Introduction
Over the past decades, significant advances in our understanding of the mechanisms underpinning the aging process have coincided with immense advances in the tools and technologies available to address these changes. No longer is the face seen as a monolithic entity that needs to be pulled or lifted. Changes in the bone structure, fat compartments, myofascial layers, and skin all contribute to restoring esthetic beauty and harmony. Rather than simply effacing lines, practitioners need to be attentive to the subtleties such as facial dynamic function, curvatures, transitions, and congruence. While surgery is effective in restoring many of the elements of facial aging, increasingly, patients seek interventions with less risk and downtime. It is estimated that by 2028, the US demand for minimally invasive treatment for facial rejuvenation will surpass $39 billion.
Facial rejuvenation procedures have been traditionally thought of as those that address the skin (texture, color, tone, dynamic function) and those that primarily address structure and volume. Increasingly the frontier between these approaches is effaced as the interdependence of skin appearance and volume becomes evident.
Botulinum Toxin
Initially used for the temporary treatment of strabismus, botulinum toxin (BoNT)-A has become a mainstay tool to address the aging face. BoNT is derived from Clostridium botulinum . Botulinum toxin blocks neuromuscular transmission by binding to presynaptic receptor sites on motor nerve terminals and inhibiting the release of acetylcholine. Initially synthetized as a single 150 kD chain, it is then cleaved into heavy and light chain molecule held by a disulfide bond. The cholinergic specificity of the heavy chain allows for binding to the presynaptic receptor while the proteolytic light chain acts as a zinc endopeptidase. When injected intramuscularly at therapeutic doses, BoNT produces temporary chemical denervation of the muscle, resulting in decrease in muscle activity. The paralytic effects of the toxin are dose- and site-related. The initial effects usually occur within 2–3 days and peak approximately 1 week after treatment. Effects generally last around 3 months, with repeated injections extending the clinical effect.
In 1992, the Carruthers and Carruthers published the first report of the cosmetic use of BoNT-A for the treatment of glabellar frown lines, which was approved for cosmetic usage in the USA in 2002.
Formulations
To date, the FDA has approved three distinct formulations of BoNT-A for both therapeutic and cosmetic purposes, and one formulation of BoNT-B for therapeutic use only. The original formulation of BoNT-A, onabotulinumtoxin-A (BOTOX/BOTOX Cosmetic/Vistabel/vistabex; Allergan, CA, USA), has been joined by abobotulinumtoxin-A (Dysport or Azzalure; Galderma, TX, USA), and icobotulinumtoxin-A (Xeomin/NT-201; Merz Pharma GmBH & Co., Frankfurt, Germany). Only one formulation of BoNT-B-rimabotulinumtoxinB (Myobloc/Neurobloc; Solstice Neurosciences, CA, USA) is available in North America. All products differ in their pharmokinetics and therefore clinical doses vary and are not interchangeable.
Cosmetic Uses of BoNT-A
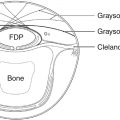
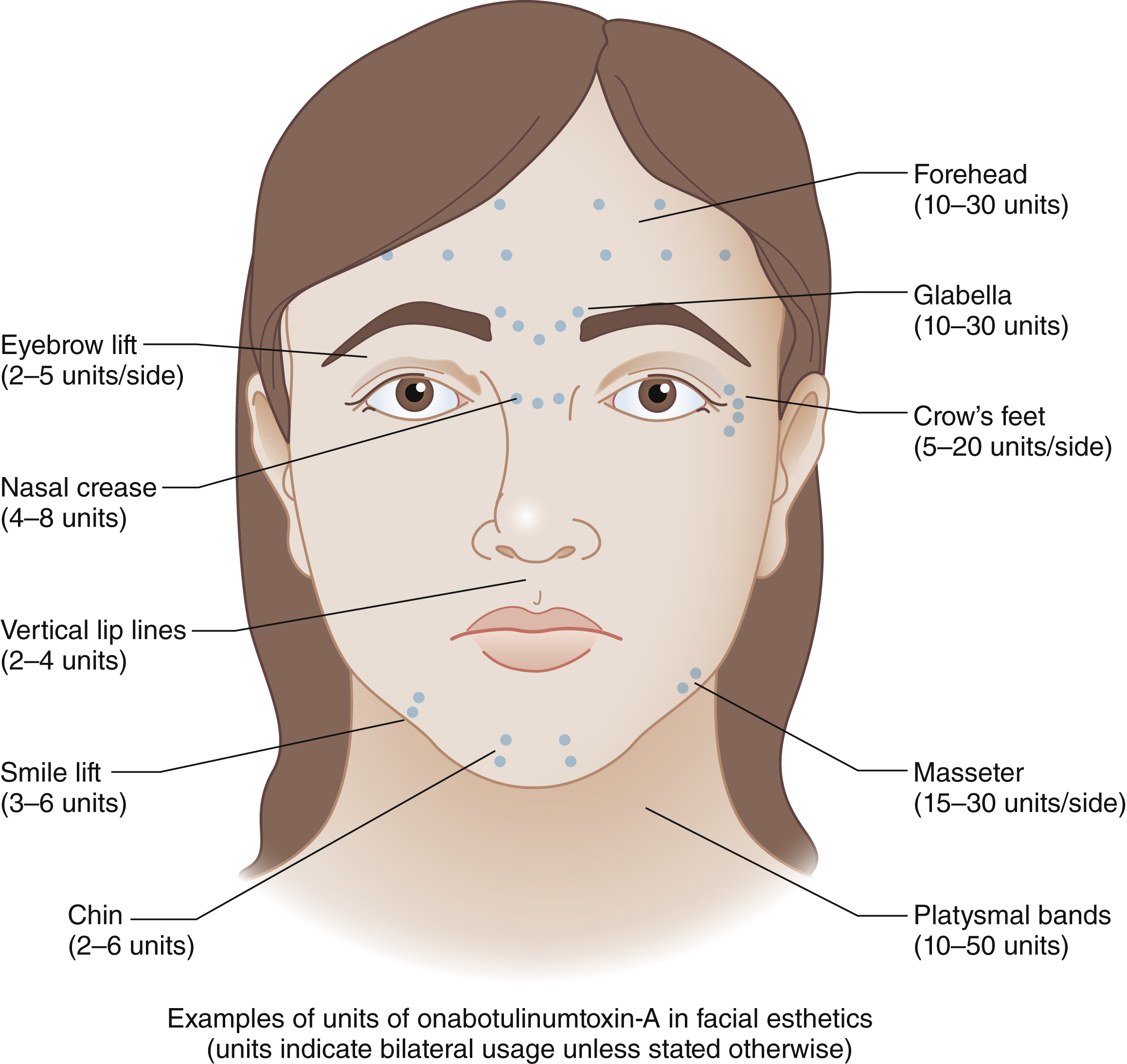
Initially approved only for injection into the glabellar rhytids, BoNT now is used for dozens of off-label applications on the face ( Fig. 61.1 ). These include controlled paresis of hyperkinetic lines of the face and neck, elevation of various facial subunits by altering muscle equilibrium between depressor and elevator muscles, and reestablishing facial symmetry by selective blockade of muscles of facial expression. BoNT is also increasingly used perioperatively to help with scar management or to stabilize brow position following brow elevation.
A number of technical factors can contribute to a comfortable and more predictable experience:
- •
Topical anesthetic is applied 30 minutes prior to the procedure (the authors prefer a triple anesthetic preparation of lidocaine, tetracaine, and benzocaine).
- •
Ice or cold compresses is applied immediately prior to injection.
- •
If a marker is used, we prefer removable white ink markers (Viscot, NJ).
- •
A 32G (or 33G) short needle is used on a hubless 0.5 syringe. Larger syringes provide less tactile feedback to the injector and can be more uncomfortable to patients.
- •
The depth of penetration of the needle follows the esthetic goals and specific muscle anatomy. When injecting the corrugators, for instance, the depth of penetration is more superficial laterally and becomes deeper medially.
- •
Injection is always made with low pressure and in small aliquots. It is preferable to inject multiple times than to inject a large quantity of fluid in a single push.
- •
It is preferable to distribute the toxin across several syringes in order to maintain tip sharpness and thus diminish patient discomfort.
- •
Where a large quantity of toxin is required (e.g., in men with a heavy corrugator complex), it may be preferable to increase the concentration of the toxin to diminish diffusion. In most instances with BoNT, the authors create a 2-mL solution of 5 u/0.1 mL for routine cases and 10 u/0.1 mL for cases where a higher concentration is required.
- •
Not all toxins have the same clinical characteristics. Abobotulinumtoxin-A for instance appears to have a wider diffusion circle. This can be advantageous in the forehead where multiple entries are often required to achieve meaningful reduction of rhytides. On the other hand, where targeting a specific muscle, highly concentrated onabotulinumtoxin-A may yield more reliable results.
- •
The areas injected are carefully mapped and the patient is seen between 7 and 14 days following injection for follow-up.
Adverse Effects and Complications
While adverse effects of BoNT are rare, they include watery, dry or crossed eyes. It may also include an incompetent mouth, the inability to whistle, dysphagia, and neck weakness. Most dissatisfaction from BoNT stems from unexpected changes in facial appearance or expression. Common complaints in include:
- •
Ptosis of the eyelid from injection into the forehead or corrugator complex.
- •
Ptosis of the brow from forehead injection.
- •
Overarching eyebrows from injection of the corrugator complex.
- •
Lower lid festoon formation from injection of crow’s feet and secondary loss of orbicularis tone.
- •
Changes in phonation or smile from perioral injection.
While these adverse effects are not entirely avoidable, they could be minimized by gaining better knowledge of the underlying facial anatomy and physiology as well as paying close attention to individual anatomic variability.
Soft Tissue Fillers
The recent decades have seen substantial increase in the use of dermal fillers to address the aging face. Fillers have been used to augment the facial skeleton, soft tissue, as well as diminish fine lines and wrinkles. Increasingly fillers are used not only to address age-related changes in the face, but also as a means to enhance appearance in youth. While the injection of autologous fat continues to be a mainstay treatment of the aging face, this chapter concentrates primarily on the use of synthetic fillers, specifically fillers based on hyaluronic acid, calcium hydroxyapatite/carboxymethyl cellulose and poly-lactic acid.
Historic Context
The era of absorbable dermal fillers was ushered in in the latter part of the 20th century with the introduction of collagen fillers. Initially made of bovine collagen, fillers based on human collagen became popular in the late 1990s. Products such as Zyderm and Zyplast that were animal-derived collagen required prior skin testing, with hypersensitivity reactions observed in as much as 10% of patients. In 2003 the US Food and Drug Administration approved the use of Restylane (Galderma), a hyaluronic-based product as a class III device to treat facial rhytids. Dozens of products have since been introduced to address a variety of facial volumetric needs.
Rheological Properties
Rheology refers to the physics of deformation and flow of materials, typically in a state between solid and liquid. An understanding of rheological properties of various injectable fillers is critical to the appreciation of their clinical behavior. G′ or elastic modulus refers to the resistance of the material to an applied strain and is commonly used as a measure of hardness or softness of a given material. G′′, or the viscous modulus, refers to the energy dissipation due to friction. Other valuable measurements include resistance to compression and maximum water uptake. These properties, when combined with experimental data from human or animal models, provide valuable insight with regard to the actual in vivo behavior of various materials. It is important to note that physical properties of the filler may not always correlate with their performance in the face over time.
Nonbiodegradable Fillers
Liquid Injectable Silicone (Purified Polydimethylsiloxane)
Primarily used in vitreoretinal surgery, liquid silicone has been used as a facial filler on an off-label basis. Silicone oil induces neocollagenesis through fibroplasia in addition to direct volume restoration. While the permanence of silicone is a highly desirable effect, the possibility of late granuloma formation and the inability to reverse the product make it a less desirable product compared to alternative options on the market. Furthermore, long-term studies are lacking on the safety and efficacy of subdermal silicone. In certain instances where the affected tissue could be excised if needed – such as a limited depressed scar – the injection of silicone may provide a valuable tool for near permanent correction of the defect without surgical intervention. However, such benefits must be weighed against significant risks of granulomas, allergic reaction, and even embolisms.
Polymethylmethacrylate (PMMA)
PMMA (ArteFill; Suneva Medical Inc., CA, USA) was approved by the FDA in 2006 for the correction of nasolabial folds. It is a third-generation filler comprising 20% PMMA microspheres and 80% bovine collagen. After implantation, the carrier solution dissipates within 1–3 months, leaving behind the nonresorbable microspheres that induce fibroplasia and become encapsulated by collagen. Since it contains bovine collagen it also requires skin testing to prevent an allergic reaction. Similar to silicone, late nodule formations are observed and can be difficult to treat in highly visible areas of the face.
Biodegradable Fillers
Calcium Hydroxyapatite (CaHA) Microspheres
CaHA microspheres (RADIESSE; Merz Aesthetics, Inc., CA, USA) comprise of biocompatible smooth CaHA spherical particles that are identical in composition to bone material and suspended in an aqueous carboxymethylcellulose gel carrier. The microspheres induce a histolytic and fibroblastic response, stimulating the production of collagen around the implant while the gel carrier is absorbed. CaHA generally lasts about 15 months before the microspheres are broken down into calcium and phosphate ions and excreted. Although CaHA has a great long-term safety profile, nodules and foreign body reactions have been reported. It has both on-label uses such as the treatment of nasolabial folds and HIV-related facial lipoatrophy as well as off-label uses for cheek augmentation and contouring. One advantage of CaHA is its ability to retain its shape over time which is why it is often used for enhancement of the facial skeleton. Unlike HA fillers, however, there is no mechanism to reverse the injection of CaHA. If injected superficially, CaHA can project as a yellow discoloration under the skin. This is why its usage is not recommended in superficial zones of the eyelid and lips.
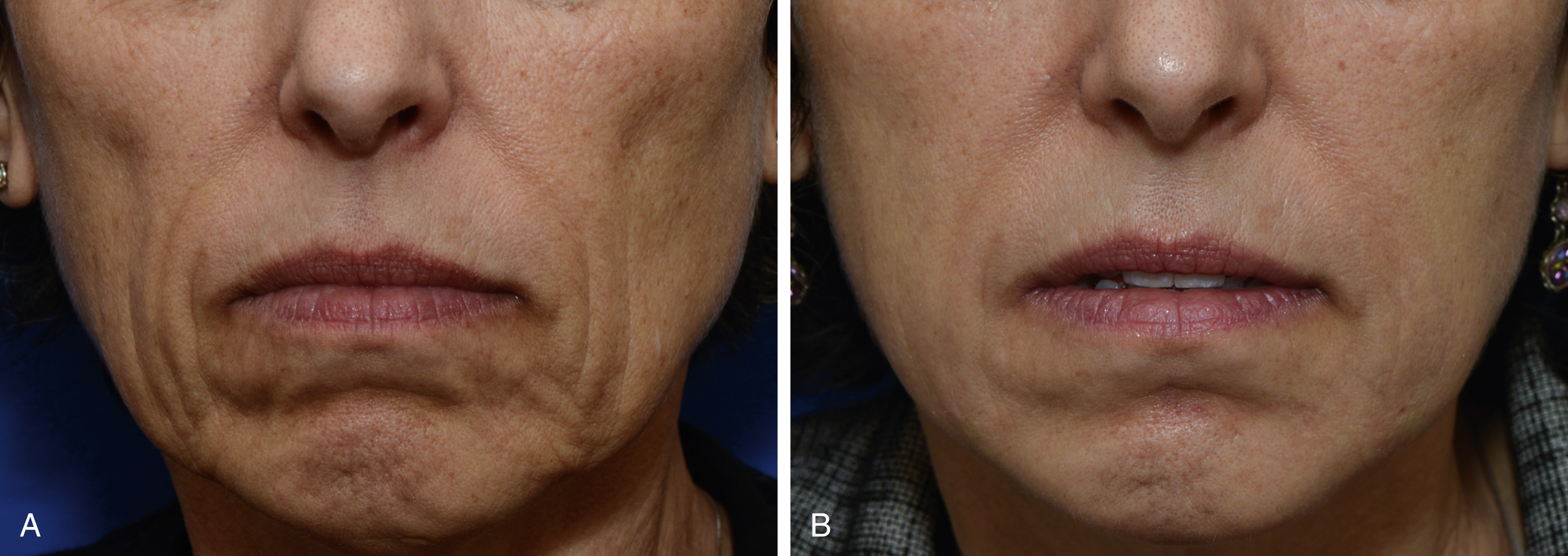
Poly-L-lactic acid (PLLA)
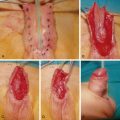
PLLA (Sculptra; Galderma) is a biocompatible and biodegradable synthetic polymer from the alpha-hydroxy-acid (AHA) family. When injected into the skin, PLLA stimulates fibroblasts to produce collagen, resulting in an increase in volume ( Fig. 61.2 ). The particles are eventually metabolized to lactic acid monomers that are further broken down by the body. Most side effects are due to the injection procedure, although late-onset papules and nodules have been reported. PLLA must be diluted prior to injection with sterile preserved water to allow adequate time for polymer hydration, and patients require a series of three injections over several months. Patients must be informed that volumetric change occurs with time and is often not observed with the first injection. Below are other practical considerations to take into account when injecting PLLA:
- •
Preparation of PLLA 5–7 days before the procedure may improve hydration and injection characteristics.
- •
The patient should be injected in a sitting position.
- •
A 25G 1–1/2-in needle is preferable as it minimizes particle blockage.
- •
Individual 1-mL Leuer lock syringes are preferable to larger syringes both because of flow control and precision.
- •
It may be valuable to slightly bend the needle with the bevel facing up for better control.
- •
Multiple cross-hatch passages are recommended to avoid overfilling a particular spot and to avoid granulomas.
- •
Small aliquots of less than 0.1 mL are placed per passage.
- •
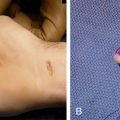
PLLA is ideal for large areas of fat loss and is best avoided in areas with thin skin such as the periocular region ( Fig. 61.3 ).
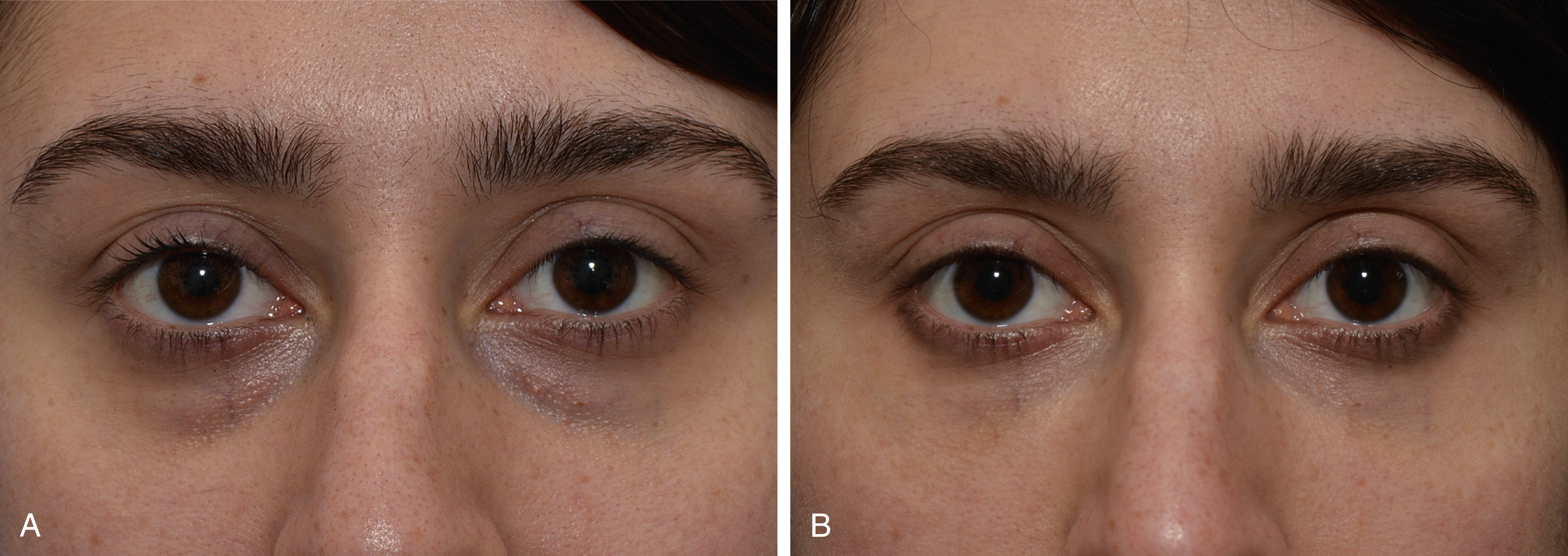
Fig. 61.3
Result of injection of filler to the periocular hollows, (A) before, (B) after.
- •
We adhere to the “rule of 5s:” massage all the areas of injection 5 times a day for 5 minutes and for 5 days to improve even distribution and avoid granuloma formation.
Hyaluronic Acids (HA)
Currently the most common form of filler, HA devices are expected to reach a market exceeding $6 billion by 2025. HA is a naturally occurring polymer of disaccharides linked via alternating beta-glycosidic bonds and one of the chief components of the extracellular matrix in eukaryotic cells. It is thus significantly less immunogenic compared to previous human- or bovin-derived collagen preparations. A chief component of HA is a long, hydrophilic, cross-linked glycosaminoglycan that acts as a scaffold for collagen and elastin and contributes to cell proliferation and migration, as well as tissue repair. The HA molecule is stabilized through cross-linking with hydroxyl groups – allowing it to resist digestion by hyaluronidase – a characteristic that largely determines its clinical behavior and longevity in the body. All HA fillers are unique in terms of concentration of HA, cross-linking, particle size, and manufacturing processes.
When injected into the skin, HA combines with the body’s own HA and binds to water, increasing skin turgor, elasticity, and volume for a period of 4–12 months, depending on the product used and the location placed. Most formulations are approved for the correction of facial rhytids and nasolabial folds but are also used off-label for volume replacement in the cheeks and midface. Side effects are generally mild and transient. More troublesome complications are rare but include granulomatous reactions and vascular ischemic complications.
Hyaluronic Acid Preparations
The choice of a particular form of HA largely depends on the esthetic concern of the patient. Uses of HA include volume augmentation of the bony support of the face (e.g., malar area or mandible), replenishment of atrophic zones of the face (e.g., temple and cheek), enhancement or effacement of discreet lines of the face (nasolabial fold, vermilion line), elevation of the facial soft tissue (e.g., jowls) or filling of scars (e.g., acne). In many instances more than one goal is achieved through 3-dimensional facial contouring (e.g., tear trough area). Below are some general considerations to optimize HA injections.
- •
The choice of the filler must be driven by the goal at hand. As a general rule, fillers with good dispersion properties (e.g., Restylane Silk; Galderma) are best used for superficial filling while those with better structural properties (e.g., Juvéderm Voluma; Allergan) are better at addressing structural support. The author’s preferences are shown in Fig. 61.4 .