div class=”ChapterContextInformation”>
67. Urethral Reconstruction in Female-to-Male Gender Affirming Surgery
Keywords
Buccal mucosa graftFemale-to-maleGender affirmation surgeryMetoidioplastyPhalloplastyUrethroplasty67.1 Introduction
Female-to-male gender affirmation phalloplasty represents one of the most challenging issues in genitourethral reconstructive surgery. Although techniques and outcomes have been vastly improved during the past decades, an ideal solution that would satisfy all patients’ requirements has not yet been reported. Creation of a cosmetically acceptable and functional neophallus, and a neophallic urethra that will enable voiding in standing position, are the most important surgical tasks. These goals can be achieved by metoidioplasty or total phalloplasty. Metoidioplasty is a variant of phalloplasty for female-to-male transsexuals, wherein a small neophallus is created from a hormonally enlarged clitoris, with urethral lengthening in a one-stage procedure. It results in male appearance of genitalia, with completely preserved erogenous sensation and voiding in standing position, but without the possibility for penetrative sexual intercourse. Total phalloplasty represents the creation of an adult sized neophallus using extragenital tissue, either as a pedicled flap or a free flap. A variety of flaps have been reported for female-to-male gender affirming phalloplasty. However, none of these methods can accomplish all goals of phalloplasty: satisfactory cosmetic appearance, sufficiently large size for safe implantation of penile prosthesis to enable penetration, preserved erogenous sensation, and a competent neophallic urethra with the meatus at the top of the neoglans [1, 2].
Neophallic urethroplasty remains a huge reconstructive challenge. Either as a part of metoidioplasty or total phalloplasty, a wide spectrum of tissues and techniques are used to create a very long and functional neourethra from the native meatus to the top of the neophallus. The technique of urethroplasty generally depends on the type of phalloplasty. Neourethra can be created using a “tube-within-a-tube” principle , as in radial forearm free flap (RFFF) phalloplasty . Vascularized genital flaps (clitoral skin, labia minora, vaginal mucosa), free flaps (RFFF, gracilis flap, ulnar flap) and grafts (skin, buccal mucosa) can be used for single-stage or staged urethroplasty [3]. Despite recently improved techniques, complication rates remain high. The most common complications, urethral fistula and stricture, are reported in 10–90% of cases, with the overall urethral complication rate of 40% [4]. Urethral complications were found to be higher in the transmale population compared to the cismale population (39.4% vs. 24.8%) [4]. We are still searching for ideal tissue for urethral reconstruction that will guarantee a successful outcome. Tissue engineering could prove promising, but there are still many technical and bioethical issues to be resolved [5].
67.2 Surgical Options for Female-to-Male Gender Affirming Phalloplasty
There are two surgical options for female-to-male gender affirming neophalloplasty – metoidioplasty and total phalloplasty. Metoidioplasty is a variant of phalloplasty for transsexuals who wish to avoid complex, staged procedures. It is established as a one-stage procedure in which a neophallus is created from a hormonally enlarged clitoris, and urethra is lengthened to the top of the glans, resulting in male appearance of genitalia, voiding in standing position and completely preserved erogenous sensation. The main disadvantage is a small penis, which does not allow penetration [6]. Phalloplasty , or surgical construction of an adult size penis, still represents one of the most difficult surgical procedures in genital reconstructive surgery. Since the original report on penile reconstruction, many authors reported their experience with different variants of local flaps for neophalloplasty, generally based on the inferior epigastric vessels [7]. Since then, different microsurgical free flaps have been developed and are regularly used in female-to-male gender affirmation surgery. Although phalloplasty techniques have improved in many aspects, they still have many drawbacks, and neither is considered superior to others. Extensive scarring of the donor area, absence of erogenous sensitivity and common urethral complications make these multi-staged procedures unacceptable for many transsexuals. The choice of a specific surgical technique mostly depends on the patient’s desires and expectations. However, it is the surgeon’s duty to inform the patient about all the advantages and disadvantages, as well as all possible complications after surgical procedure, their severity and management.
Voiding in standing position is one of the main goals in female-to-male gender affirmation surgery, and it requires a functional neophallic urethra. To achieve this goal, a complex urethroplasty has to be performed. It includes the creation of a competent neophallic part and a funnel-shaped bulbar part that will bridge the native female meatus and the neophallic urethra. This funnel part of the urethra and anastomosis with the native meatus has a predilection for fistula and stricture formation, due to the high pressure and relative obstruction of the urinary stream at this point. This is why additional support with well-vascularized tissue is preferable in all cases [3, 8]. Many techniques and materials, genital and extragenital, have been used in order to achieve a successful outcome. Usually, the type of phalloplasty determines the type of urethral reconstruction. There are some general principles though, for the creation of the neourethra in transgender. It is possible to create it in the first stage, using a “tube-in-a-tube” technique in total phalloplasty, or combined flap and graft technique in metoidioplasty. Another option is to perform a staged urethroplasty, based on the Johanson principle [9]. However, lack of spongiosal and supportive tissue, frequent urethral complications and common failure to achieve good voiding stream while standing, make urethral lengthening the biggest reconstructive challenge in female-to-male gender affirmation phalloplasty.
67.2.1 Metoidioplasty
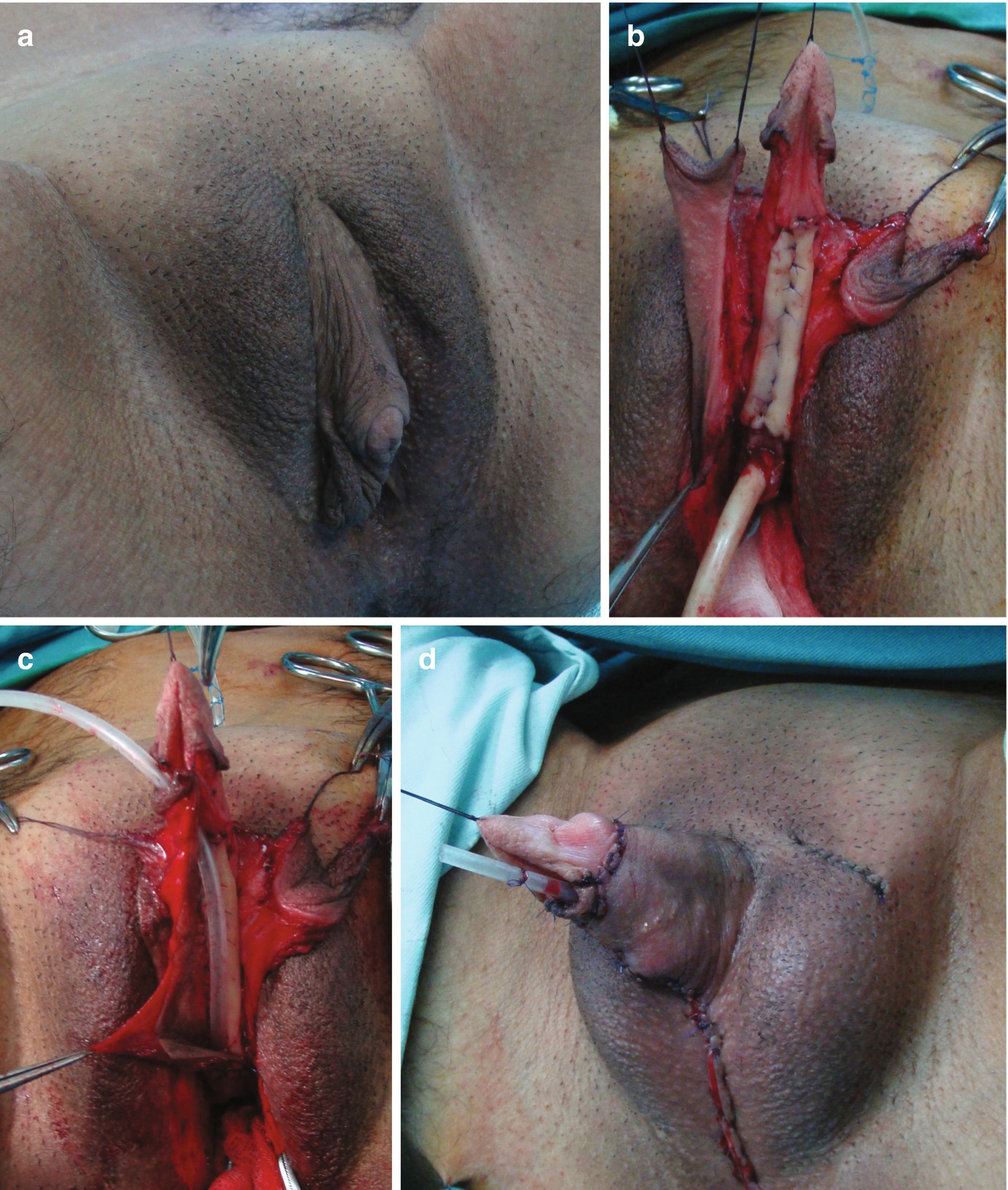
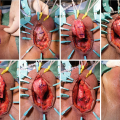
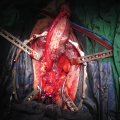
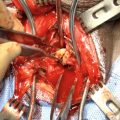
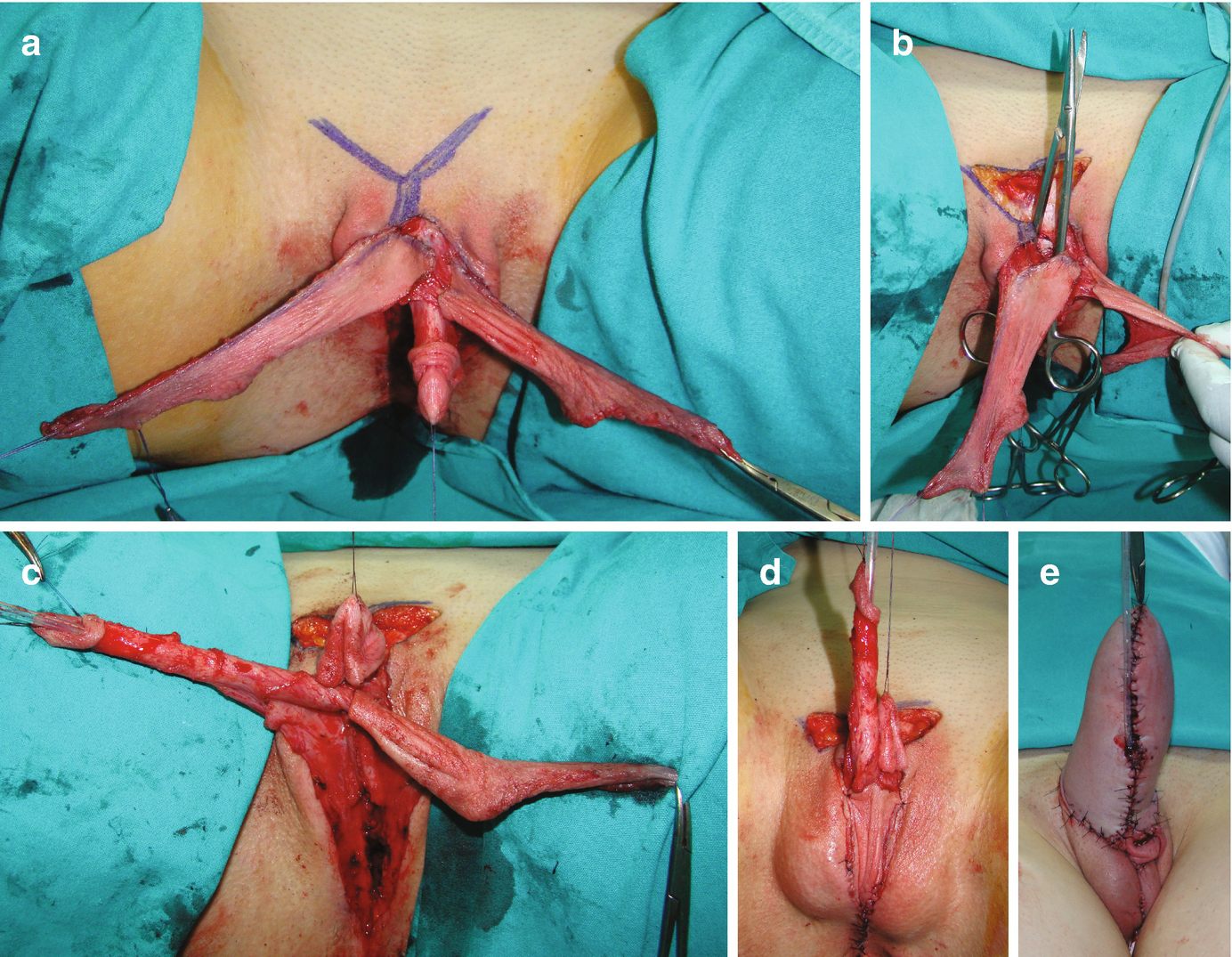
Metoidioplasty technique (combined graft and flap urethroplasty). (a) Preoperative appearance . Clitoris is hormonally enlarged; (b) The short urethral plate is divided, creating a gap. This maneuver is necessary to straighten and lengthen the clitoris. Buccal mucosa graft is placed and quilted to cover the gap. Flap is harvested from the right labia minora, to be joined with buccal mucosa graft; (c) Bulbar part of the neourethra is formed by joining the periurethral tissue and the proximal part of the urethral plate. The right labia minora flap is joined with a buccal mucosa graft to create the penile neourethra. Distal part of the urethral plate is tubularized creating the distal neourethra; (d) Appearance after metoidioplasty. Two testicular implants are inserted into the scrotum created from both labia majora
67.2.1.1 Urethral Lengthening in Metoidioplasty
Urethroplasty presents the most difficult part of one-stage metoidioplasty. Originally, the urethral plate was left intact and the penis was short and curved [12]. In order to improve outcomes and prevent complications, modifications of urethroplasty were reported [13]. A very long flap of anterior vaginal wall (5–7.5 cm) was used to create bulbar urethra. Additional support was provided by one labia minora flap, used to cover suture lines. Penile neourethra was created from a labia minora flap and the urethral plate, which was divided at the level of the female urethral opening. As the course of dissection was from proximal to distal, it could compromise the vascularization of the mobilized urethral plate. The same authors reported long-term results of this technique, with complication rate of 85% [14]. They stated that an average of 2.6 procedures per patient were performed in their series to complete genital reconstruction.
There are certain issues that led to significant improvements in urethral lengthening in metoidioplasty. New general principles of urethroplasty, introduction of buccal mucosa as the best material in reconstruction of the urethra, improved knowledge on clitoral anatomy and neurophysiology, as well as rich experience in gender affirmation surgery and severe hypospadias repair, led us to establish the current, one-stage metoidioplasty [15–18]. Advanced urethroplasty with voiding while standing is possible with simultaneous use of buccal mucosa graft and genital flaps , providing lengthening of the native female urethra to reach the tip of the glans, as in males. Masculine appearance of the external genitalia is further enhanced by scrotoplasty and the insertion of two testicular implants. This approach was first reported in 2009, with the main advantage of being a one-stage procedure: removal of the vaginal mucosa, metoidioplasty, and the creation of the entire neourethra and scrotum [19].
In considering metoidioplasty, it is important to recognize all similarities between male and female anatomy. The clitoris, like the penis, is composed of two paired corporal bodies, a clitoral corona, and a dorsal neurovascular bundle. Unlike in males, the ventral portion of the clitoris is made up of a short, wide urethral plate. In order to straighten and lengthen the clitoris as much as possible, dorsal suspensory ligaments need to be completely detached from the bone. Also, the wide and short urethral plate is carefully dissected from the clitoral bodies and divided at the level of glanular corona, additionally lengthening the clitoris but creating a urethral plate defect. Urethral lengthening includes reconstruction of bulbar, penile and glandial urethra [17].
Bulbar Neourethra
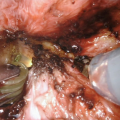
Urethroplasty starts with the reconstruction of the bulbar part. One of the main advantages of this technique is the simultaneous removal of the vaginal mucosa and use of periurethral mucosa in creation of the bulbar urethra. Periurethral tissue proved to be better supportive material compared to the previously used vaginal flap, in terms of postoperative complications [2]. The bulbar urethra suffers the highest urinary stream pressure and therefore represents a high-risk point for postoperative fistula formation. The joining of the clitoral bulbs over the neourethra and the additional covering with vascularized surrounding tissue is considered a key for successful fistula prevention at the place of anastomosis with native female urethral orifice.
Penile Neourethra
Further urethral lengthening is performed using a buccal mucosa or skin graft and vascularized genital skin flaps [20, 21]. Buccal mucosa graft has become the gold standard material in urethral reconstruction, especially due to its’ histological attributes. Graft harvesting is a safe procedure without morbidity of the donor site [15, 22]. The graft is prepared and sutured to cover the defect left after the division of the short urethral plate (Fig. 67.1b). Split-thickness skin graft can be used as an alternative, harvested from labia minora or other genital area. Quilting of the graft to the corporeal bodies is very important for better survival. Urethral reconstruction is completed either by using a longitudinal dorsal clitoral skin flap button-holed ventrally, or a flap harvested from the inner surface of the labia minora. Labial flap combined with buccal mucosa graft is reported as the best option, resulting in a complication rate lower than 7% [20]. Either skin or labial flap is joined with buccal mucosa graft over a 10–12 Fr catheter to form penile neourethra (Fig. 67.1c). In both cases, all suture lines are covered with well vascularized tissue to prevent fistula formation.
Glandial Neourethra
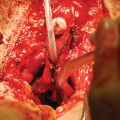
Glans wings are mobilized like in hypospadias repair [16]. Distal part of the urethral plate and glans wings are used to form glandial neourethra and lengthen the urethra to the top of the neophallus. Penile shaft is reconstructed using the remaining clitoral and labial skin. Labia majora are joined in midline to create the scrotal sac, and two silicone testicular implants are inserted (Fig. 67.1d).
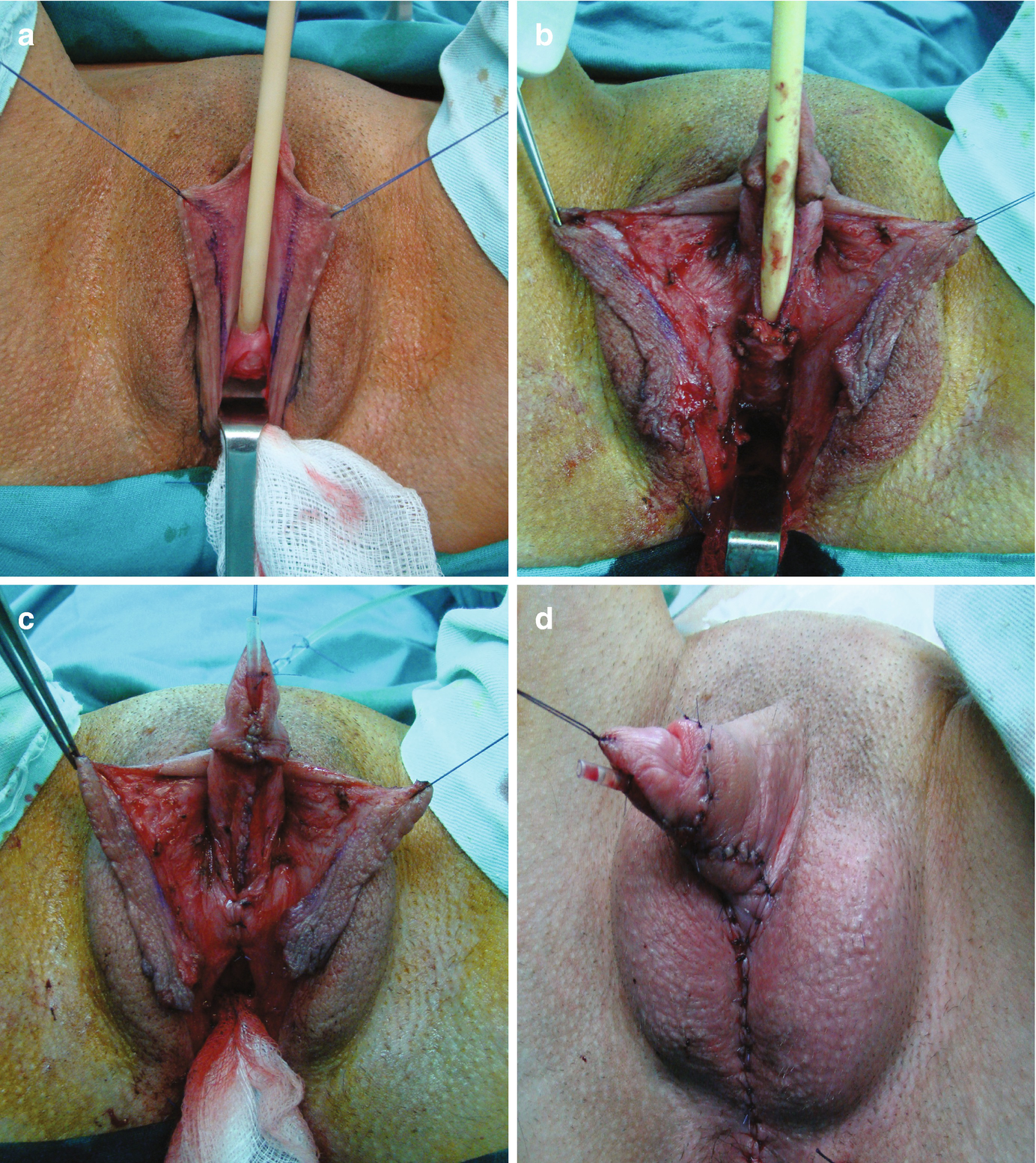
Metoidioplasty technique (urethral plate tubularisation). (a) Preoperative appearance. Urethral plate is wide and well developed; (b) Bulbar urethra is created using a well-vascularized vaginal flap and proximal urethral plate; (c) Urethral plate is tubularized to create penile and glandial neourethra. Proximal neourethra is covered with surrounding tissue to prevent postoperative fistula; (d) Outcome after metoidioplasty. Scrotum is formed by joining labia majora, and testicular implants are placed
Perioperative Care
Suprapubic tube is inserted into the bladder for urine derivation, and 10–12 Fr catheter is left in the neourethra as a urethral stent. Broad spectrum antibiotics and anticholinergic drugs are prescribed while the catheter is in place. Urethral stent is removed 10 days after surgery, and neourethra irrigated with 5 ml of saline every day until beginning of voiding. Patient starts to void 3 weeks after surgery, and suprapubic tube is removed safely the next day.
This single-stage metoidioplasty has good aesthetical and functional outcomes, according to patients’ evaluation. Reconstruction of the neourethra enables voiding while standing in majority of reported cases. Postoperative complications related to urethroplasty can be minor (dribbling, spraying) and major (urethral fistula and stricture). Urethral fistula occurs in 7–25% of all reported cases in the long-term, and is repaired by excision of the fistula and overlaying with available local vascularized flaps. Anastomotic or buccal mucosa graft urethroplasty is performed for urethral stricture repair [18–20].
67.2.2 Total Phalloplasty
Phallic reconstruction in male transsexuals should ideally create an aesthetically pleasing phallus with sufficient length for vaginal penetration, which has tactile and erogenous sensibility, enables voiding in standing position and with acceptable donor site morbidity. A variety of neophalloplasty techniques are described, using pedicled vascularized flaps or free flaps with microvascular anastomosis . The radial forearm free flap (RFFF) is the gold standard technique in female-to-male gender affirmation phalloplasty. Other frequently used flaps include: pedicled or free anterolateral thigh (ALT) flap , musculocutaneous latissimus dorsi (MLD) free flap, abdominally based flaps and osteocutaneous fibula free flap .
67.2.2.1 Radial Forearm Free Flap (RFFF) Phalloplasty
The fasciocutaneous RFF flap was originally described for the release of cervical skin contracture after burns, but nowadays is most widely used flap for penile reconstruction. This flap enables creation of an adult-sized, sensate neophallus and voiding while standing in a single-stage surgery. Penetrative sexual intercourse is achieved if the flap is used as an osteocutaneous flap, or if a penile prosthesis can be implanted at a later stage [23].
The classical approach for RFFF phalloplasty includes simultaneous work of three teams: gynecology, urology and plastic surgeons’ team. The fasciocutaneous forearm flap is harvested from the non-dominant forearm, measuring approximately 14–18 cm in length and 14–16 cm in girth. If an osteocutaneous flap is harvested, not more than one third of the radius diameter is used with soft tissues. The flap is tubularized while connected to the neurovascular pedicle, then transferred to the pubic area and fixed in the proper position. Microvascular anastomosis is performed between radial artery and inferior epigastric artery or femoral artery; as well as cephalic and great saphenous veins. Eventually, a second venous anastomosis can be performed between a deeper radial concomitant vein and the great saphenous vein. The medial and lateral antebrachial nerves are anastomosed to the ilioinguinal or genitofemoral nerve, as well as dorsal clitoral nerve, in order to allow for neophallus sensation. The forearm defect is covered with full-thickness or split-thickness skin grafts [24]. Still, unsightly appearance of the donor area, insufficient volume of the neophallus for safe insertion of two cylinders of penile implant, and high incidence of urological complications, are important disadvantages of RFFF phalloplasty. Urethral reconstruction in RFFF phalloplasty is performed using “tube-in-a-tube” technique , or two-staged neourethra prelamination technique.
“Tube-in-a-Tube” Urethroplasty
This urethroplasty technique was originally described in radial forearm flap phalloplasty, and used afterwards in groin flap and anterolateral thigh flap phalloplasty [25–27]. It includes the design of an additional part of the flap that is tubularized over a 14 Fr catheter by inverted skin tubing to approximate free skin edges and make the urethral lining. The subcutaneous sutures are placed without tension, minimizing the exposure of suture materials in the neouretheral lumen. A penile flap is wrapped around the neourethra and sutured to skin edges to create the penile shaft. However, additional tissue is necessary to cover the gap between the neophallic and native urethra and prevent possible complications. A vaginal flap and labia minora flap are used to create this bulbar, funnel-shaped part of the neourethra and extend the channel to the top of the neophallus. A wide-based vaginal flap is harvested from the anterior vaginal wall, containing the mucosa and muscle of the vagina. Both labia minora flaps are raised as well, joined and anastomosed with the vaginal flap in a funnel-shaped fashion. Suture lines are covered with an additional layer of available, well-vascularized local tissue. Another option is to use combined wide urethral plate and labia minora flap, in selected cases. A suprapubic catheter is inserted for urine derivation. Urethral catheter is removed 2 weeks after surgery. After that, the patient starts with voiding. Suprapubic catheter is removed once the patient is able to void without significant residual volume. Urethral fistula and strictures occurred in more then 40% of cases, but the incidence of complications has been reduced recently due to refinements of the technique [4, 23, 28].
67.2.2.2 Anterolateral Thigh (ALT) Flap Phalloplasty
Perforator flaps have recently become very popular for neophallic reconstruction, due to evident benefits: great amount of favorable tissue, easier microsurgical procedure, better appearance of the donor area. Anterolateral thigh flap is a frequently used perforator flap, both as a free flap and pedicled flap. It is nourished by the descending branch of the lateral circumflex femoral artery. Pedicle length and position are of utmost importance in the planning of pedicled flap phalloplasty. Since perforator anatomy is not predictable, a detailed preoperative assessment of perforators by CT angiography is necessary for a successful outcome [29].
The flap is harvested by dissecting the perforator until the vascular pedicle of the rectus femoris muscle is reached. When dissection of the perforator is finished, the flap is elevated and carefully transferred through a previously created muscle tunnel to reach the recipient site. Care must be taken to avoid torsion or kinking of the pedicle, as well as injury of nerves inside the tunnel. If the pedicle is too short, or if there is tension in the pedicle, conversion to a free flap is indicated. Two nerves are always taken with the flap and anastomosed with ilioinguinal and dorsal clitoral nerves. Split-thickness skin graft is used to cover the donor site.
Several methods for urethral reconstruction in ALT flap phalloplasty are described. The anterolateral thigh flap is seldom thin enough to be reconstructed using the tube-in-a-tube principle, especially in female-to-male persons, who have gynoid habitus, thicker thighs, and consequently excess flap thickness. For this reason, ALT phalloplasty in this population requires other urethroplasty techniques, and various free flaps and grafts are reported as a solution in these cases – radial free forearm flap, superficial circumflex iliac artery perforator (SCIAP) flap , flap prelamination with skin graft, peritoneal flap. However, most of these procedures have uncertain outcomes [30].
Free Flap Urethroplasty
Free flap urethral reconstruction was originally described in oncological cases, but also as salvage option after partial necrosis of the neourethral part of a tube-in-tube phalloplasty, or in patients who are not eligible for a tube-in-a-tube RFFF or ALT phalloplasty, but who do wish to void in the standing position. The use of the RFFF, gracilis flap, ulnar flap and pedicled superficial circumflex iliac artery perforator flap (SCIPF) are described for urethral reconstruction [27, 31, 32]. The RFFF is a superior flap, with thin skin and a long vascular pedicle, which makes is suitable for urethral reconstruction and microsurgical vascular anastomosis in the groin. It provides the most effective single-stage urethral reconstruction, without the distinctive scar in the forearm. The disadvantage is that it requires a complex, microvascular surgery, performed together with a pedicled perforator flap. A rectangular RFFF of approximately 3 × 16 cm is designed and harvested from the non-dominant forearm. Donor site is closed primarily or covered with a split-thickness skin graft. The vascular pedicle is carefully dissected from the distal to the proximal end, to its origin at the brachial artery. The flap is tubularized in two layers around an 18 Fr Foley catheter with the skin facing the catheter. Labia minora flaps are created on both sides, and conjoined over a catheter to lengthen the native urethra. After elevation of the RFFF and pedicle dissection, the flap is transferred to the genital region. First, the urethral anastomosis with previously formed bulbar part is performed in a two-layer fashion. The vascular pedicle is tunneled to the groin, the radial artery is anastomosed to the common femoral artery and the cephalic vein to the great saphenous vein. In this way, complete urethral lengthening is performed as a single-stage procedure. However, long-term urethral complications are reported in more then 50% of cases, including urethral stricture, fistula and hairy urethra. Also, microsurgical anastomosis carries a risk of vascular accidents and flap loss [27, 32].
67.2.2.3 Musculocutaneous Latissimus Dorsi (MLD) Free Flap Phalloplasty
The musculocutaneous latissimus dorsi flap has useful anatomical attributes for phallic reconstruction (good size, volume and length of the neurovascular pedicle), and therefore represents our method of choice for neophalloplasty in male transsexuals. In gender affirmation surgery, this technique includes creation of a neophallus using a latissimus dorsi musculocutaneous free flap, its fixation in the pubic region, microvascular anastomosis at the recipient site and reconstruction of the scrotum [33].
Clitoral lengthening and repositioning are performed by complete detachment of dorsal clitoral ligaments from the pubic bone, to mobilize the clitoris and enable its fixation at the base of the neophallus. All vascularized labial and clitoral flaps are used for maximal urethral lengthening. The perineum is reconstructed to achieve a male appearance, the scrotum is created using labia majora. A 15 × 13 cm average-sized flap is designed on the non-dominant side, consisting of two parts – a rectangular part for the neophallic shaft and a circular part for the neoglans. Flap harvesting starts with an incision of the anterior skin margin down to the deep fascia. The plane is developed between the latissimus and anterior serratus muscle, using sharp and blunt dissection, and the flap is lifted to expose the neurovascular pedicle. The pedicle, surrounded by fatty tissue, is identified and dissected up to the axillary vessels. The thoracodorsal nerve is identified and isolated proximally for 3–4 cm, preserving its vascularization. The flap is completely elevated, but the neurovascular bundle is left in place and is not transected until the recipient vessels and nerve have been prepared for anastomosis. The flap is tubularized while still on the pedicle, creating the neophallus. The distal end is closed to create the glandial part of the neophallus. Completely created neophallus is then detached from the axillar region, after clamping and dividing the subscapular artery, vein and thoracodorsal nerve at their origin, in order to achieve maximal pedicle length. The donor site is approximated and closed directly. In case of significant tension, the defect is covered with split-thickness skin graft. All neurovascular structures at the recipient site are identified, the neophallus is transferred to the pelvic area and a microsurgical vascular anastomosis is performed. The neophallic base is positioned and fixed at the recipient site [34].
Urethral lengthening in MLD free flap phalloplasty can be performed in the same stage, using the same principles of urethroplasty like in metoidioplasty, or as a staged procedure. In one-stage urethroplasty, all available vascularized genital flaps (labia minora, clitoral skin) are used to lengthen the urethra to the proximal third of the neophallus. In some cases, it is enough to enable voiding in standing position. For additional lengthening, staged urethroplasty with buccal mucosa graft is performed.
One-Stage Urethroplasty
The native female urethra needs to be lengthened at least 13–15 cm to enable voiding in standing position. Using the same principle of urethral reconstruction as in metoidioplasty, it is possible to create a neourethra long enough to make voiding while standing feasible after one-stage surgery [2, 34].

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree