Key points
- •
There are shortcomings in U.S. Food & Drug Administration (FDA)–approved treatments for androgenetic alopecia (AGA) that are currently available.
- •
There is a growing body of double-blind, placebo-controlled, randomized studies that demonstrate both qualitative and quantitative benefits of nutritional supplements to combat hair thinning.
- •
Larger studies with longer follow-up times are needed for each individual supplement to assess optimal dosing and frequency, duration of effect, and less common adverse events.
- •
Nutritional supplements can lead to a variety of side effects, interactions with medications, and interference with laboratory testing.
Background, definition, and history
Androgenetic alopecia (AGA) is prevalent, with estimates that it affects 43% of women and 83% of men by age 60 (level of evidence: 2c). Depression, anxiety (level of evidence: 2b), and decreased quality of life (level of evidence: 1b) are frequently seen in these patients. Despite AGA’s prevalence and morbidity, topical minoxidil in women and men and oral finasteride in men remain the only treatments approved by the U.S. Food & Drug Administration (FDA). The side effects of irritant and allergic contact dermatitis and hypertrichosis with minoxidil (level of evidence: 1a) and erectile dysfunction and decreased libido with finasteride (level of evidence: 2a) may affect long-term adherence. Other treatments, such as low-level laser light, show inconsistent results, and many of the supportive studies provide low-quality evidence (level of evidence: 1a).
A need for additional effective and well-tolerated therapies may account for the use of complementary and alternative medicine (CAM), which is used by an estimated 35% to 69% of dermatology patients over their lifetimes (level of evidence: 3a) ( Pearl 4.1 ). One facet of CAM is nutritional supplementation. A nutraceutical is any food substance (vitamin, mineral, proprietary blend, genetically engineered food, or herbal product) that “provides medical or health benefits, including the prevention and treatment of disease” (level of evidence: 5). The FDA does not regulate nutraceuticals as it does drugs, so there are no premarket safety and efficacy studies. Therefore nutraceutical companies are not permitted to market claims that their product can diagnose, prevent, treat, or cure disease (level of evidence: 5). If a company does make a “structure-function” claim (e.g., saw palmetto cures androgenic alopecia), then the product must carry a disclaimer that it was not reviewed by the FDA. This may be confusing to patients, who often read about conflicting associations between nutrients and diseases reported in the media. A systematic review found that 75% of the risk estimates in studies claiming associations between foods and cancer had either weak or no statistical significance (level of evidence: 3a). Providers must bridge this knowledge gap and offer factual advice on nutraceutical risks and benefits.
A dearth of effective and well-tolerated therapies for androgenetic alopecia may account for the use of complementary and alternative medicine.
Indications and patient selection
Vitamin and mineral supplementation is indicated in any patient complaining of hair loss with a documented deficiency ( Pearl 4.2 ). Certain populations are at higher risk for deficiencies, and a thorough history of diet, lifestyle, use of supplements, and current medications should be sought in all cases ( Pearl 4.3 ). If responses raise concern for a vitamin or mineral deficiency, then laboratory testing may be pursued to determine whether supplementation is needed.
Vitamin and mineral supplementation is indicated in any patient reporting hair loss who has a documented deficiency.
Certain patient populations are at increased risk of vitamin and mineral deficiency.
A large population at risk for nutritional deficiency is the elderly. More than 60% of senior adults have been found to have vitamin and mineral intakes lower than the estimated requirements (level of evidence: 2c). Malnourishment in this population is multifactorial, and causes include decreased sensory reception in smell and taste of foods, impaired ability to chew and swallow food, reduced nutrient absorption as a result of medications, and higher rates of dementia, depression, and social isolation (level of evidence: 5).
Certain medical conditions are risk factors for nutritional deficiencies. Malabsorption in patients with pancreatic or biliary disease, Crohn’s disease, celiac disease, amyloidosis, parasitic infections, scleroderma, alcohol abuse disorder, or a history of bariatric weight-loss surgery renders them at particular risk for deficiencies of fat-soluble vitamins A, D, E, and K (level of evidence: 5). Patients with ongoing blood loss from menorrhagia (level of evidence: 2b), inflammatory bowel disease (level of evidence: 5), or colon cancer (level of evidence: 2c) are at risk for iron deficiency. Iron deficiency may also be seen in pregnant women (level of evidence: 5).
Pregnancy (level of evidence: 3b), lactation, and alcohol use disorder (level of evidence: 5) may lead to biotin deficiency, while burns, sickle cell disease, collagen vascular diseases, and chronic diarrhea can cause zinc deficiency (level of evidence: 5). Lastly, patients on hemodialysis are at risk for selenium deficiency caused by its removal of selenium from the blood (level of evidence: 2b).
Dietary practices have been found to be associated with specific deficiencies. Vegetarians are at higher risk for iron and zinc deficiencies, because plant-based sources of those nutrients have less bioavailability than their animal-based sources (level of evidence: 5). Furthermore, phytates, such as those found in cereals, whole grain bread, and legumes, are prevalent in vegetarian diets and are known to chelate zinc, making it unavailable for absorption (level of evidence: 5). Overconsumption of raw egg whites can lead to biotin deficiency, because eggs contain a protein component called avidin that binds to biotin and renders it nonfunctional (level of evidence: 5).
Various medications can impair the absorption of nutrients. Proton pump inhibitors (e.g., omeprazole) decrease iron absorption by reducing the acidic environment of the stomach that is necessary to transport iron across the intestinal lumen (level of evidence: 5). Diuretics, valproic acid, and penicillamine lower zinc levels (level of evidence: 5), , while isotretinoin (level of evidence: 1b) and valproic acid (level of evidence: 1b) lower biotin levels.
Geographic location is yet another consideration. Those with inadequate sun exposure are at risk for vitamin D deficiency (level of evidence: 5), while those living in places with low selenium soil content, such as Tibet, Siberia, and parts of China, are at risk for selenium deficiency (level of evidence: 5).
Guidelines for laboratory testing of vitamins and minerals have not been established. In 2014, the U.S. Preventive Services Task Force (USPSTF) reviewed the utility of screening for vitamin D deficiency and found that there was insufficient evidence to assess the pros and cons of screening asymptomatic individuals (level of evidence: 5). There are currently no laboratory testing recommendations for patients with AGA. Laboratory testing itself is not perfect. For example, biotin levels in women reporting hair loss have been shown to fluctuate up to 100% in 1 day (level of evidence: 2b). Additionally, laboratory testing is not available for some ingredients of nutraceuticals, as they are not known to be essential for bodily function. Without a documented deficiency, the decision to start a nutraceutical rests on the balance between clinical trial evidence and side effect profile.
Expected outcomes
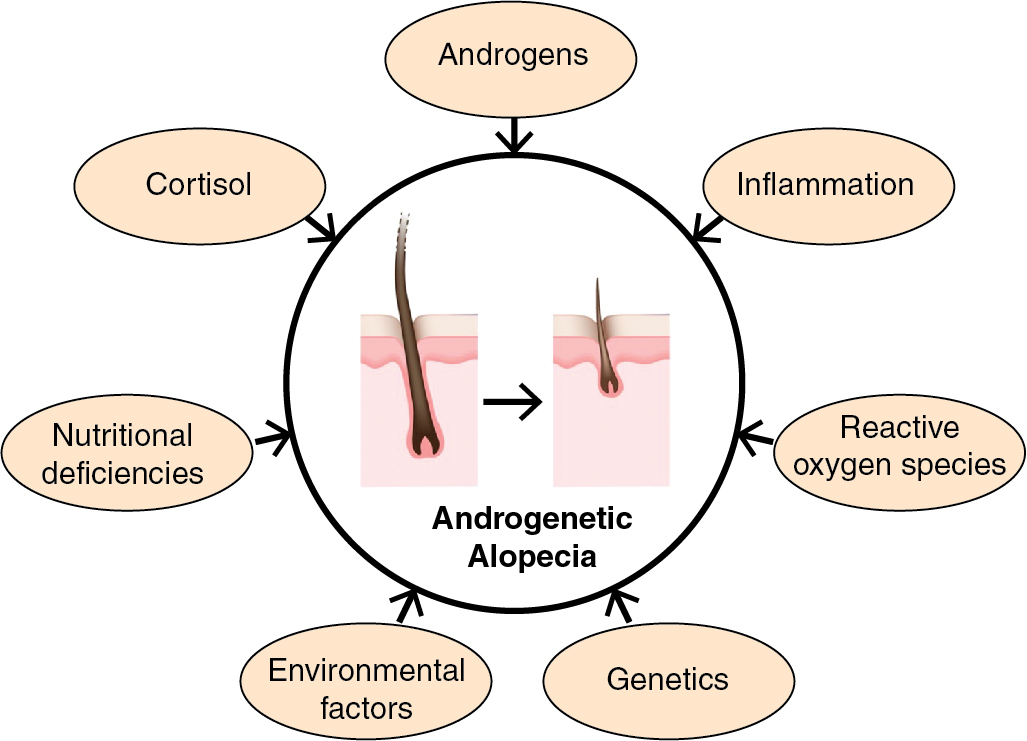
AGA is the result of progressive miniaturization of terminal hair follicles on the scalp ( Fig. 4.1 ) (level of evidence: 4). Miniaturization occurs as the hair follicle cycles from the anagen (growth) phase into the catagen (regression) phase, and subsequently the telogen (resting) phase ( Fig. 4.2 ). During the transition, hair shafts become thinner in diameter and hair bulbs move more superficially from the subcutis to the dermis. It is hypothesized that this process is driven by a combination of hormones, stress, genetics, and environmental exposures ( Fig. 4.3 ) (level of evidence: 5). Accordingly, nutraceuticals have been studied on molecular levels for their antiandrogenic and anti-inflammatory effects, which would theoretically increase the number and duration of terminal scalp hair follicles in the anagen phase.
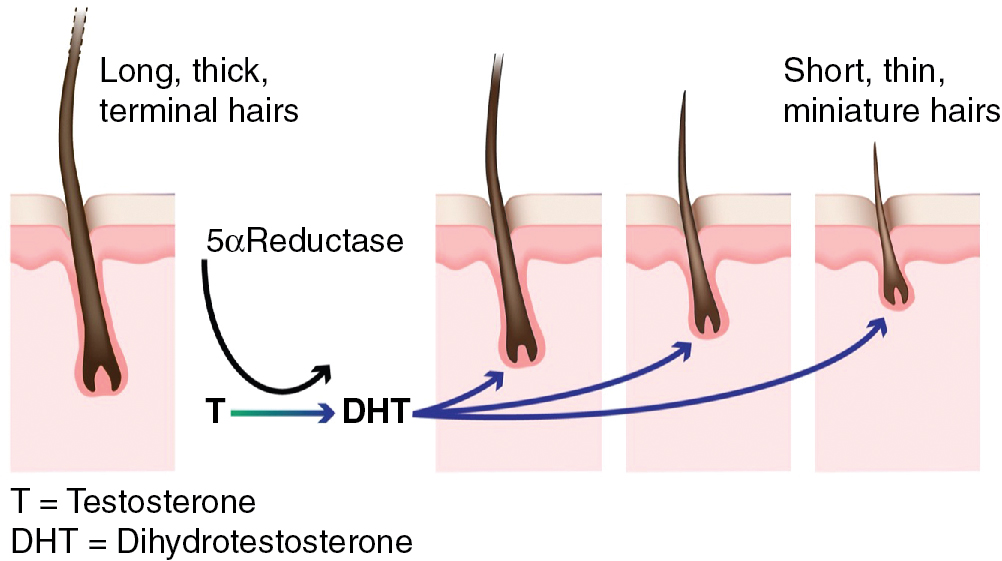
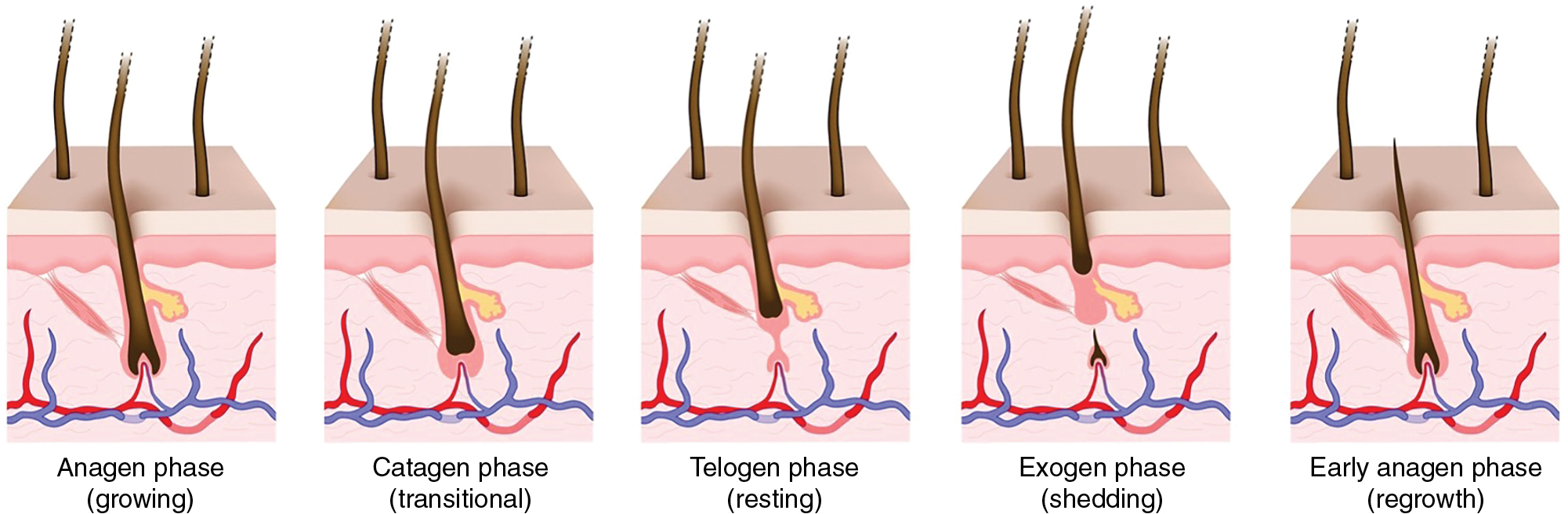
If the treatment of AGA involves prolonging the anagen phase and reversing miniaturization, one should expect to see an increase in the percentage of hair follicles in the anagen phase, hair shaft diameter, number of terminal hairs, and hair fullness. Studies have shown this to be true with topical minoxidil, which leads to a greater proportion of hairs in the anagen phase (level of evidence: 5) and increased hair counts (vellus, terminal, and total) (level of evidence: 1b). Nutraceutical studies have evaluated similar endpoints to capture the reversal of miniaturization; they have included objective measures of hair-shaft diameter and hair counts (vellus, terminal, and total hairs) and subjective measures of reviewer and patient perceptions of hair fullness, thickness, and density.
Many nutraceutical studies have evaluated endpoints between 3 and 12 months of treatment. This variability may be in part caused by the lack of studies on the duration of hair regrowth in in vivo human hairs (level of evidence: 5). Knowledge of the duration of hair follicle cycles has come from mouse xenograft models, which have shown that donor human catagen follicles will progress to the anagen phase in recipient mice somewhere between 60 and 90 days. , Two randomized, double-blind, placebo-controlled studies on the proprietary nutraceuticals Nutrafol (level of evidence: 1b) and Viviscal (level of evidence: 1b) both measured results after 90 and 180 days of treatment. This is similar to prior studies on topical minoxidil, which assessed endpoints at 16 weeks (112 days) and 24 weeks (168 days) (level of evidence: 1b). Although it is known that discontinuation of minoxidil does not maintain treatment response (level of evidence: 1b), , studies on nutraceuticals have not assessed the duration of effects once patients stop treatment. There is a need for more studies on optimal dosing and length of nutraceutical supplementation.
Treatment technique/best practices ( Pearl 4.4 )
Ashwagandha
Ashwagandha, also known as Withania somnifera , is a botanical herb long thought to have a role in maintaining homeostasis by modulating the stress response ( Fig. 4.4 ). Daily intake has been shown to significantly reduce circulating levels of stress hormones. An 8-week, double-blind, placebo-controlled, randomized trial demonstrated that daily intake of ashwagandha significantly reduced serum cortisol levels in healthy, stressed adults, especially at higher doses (600 mg/day > 250 mg/day) (level of evidence: 1b). Study participants also reported decreased levels of stress and anxiety, and increased quality of sleep. Similar results were found in a 60-day, double-blind, placebo-controlled, randomized trial that demonstrated reduction in both serum cortisol and dehydroepiandrosterone-sulfate (DHEA-S) levels in healthy adults taking 240 mg of ashwagandha daily (level of evidence: 1b). However, compared with placebo, an increase in testosterone was found in young men undergoing resistance training supplemented with 300 mg of ashwagandha twice daily (level of evidence: 1b). Increased serum testosterone levels could theoretically initiate, or hasten, AGA.
Routine vitamin and mineral supplementation for all patients with androgenetic and other forms of alopecia is not advisable.
Ashwagandha is an ingredient in Nutrafol, a proprietary blend of supplements that has been shown to stimulate hair growth. It is unclear if ashwagandha contributes to this observed effect given that no clinical trial has evaluated the direct effect of ashwagandha on hair growth.
Biotin
Biotin is a water-soluble B-complex vitamin. It serves as a cofactor for enzymes involved in mitochondrial function of hair follicles and other metabolic functions, gene regulation, and cell signaling (level of evidence: 5). , Biotin is often given as a supplement to patients with hair loss, because hereditary deficiency of biotin is associated with alopecia; it is also inexpensive and readily available. One retrospective cohort study found that 38% of healthy women complaining of hair loss had low levels of biotin (level of evidence: 2b); however, many of these women had concurrent seborrheic dermatitis or a risk factor for biotin deficiency (e.g., inflammatory bowel disease, isotretinoin use, antibiotic treatment). Additionally, the studied population had not been diagnosed with AGA. A different study on biotin and zinc levels in male patients with AGA revealed low levels of both compared with matched controls, although biotin levels did not correlate with age or disease duration (level of evidence: 2b). Furthermore, biotin levels fluctuate daily, and there are multiple compounding factors in their measurements (level of evidence: 5). Although biotin has been shown to promote hair growth in biotin-deficient patients (level of evidence: 4), there are no randomized, controlled trials demonstrating a beneficial effect of biotin in AGA or other types of alopecia.
Curcumin
Curcumin is the active ingredient of turmeric, a naturally occurring botanical in the ginger family ( Fig. 4.5 ). Curcumin has been used in Ayurvedic medicine for centuries due to its anti-inflammatory effects, although it also has antioxidant, antimicrobial, antineoplastic and antiandrogenic properties (level of evidence: 5). Although it can be made as an individual oral supplement, it is poorly absorbed and rapidly metabolized. The botanical piperine, which is found in black pepper, is added to commercially available supplements with curcumin to drastically increase bioavailability (level of evidence: 4).
There are no clinical trials on oral curcumin for hair loss. However, a 5% topical extract has been studied with and without 5% minoxidil in comparison to placebo (level of evidence: 1b). Combined therapy demonstrated statistically significant improvements in hair count, regrowth, and shedding, while treatment with topical curcumin alone did not yield significant improvements. This suggests that topical curcumin increases the penetration of topical minoxidil, which has been verified in ex vivo foreskin models (level of evidence: 5). Although supportive evidence for curcumin alone is lacking, it may be beneficial in enhancing treatment with topical minoxidil.
Equisetum
Equisetum , or horsetail, is an herb that has long been used for its suspected anti-inflammatory, antioxidant, and antimicrobial properties ( Fig. 4.6 ). It has been shown to inhibit 5-α-reductase in vitro , theoretically slowing the progression of AGA (level of evidence: 5). Equisetum is believed to be effective for hair growth because of its high concentration of silicon. Physiologic concentrations of orthosilicic acid (the bioavailable form of silicon) have been shown to stimulate synthesis of type I collagen in in vitro human osteoblast-like cells (level of evidence: 5). It is hypothesized that by activating fibroblasts, orthosilicic acid induces a favorable cytokine milieu for hair growth. A double-blind, placebo-controlled, randomized trial assessed the effects of 10 mg of choline-stabilized orthosilicic acid daily in women with fine hair (level of evidence: 1b). After 9 months, the treatment group had significantly thicker hair (as measured by the cross-sectional area) and a positive effect on tensile strength. Although these results are promising, the supplement has not been tested on patients diagnosed with AGA.
Iron
Iron is an essential element, which means it must be obtained from the diet. It plays many crucial roles in bodily functions, including red blood cell production, oxygen transport, mitochondrial respiration, DNA replication and repair, immune function, and cell signaling (level of evidence: 5). Mice with a TMPRSS6 protein (matriptase-2) mutation, which causes poor iron absorption and iron deficiency, experience a loss of body hair (level of evidence: 5). It has been shown that giving these mice iron supplements can reverse the iron deficiency and restore hair growth. However, results of studies on iron and hair loss in humans are conflicting, and often AGA and telogen effluvium (TE) are not distinguished (level of evidence: 5). One study on ferritin levels in women with different types of nonscarring hair loss revealed that those with AGA and alopecia areata (AA) had significantly lower levels than controls (level of evidence: 2b). Another study did not find any significant decrease in ferritin levels in women with AGA compared with those with TE and controls (level of evidence: 2b).
Marine proteins
Marine proteins are a group of fish, mollusk, and marine plant species that have been isolated for the purposes of human supplementation. The first reported use of marine proteins for AGA came from a study on men in Finland comparing marine proteins mixed with equisetum (Viviscal) to a fish extract of similar composition ( Fig. 4.7 ) (level of evidence: 1b). The marine protein group demonstrated a lack of hair loss in all participants after 2 months, a significant increase in nonvellus hair counts, and less inflammation and improvement in follicular size on biopsies at 6 months. Although multiple randomized controlled trials have also shown that Viviscal leads to clinical and histologic improvement in hair growth, many have been marred by the inclusion of different types of hair loss, the lack of placebo groups, small study sizes, and follow-up times of less than 4 months (level of evidence: 5, 1b, 1b). It should be noted that Viviscal is a proprietary blend of shark cartilage, oyster powder, apple extract powder, vitamin C, L-cystine, L-methionine, and biotin, so conclusions about the efficacy of marine proteins alone cannot be made. Other proprietary supplements contain marine proteins; Nourkin has fractionated fish extract with specific lectican proteoglycans, and Nutrafol has marine collagen from cod fish scales.
Pumpkin seed oil
Pumpkins are members of the squash family and are native to North America ( Fig 4.8 ). Pumpkin seed oil contains varying amounts of magnesium, potassium, calcium, sodium, selenium, iron, polyunsaturated fatty acids, tocopherols, and sterols (level of evidence: 5), , which lend antioxidant and anti-inflammatory properties. Wounded rats treated with pumpkin seed oil revealed full reepithelialization with reappearance of skin appendages and well organized collagen fibers, suggesting a mechanism for hair regrowth (level of evidence: 5). In a clinical study of 76 men with AGA treated with a supplement containing 400 mg of pumpkin seed oil, hair counts were significantly improved at 12 and 24 weeks compared with placebo, but hair diameters improved in both the treated and placebo groups (level of evidence: 1b). However, the product used (Octa Sabal Plus) also contains mixed vegetable powder, octacosanol, corn silk, evening primrose powder, tomato powder, and red clover powder, so it is not known which ingredient, or combination of ingredients, was responsible for the observed results.

Saw palmetto
Saw palmetto, or Serenoa repens , is a botanical extract made from the berries of the American dwarf tree, which can be found in the southeastern United States ( Fig. 4.9 ) (level of evidence: 5). It has been widely used in the treatment of benign prostatic hyperplasia as a result of is antiandrogenic effects. Saw palmetto is a competitive, nonselective inhibitor of 5-alpha reductase (level of evidence: 5) and inhibits 50% of dihydrotestosterone from binding to nuclear androgen receptors in cultured human foreskin fibroblasts (level of evidence: 5). There has been some evidence in clinical trials that saw palmetto is efficacious in treating AGA. A small pilot study showed that 200 mg of saw palmetto daily resulted in “improvement,” as determined by blinded investigative staff in 6 of 10 healthy male subjects with mild-moderate AGA (60%, compared with 11% of subjects given placebo) (level of evidence: 1b). A larger study compared 320 mg of saw palmetto daily to 1 mg of finasteride daily in males with mild to moderate AGA for 24 months, finding significantly higher hair density scores for both saw palmetto (38%) and finasteride (68%) (level of evidence: 1b). Additionally, saw palmetto “stabilized” AGA in 52% of cases, while 10% of the subjects continued to have progressive hair loss. The study showed that both saw palmetto and finasteride are effective treatments, but that finasteride was superior, especially for men with more severe AGA.