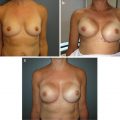
Fig. 16.1
Preoperative asymmetry after right lumpectomy and radiation
As the use of nipple-sparing mastectomy is becoming more widespread, the selection criteria have been broadening significantly. Most surgical practices are shifting toward including more complex patients including patients with macromastia , ptosis, tumors less than 2 cm from the nipple, older patients, patients with positive nodes, and those receiving neoadjuvant chemotherapy [2, 3]. A recent publication from Mayo Clinic showed that 7.8 % of their patients undergoing nipple-sparing mastectomy had received preoperative radiation therapy whereas it was previously considered a relative contraindication in their practice [2].
Patients who decide to save their nipple areolar complex have been shown to have a higher patient satisfaction when they undergo nipple-sparing mastectomy versus skin-sparing mastectom y [4]. Patients with previous radiation who have preoperative asymmetry and poor skin quality must be informed that they will potentially have persistent asymmetry postoperatively and that previously radiated skin carries a higher risk of postoperative complications such as infection, flap necrosis, high-riding nipple, asymmetry, and implant loss [5]. Careful patient selection , preoperative planning , meticulous technique , perioperative antibiotic coverage , and maintenance of the blood supply are important in order to reduce the risk of complications.
Pathophysiology of Radiation and Surgical implications
Radiation can obscure normal tissue planes, cause reduced tensile strength, decrease elasticity of the skin and impair wound healing [6, 7]. It can also cause inhibition of revascularization which results in reduced blood supply and subsequent tissue hypoxia. This can impair the normal inflammatory response to wound healing, which can in turn predispose the surgical site to bacterial invasion [6, 7].
Radiation treatment causes excitation and ionization of electrons, which results in direct damage to DNA as well as the production of free radicals. These free radicals can damage cell membranes, proteins, and DNA which ultimately causes apoptotic cell death.
The effects of radiation are divided into early and late effects. Early effects occur within the first 10 to 14 days after starting treatment. They are caused by vascular permeability, vessel wall edema and thrombosis, fibroblast injury, insufficient collagen production, inability to encourage maturation, reduced response to stimulatory effects of local growth factors, and reduced neovascularization. The late effects are seen more than 3 months after starting treatment and include atrophy, contraction, loss of vessels, fibrous tissue replacement, and damage to melanocytes, altering skin pigmentation [7]. In radiated tissue, TGF-beta receptors are upregulated and become chemotactic for mast cells, fibroblasts, monocytes, and macrophages. TGF beta 1 is considered a major stimulant of radiation fibrosis [6].
Impact of Time Interval Between Radiation and Surgery
Previously, many surgeons felt that the longer the time from the radiation, the longer the time the skin had to repair and recover and the fewer the complications . However, there has been insufficient data examining the effect of length of duration from radiation to complications to draw this conclusion [8]. In 2011, Khansa et al. found no relationship between time interval from radiation to final mastectomy and reconstruction outcome [9]. In the sarcoma literature, there was no correlation between preoperative radiation and time to surgery with wound complications in patients undergoing lower extremity resections [10]. Many have proposed that there may be a genetic predisposition in how a patient responds to radiation therapy [11].
Outcomes After Reconstruction in Previously Radiated Patients
Several studies have shown that the failure of reconstruction is much higher in patients who have been treated with previous radiation, at a rate of 19 % versus around 5 % in those who have never been radiated [12–14]. The surgical outcomes of nipple-sparing mastectomy in previously radiated patients are unpredictable due to the side effects of radiation on the skin. Several recent studies have reported on these decreased outcomes. A study in 2011 from Khansa et al. looked at 113 patients who had undergone breast conservation therapy followed by mastectomy with reconstruction. The overall complication rate in this group was 36.5 % compared to 27.1 % in those who had never had radiation. They ultimately found that the only statistically significant complication was an increased rate of mastectomy skin flap loss in breasts that had been radiated (12.4 % vs. 6.8 %, p = 0.024) [9].
The following year in 2012, Hirsh et al. reported on a series of 71 breasts from 66 patients that had undergone prior breast conservation therapy followed by mastectomy with tissue expander reconstruction. They found an overall rate of major complications requiring surgical intervention including loss of implant or reconstruction with autologous flap reconstruction of 28.2 %, with an 8.5 % rate of minor complications. The overall rate of successful completion of reconstruction with expander exchange for implant was 62 % [15].
Technical Surgical Aspects and Pearls
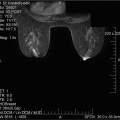
Patients with radiated skin do not tolerate even the slightest complication such as infection, extreme tension on skin flaps, hematoma, or ischemia. Since the outcomes may not be ideal and there is a higher rate of complications in previously radiated patients, appropriate patient selection, careful preoperative planning, and surgical technique are of utmost importance. Patient with smaller breasts, lack of severe ptosis, and minimal skin changes after radiation are more ideal for this procedure. It is important to work closely with the reconstructive surgeon during the preoperative planning stage. The imaging work up should include a mammogram and possible sonogram, as well as a preoperative MRI in order to assess the distance of the tumor to the skin and nipple areolar complex (Fig. 16.2a, b). This facilitates the planning of the surgical incision.
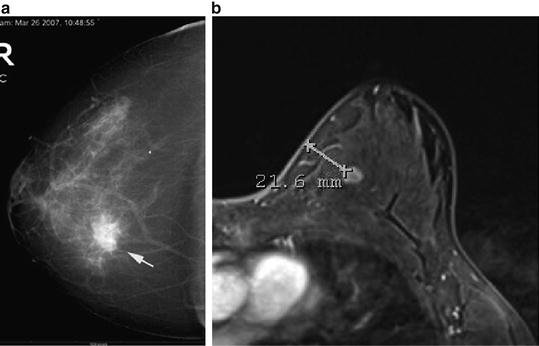

Fig. 16.2
(a) Mammogram showing location of cancer in relation to skin and nipple areolar complex. (b) MRI showing location of cancer in relation to skin
All patients who undergo breast reconstruction should receive perioperative antibiotics with gram-positive coverage. When technically feasible, an inframammary fold (IMF) incision is preferable as it has been associated with decreased complications (OR, 0.018). A large incision measuring at least 12 cm in length can be used for adequate exposure and decreased tension on the flaps [16]. In nonradiated patients, the IMF incision is usually the length of the width of the surgeon’s hand, or at least 9 cm, starting at the medial aspect of the areola and extending laterally. In a previously radiated patient, it is preferable to extend the incision at least 12 cm (Fig. 16.3a, b). If a previous lumpectomy scar is tethered, or the cancer is near the lumpectomy scar, then this skin may need to be excised and incorporated into the new incision, depending on its location.
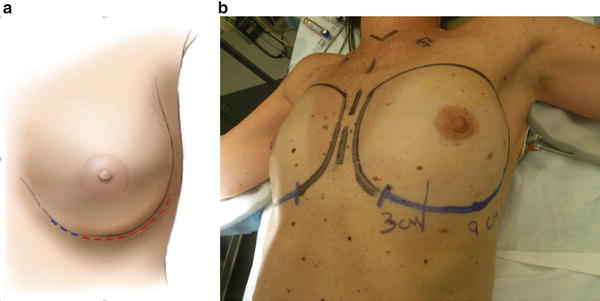

Fig. 16.3
(a) Nonradiated skin, perform approximately 10 cm incision length along IMF (red line), however, in radiated skin extend incision to at least 12 cm for maximal exposure and less flap tension (blue line). (b) Shows extension of IMF in a previously radiated patient for better exposure
Meticulous dissection is important for all patients undergoing nipple sparing mastectomy in order to preserve the blood supply to the flap as well as the nipple areolar complex. This is technically more challenging in patients undergoing an inframammary fold incision where the length of the flap is even longer and the watershed area inferior to the nipple is more compromised. The flap is made slightly thicker at the skin incision to prevent necrosis of the skin edges. It is important to identify and develop the layer between the anterior fascia of the breast and the patient’s subcutaneous layer. This anterior plane is a relatively avascular plane with only small vessels in the patient’s Cooper’s ligaments connecting the breast tissue to the skin. The boundaries of the mastectomy are the clavicle superiorly, the sternum medially, the latissimus laterally, and the sixth rib inferiorly. The posterior plane is between the pectoralis major muscle and the posterior aspect of the breast. We routinely remove the posterior fascia of the breast along with the specimen.
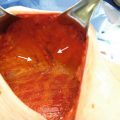
The largest blood supply to the breast comes from the second intercostal perforator off the internal mammary artery, followed by the lateral thoracic artery (Fig. 16.4). Preservation of the second intercostal artery is highly recommended in order to maintain perfusion to the flap (Fig. 16.5). We also recommend preserving the venous plexus in the subcutaneous layer to ensure adequate perfusion to the flap and prevent venous congestion.
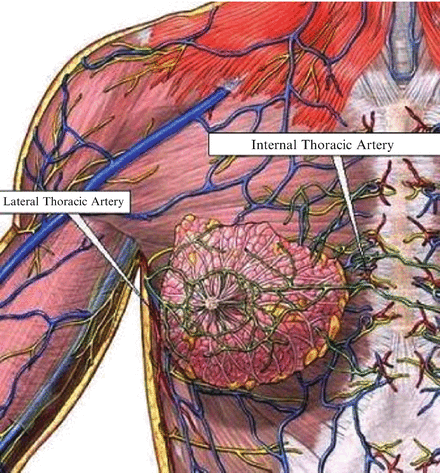
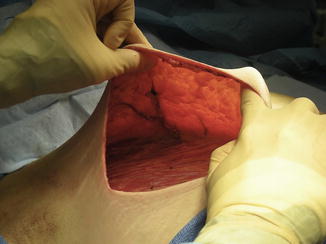

Fig. 16.4
Blood supply to breast

Fig. 16.5
Intraoperative photo shows largest blood supply to the breast, the second intercostal perforator
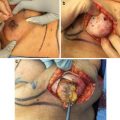
When operating on radiated patients, it is important to minimize tension on the flaps both intraoperatively as well as postoperatively. We use noncorrugated lighted retractors to assist with creation of the flaps, and periodically remove the retractors to alleviate tension from the flaps thus giving them a chance to reperfuse (Fig. 16.6). When performing the dissection behind the nipple, the assistant’s finger is used to provide gentle upward traction.
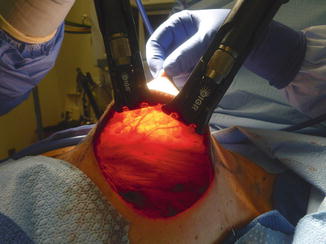

Fig. 16.6
Intraoperative photo shows extended incision along the IMF with good exposure and no tension on flaps
Oncologic Safety of NSM
Controversy has long surrounded performing NSM in patients with cancer fearing that retained breast tissue behind the nipple areolar complex may harbor a future breast cancer. However, several retrospective studies comparing local recurrence in skin sparing versus nipple sparing patients reveal a similar local recurrence rate in the two groups. A large study on 657 breasts in 428 patients undergoing nipple sparing mastectomy revealed a recurrence rate of 2.4 % after 3 years follow up [17]. In the most recent largest study of 982 patients undergoing NSM, ten patients had locoregional recurrences. However, none of these recurrences occurred in patients who had been treated with previous radiation at a mean follow up of 24 months [18]. A recent meta-analysis of 27 studies in 2013 showed an overall local regional recurrence rate of 2.8 % in patients undergoing NSM [19]. The largest study to date of nipple sparing mastectomy in radiated patients followed for 22 months revealed no local recurrences in 43 patients with previous radiation [12].
Outcomes and Associated Complications
The most common complications in patients who undergo nipple-sparing mastectomy and reconstruction after previous radiation include infection, flap necrosis, capsular contracture, fibrosis, asymmetry, nipple malposition, and a higher revision rate. Several studies in recent years have reported on complications and outcomes in this patient population (summarized in Table 16.1). Colwell et al. found that preoperative irradiation was a positive predictor for nipple necrosis with an odds ratio of 4.86 compared to patients who had no radiation [16]. In a large review of 318 patients undergoing nipple-sparing mastectomy, 20 were identified who had been treated with previous radiation [20]. This review found a 30.0 % rate of postoperative nipple-areola complex ischemia, compared to the group of NSM without prior lumpectomy or radiation (n = 187) which had a NAC ischemia rate of only 18.7 %.
Table 16.1




Literature review from 2004 until 2015 showing risk of complications in previously radiated patients undergoing NSM
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree