgender people is alarmingly high at 32% [3]. Gender affirmation therapy has been shown to significantly decrease psychopathology in the gender dysphoria population [4]. While gender affirming surgery is not life-saving the way trauma surgery can be, it can nonetheless be a life-saver. With the number of gender affirming procedures on the rise, so too will its related complications, and the reconstructive urologist must be prepared to treat them.
69.2 Complications in the Transgender Male
69.2.1 Metoidioplasty Surgery Overview
The choice between metoidioplasty and phalloplasty is both practical and personal. The ultimate goal of transgender surgery is always relief of the underlying gender dysphoria . Phalloplasty may take a two-surgeon team at least 8 hours to complete while metoidioplasty can be accomplished by a single surgeon in 4 hours or less. Patients may realize that their gender dysphoria may be relieved with the smaller surgery and opt for the less morbid metoidioplasty.
Some other reasons why someone would choose metoidioplasty is that it offers absence of a donor site while allowing for upright micturition. The patient must understand, however, that performing penetrative sexual intercourse is unlikely. Perovic and Djordjevic reported a mean neophallus length of 5.7 cm after metoidioplasty, although this length is often not achievable even with maximum phallic lengthening. In this study 17/22 patients (77%) were satisfied, while 5/22 (23%) later decided to undergo phalloplasty [5]. This highlights another benefit of metoidioplasty: if the metoidioplasty proves to be functionally or psychologically inadequate, that the patient first underwent metoidioplasty does not forfeit the option for phalloplasty. In fact, there is benefit to this route as it essentially stages phalloplasty, making surgery less difficult for the surgeon and less taxing for the patient. Lastly, metoidioplasty complications tend to be fewer and more minor than those associated with phalloplasty. These include urethrocutaneous fistulae, urethral stricture, and persistent vaginal remnant. Frey et al. report in their review that the overall complication rate after metoidioplasty is 0.43 per patient versus 0.88 after radial forearm flap phalloplasty [6].
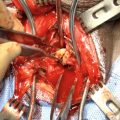
Phallus lengthening and urethral lengthening in metoidioplasty
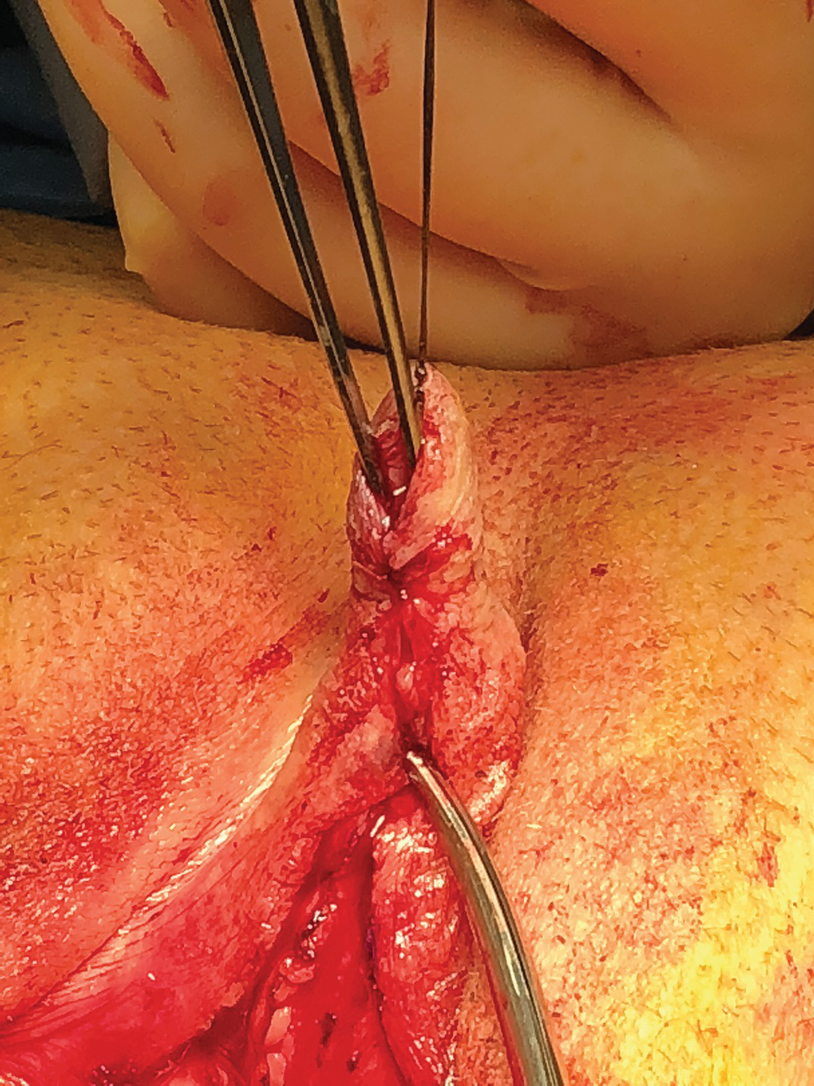
Flat male-type perineum after vaginectomy. Scrotoplasty using labia majora flaps, a technique used in both phalloplasty and metoidioplasty
69.2.2 Phalloplasty Surgery Overview
Historically, the ideal phalloplasty was described as producing an (1) aesthetically pleasing neophallus with a (2) neourethra that permits standing micturition and (3) enough bulk to accommodate a prosthetic stiffener while (4) providing tactile and erogenous sensation and (5) being reproducible with (6) minimal donor site morbidity [8]. Patients wanting the ability to achieve penetrative intercourse will preferentially choose phalloplasty.
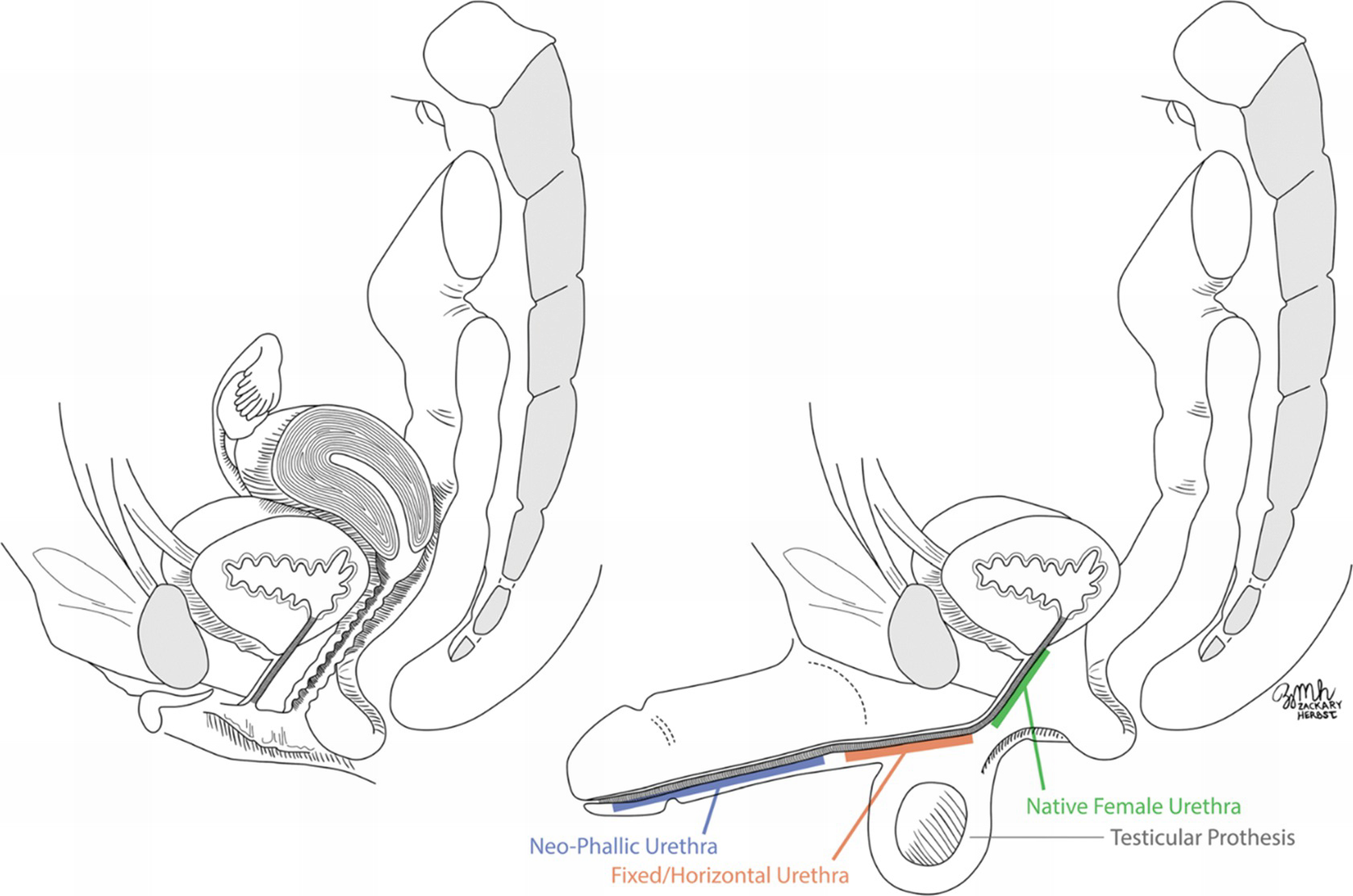
Native female (left) and phalloplasty anatomy. The pars fixa (fixed urethra) is lengthened from the native urethra and continues on as the pars pendulans (penile/neophallic urethra). The anastomosis between the pars fixa and pars pendulans is a common site for urethral complications. Used with the permission of the publisher [31]
A variety of phalloplasty techniques have been described in the literature, including radial forearm flap (RFF), anterolateral thigh (ALT) pedicled or free flap, latissimus dorsi free flap, suprapubic pedicled flaps, and osteocutaneous free flaps (mostly historical). Radial forearm free flap has emerged as the most commonly used method of phalloplasty followed by anterolateral thigh. Early attempts at free radial forearm flap phalloplasty had a 100% complication rate with total graft loss rate of 60% due to thrombosis [9]. In 2011, Doornaert et al. reported that 7.2% of their radial forearm flap phalloplasties demonstrated a minor degree of partial flap necrosis, while Ascha et al. reported in 2018 a partial neophallus loss rate of 3.4% [10, 11]. Phalloplasty technique has advanced a great deal with great acceleration in recent years, but complications still abound. The following are the most common urologic complications of female-to-male gender affirming surgery.
69.2.3 Urethrocutaneous Fistula
Urethrocutaneous fistula is the most common complication of both metoidioplasty and phalloplasty, with an incidence of 10% to 64% [9–14]. Generally older series report higher complication rates, while improvements in technique have resulted in lower rates of fistula in more recent series. While urethrocutaneous fistulae can be found anywhere along the urethra, they will almost exclusively form at the anastomosis between the fixed urethra to the pendulous urethra , also known as the anastomotic urethra [15]. In 1999, Fang et al. reported a urethrocutaneous fistula rate in radial forearm free flap of 63.6%, of which 36% of these individuals healed spontaneously within 2 months. The remainder required an average of 1.55 surgeries to correct their fistula. All urethrocutaneous fistulae occurred at the junction of neophallus to neoscrotum [12]. In the largest reported cohort of RFF phalloplasties to date, Doornaert et al. reported a urologic complication rate of 40% (127/316). Fistula was the only complication in 25% (80/316); 66% (53/80) of these fistulas closed spontaneously [10]. Recent advancements in phalloplasty technique have yielded an even lower fistula rate of 10% with a minimum follow-up of 6 months in 149 patients undergoing RFFF phalloplasty [11]. An important note is the impact of basic reconstructive tenets, such as multiple-layer closures. With vaginectomy, the bulbospongiosus muscle, which lines the vestibule, is available to form an additional layer over the newly created fixed urethra. Massie et al. have highlighted the importance of this layer by showing that omission of additional layers of vascularized muscle tissue, particularly in vagina-sparing phalloplasty , increased the risk of urethrocutaneous fistula 400% from 14% to 56% [16]. Even with solid technical skills, urethrocutaneous fistula will occur. Many will spontaneously resolve, especially in cases where the leak is small and there is a large amount of overlying tissues, such as a urethrocutaneous fistula tracking through the neoscrotum. Many other fistulae will not spontaneously close and will need surgical intervention. For example, if the fistula is large and superficial, there simply is no tissue to heal over this area. The need for surgery is often obvious to the clinician in these cases.
Urethrocutaneous fistulae usually present obviously with patients complaining of urinary leakage from an ectopic site when they void. Other symptoms include decreased force of stream, straining, incomplete voiding, dysuria and dribbling, especially if a urethral stricture is also present. Physical examination may reveal a visible fistulous tract, induration, erythema, tenderness, a fluctuant mass with expressible purulence, and urinary retention (aided by bladder scan). Ultrasound may be a useful adjunct to detect fluid collections, including abscesses, especially when there is a coexisting urethral stricture that may cause pooling and urostasis. Of note, while urethrocutaneous fistula can coexist with a downstream urethral stricture causing increased voiding pressures, fistulae tend to occur early while strictures will present later.
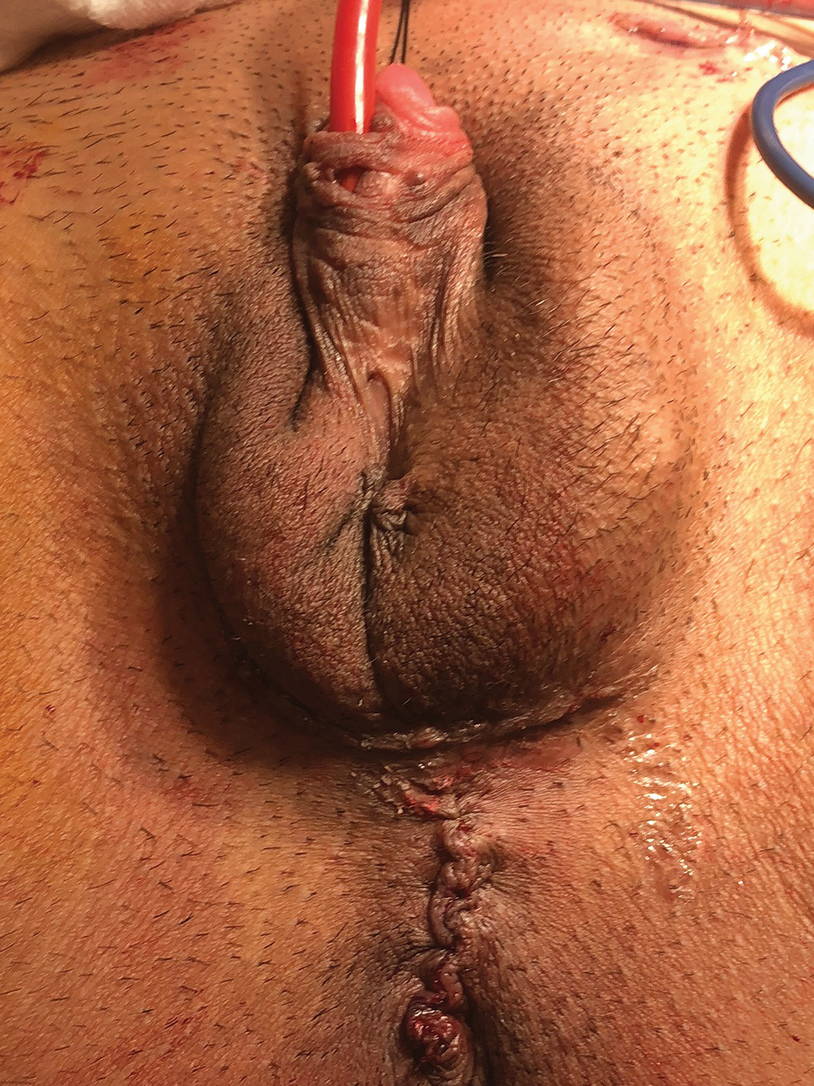
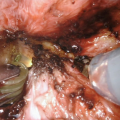
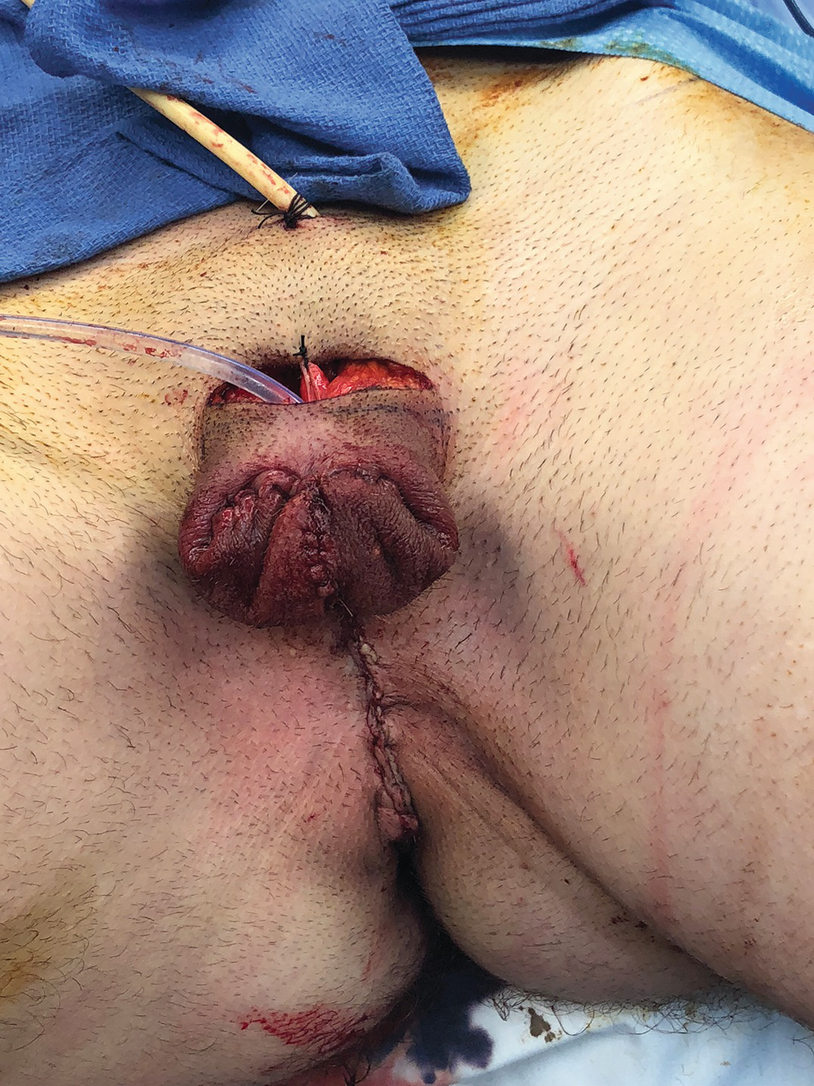
Distal urethral fistula after previous metoidioplasty
69.2.3.1 Conservative Management of Urethrocutaneous Fistula
Most urethral fistulae are early events after surgery, often when a suprapubic tube is in place already from the original surgery. Surgeons may opt to continue urinary diversion for longer than the standard time (about 30 days for phalloplasty and about 21 days for metoidioplasty at our center) as initial treatment of fistula. However, data to support improved outcomes from longer urinary diversion times do not exist. Further, some experts at very high-volume centers in Austin, Texas and Ghent, Belgium believe that prolonged urinary diversion is not helpful after GAS. They assert that fistulae that are unlikely to spontaneously heal will never heal with any amount of prolonged diversion, and conversely that fistulae that will heal in time will do so with or without urinary diversion. More research is clearly needed to clarify the role of urinary diversion in healing of fistula. Ascha et al. report that when they do heal spontaneously, most of these fistulae will close within 3 months [11].
69.2.3.2 Surgical Management of Urethrocutaneous Fistula
The patient is usually placed in low dorsal lithotomy. Usually we pass a flexible cystoscope because this, together with external physical exam, is all that is needed to understand the anatomy well. Fistula repair follows the standard template: closure should be performed in many nonoverlapping layers. Local advancement flaps of well vascularized tissue should be used if possible. If a urethral stricture is present in close proximity to the fistula, then they should be repaired simultaneously; the stricture and fistula can often be removed entirely and anastomotic urethroplasty performed. In cases where urethral strictures are to be treated with a first stage Johanson urethroplasty, the urethrocutaneous fistula will be incised and effectively treated. If the urethral stricture and urethrocutaneous fistula are separated by a significant distance, then two separate operations should be done.
69.2.4 Urethral Stricture
Methods of urethroplasty at the time of metoidioplasty or phalloplasty all share the commonality of requiring long suture lines and anastomoses. These blood supply “watershed” points are the epicenter of stricture formation, the incidence of which ranges from 13.7% to 57.1% [10–12, 14, 15, 17]. Lumen et al. reports that the rate of urethral stricture by location after phalloplasties are the urethral meatus in 15%, pendulous urethra in 24%, anastomotic urethra in 41%, fixed urethra in 13% and multifocal in 8% [18]. In our practice, we have found that virtually all phalloplasty strictures are short (<2 cm) and almost universally located at the anastomosis between the pars fixa and the distal skin (penile) urethra. The remainder are distal, meatal type strictures, generally caused by distal flap loss and scarring over of the previously well-open meatus. Each of these entities have their preferred approach which we will describe in the following section.
The diagnostic workup for urethral strictures is similar to urethrocutaneous fistula. Imaging the urethra is important to determine stricture length. The surgical approaches are essentially the same as those applied to native urethral strictures: dilation/DVIU, anastomotic, first stage Johanson followed by possible second stage, buccal or skin grafts when necessary and the use of flaps for more complex repairs. Despite these many options, we have found that almost all phalloplasty strictures today are amenable to either EPA or Heineke-Mikulicz urethroplasty . One important precept not to be violated is to allow enough time to pass for post-surgical inflammation to fully resolve and regain strength and elasticity. We prefer to wait at least 6 months. In rare cases we will reluctantly proceed with repair in as few as 4 months if the tissue has healed quickly.
When urethral stricture presents prior to the 6-month mark, the surgeon must use temporizing measures for urinary drainage. Options include urethral dilation (often repeatedly), direct vision internal urethrotomy (DVIU) or suprapubic cystotomy . Success of DVIU as a definitive treatment for urethral stricture is extremely low at 13% [19]. As in natal males, an extremely short stricture may enjoy success with DVIU according to some reports; however, we ourselves have not found DVIU useful as a definitive treatment [20, 21]. Unlike natal males who have a robust blood supply in the corpus spongiosum, the neophallus has a tenuous blood supply and it is our experience that hypoperfused, scarred skin is resistant to any treatment other than excision or staged urethroplasty. DVIU and urethral dilation should only be used as a temporizing measure to reach the 6-month mark at which point definitive surgery may proceed.
69.2.5 Urethral Stricture after Metoidioplasty
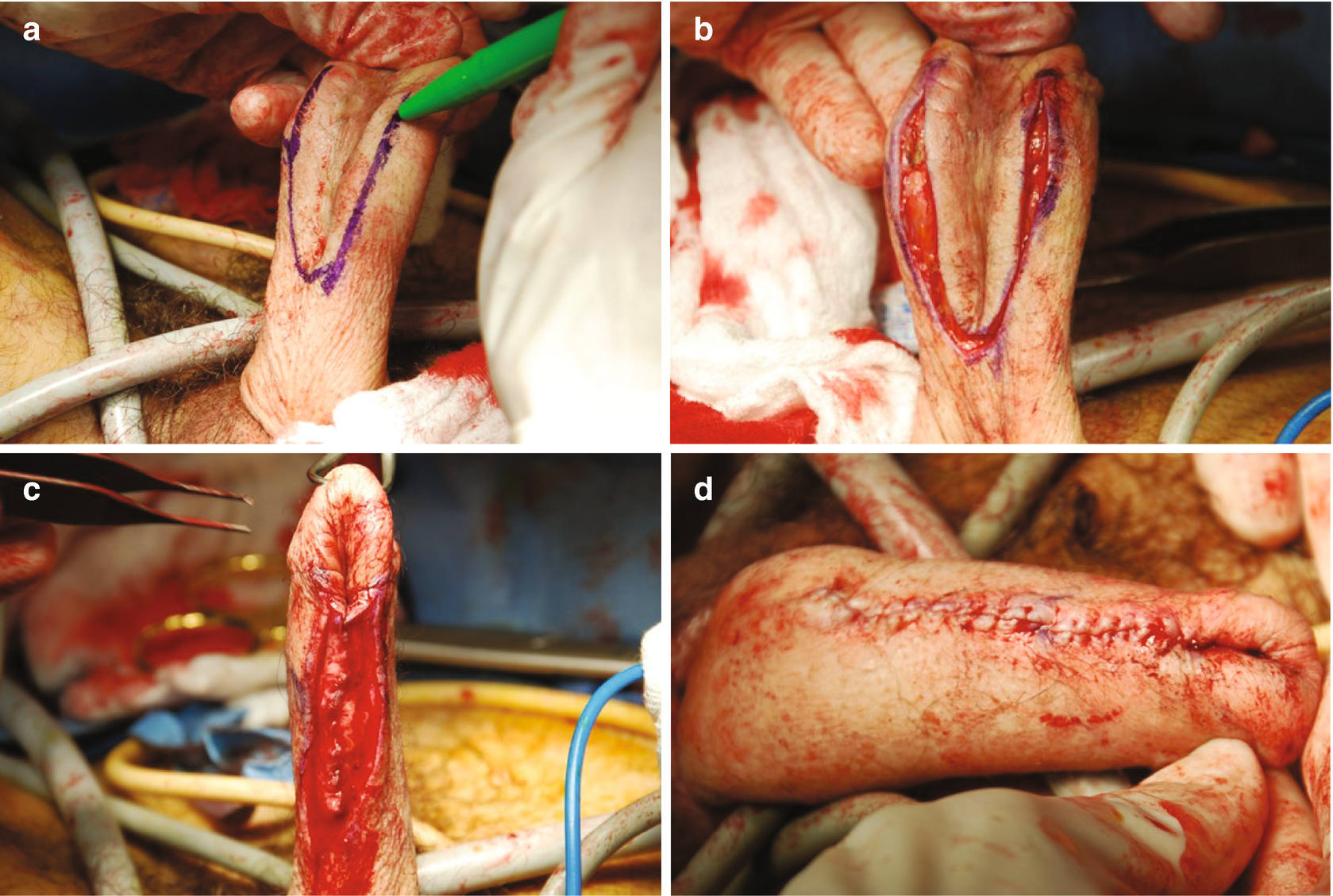
Metoidioplasty urethral stricture can be thought of as two basic entities: meatal stenosis versus all other types of stricture. If only the meatus is involved, then a standard meatoplasty may suffice. If, however, the stricture is more substantial than a meatal stenosis, then the options are either a single-stage buccal mucosa graft dorsal inlay versus a staged Johanson-type repair. A single-stage buccal mucosa dorsal inlay is preferred when the patient has a short urethral stricture. Otherwise, the author prefers to perform a staged Johanson urethroplasty with buccal mucosal graft , as needed.
69.2.6 Urethral Stricture after Phalloplasty
69.2.6.1 Meatoplasty
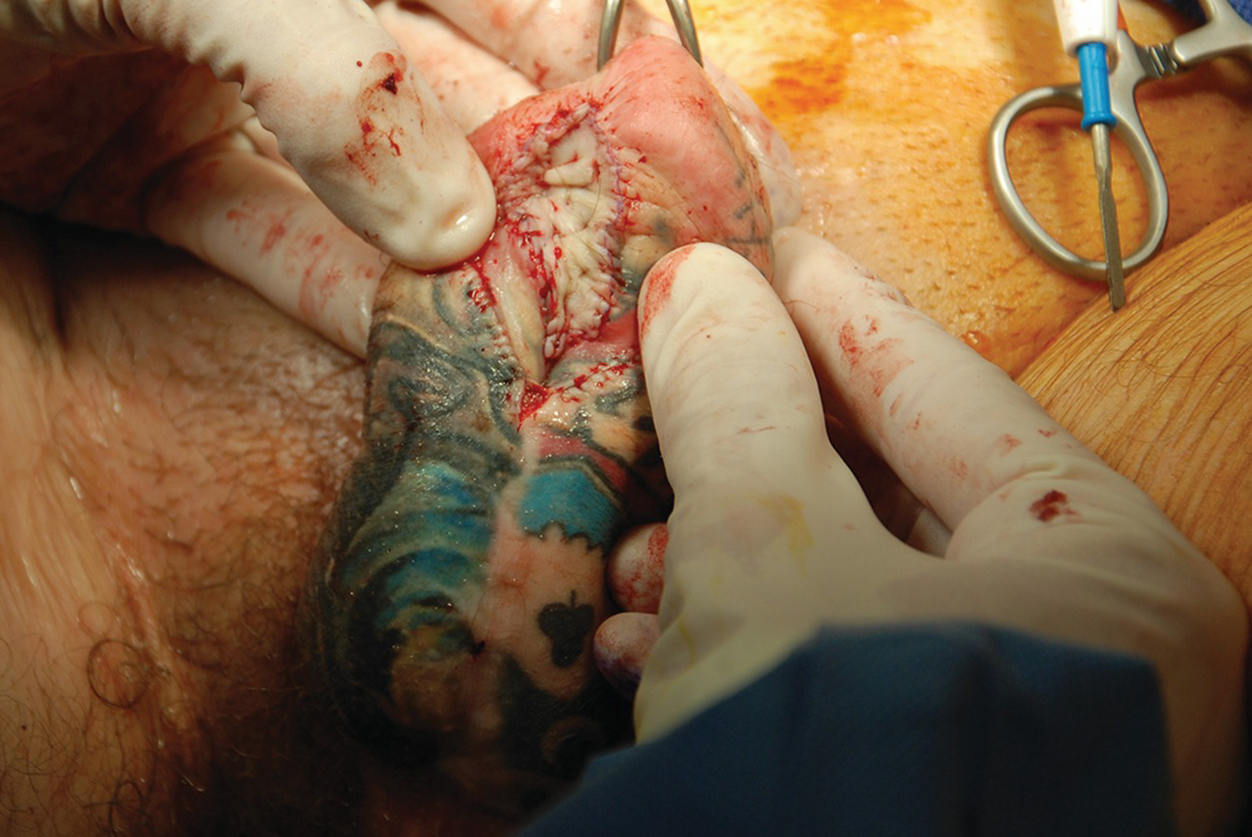
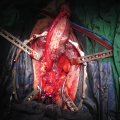
Distal urethral stricture after phalloplasty treated with a dorsal skin graft and one stage urethroplasty
69.2.6.2 Short Urethral Stricture Repair
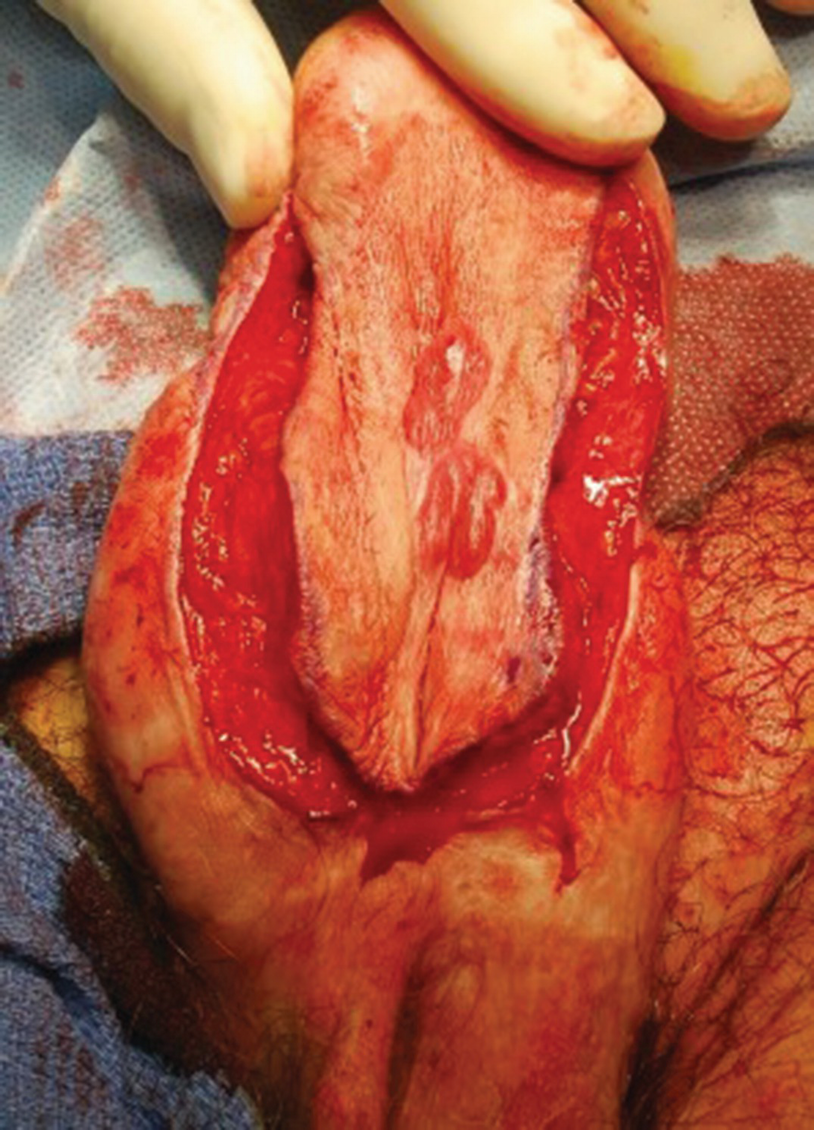
Second stage Johanson urethroplasty for long penile urethral stricture in an older model “nonmicrovascular” phalloplasty. Note near total contraction of the previously placed buccal grafts, center. Poor performance of buccal grafts in phalloplasty strictures has led to us abandoning their use
The majority of urethral strictures will be found at the anastomosis of the fixed urethra. Very short strictures (~1 cm) are amenable to a Heineke-Mikulicz principle-based nontransecting anastomotic urethroplasty in which the stricture is incised longitudinally and closed horizontally. Placement of stay sutures on either side of the longitudinal incision helps facilitate a transverse closure. Nontransecting anastomotic urethroplasty success rate is reported at 58% [18].
When the urethral stricture is slightly longer (~1.5 cm) or particularly scarred due to fistula or infection, excision and primary anastomosis (EPA) is necessary. When performing EPA on a natal male, one must mobilize the urethra proximally and distally to create a tension-free anastomosis. This native urethral blood supply is absent in the neophallus, and urethral mobilization must be modified. We tend to free the proximal stump more thoroughly than the distal stump, and to spare the dorsal blood supply of the pars fixa by freeing up the ventral tissue connections most thoroughly. The strictured segment is excised, and the remaining segments of urethra are spatulated and anastomosed with absorbable suture, yielding a success rate of at least 57% with a mean follow-up of 34.9 months [18]. Any failures after primary anastomotic urethroplasty are treated with Johanson urethroplasty with skin graft.
69.2.6.3 Long Urethral Stricture Repair
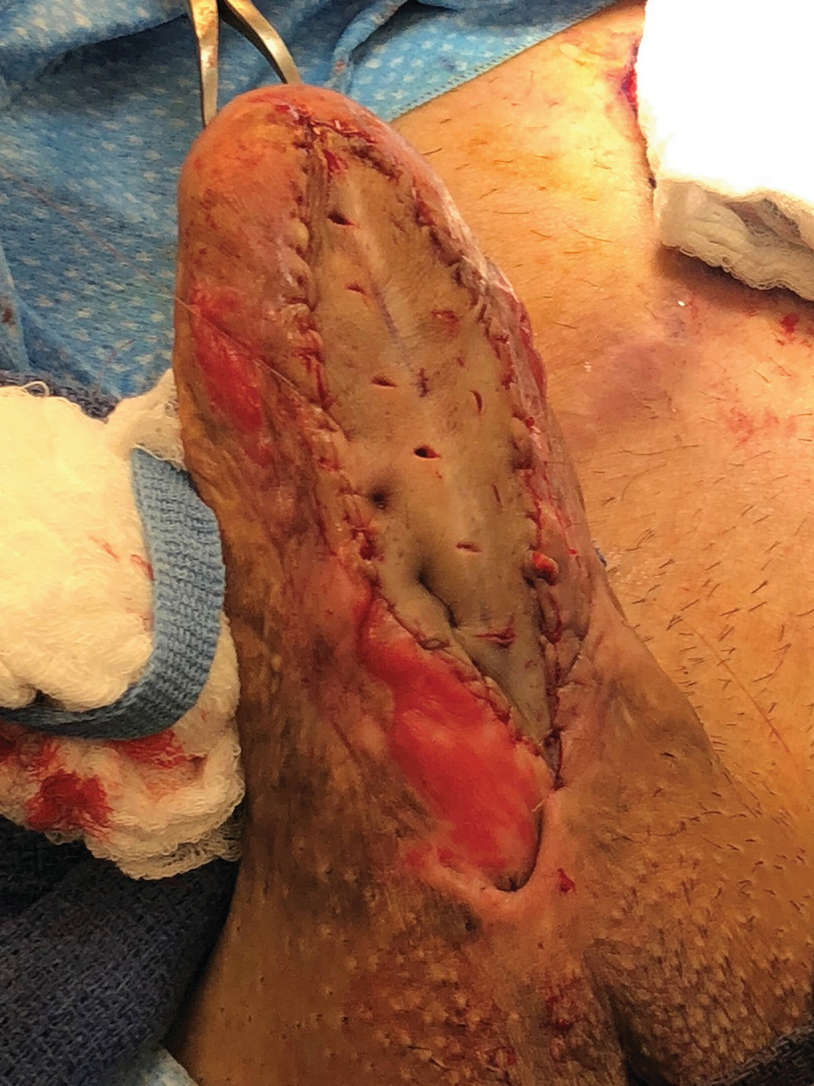
First stage Johanson urethroplasty of long penile stricture with split thickness skin grafts

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree