Fig. 28.1
Skin island designed over the flank (a) and anterior abdominal wall muscles. (b, c) View of extended groin flap harvested on the superficial epigastric artery (white arrow) before cutting iliolumbar artery (black arrow). (d) The silicon sheet lying over the anterior and lateral abdominal wall muscle from the costal cage to thigh muscle. IS iliac spine
EG Flap Transplant Groups
Two subgroups were prepared as isograft and allograft transplants. Isograft transplants were performed between Lewis (RT11) rats (N = 6). EG flaps were elevated as described above; their pedicles were followed under the inguinal ligament and femoral vessels, and were transected at this level. Femoral vessels were transected before the bifurcation at the popliteal region to provide long recipient vessels. Abdominal and flank skins were resected from the recipient rat to create a defect proportionate to the flap size. Flaps’ pedicles, as well as femoral arteries and veins of recipient rats, were anastomosed using the end-to-end technique with 10/0 nylon. The EG flap was sutured into the created defect with absorbable material (4/0 Vicryl, Ethicon). In the allograft transplant group (N = 6), EG flaps were transplanted from LBN (RT 1+n) donor to Lewis (RT11) recipients, as described above. Immunosuppressive protocol was based on tapered dosage of cyclosporine A (CsA) monotherapy starting from 16 mg/kg/day during the first week, tapered to 2 mg/kg/day during the fourth week, and maintained at this level during follow-up period.
Assessment of Flap Viability
Direct Observation
The same surgeon (SN) assessed viability of the skin island, and subsequently observed both isograft and allograft transplants daily. The survival of the EG flap skin transplant was evaluated during the postoperative follow-up period by gross inspection of the flap color, edema, erythema, hair loss, and necrosis compared with normal skin.
Microangiography
Microangiography was performed to demonstrate the vascularization of the total abdominal wall flap in two animals in the isograft transplant group on post-transplant day 15. After cannulation of the common carotid artery, 50 cc of a mixture (at 70 °C) composed of 50 g of silver oxide, 5 g of gelatin, and 100 cc of 0.9 % sodium chloride was injected with a syringe. The flaps were stored at 4 °C for 4 h. Flaps then underwent radiography with a soft X-ray machine, using Microvision-C mammography film.
India Ink Injection Studies
In two isograft transplants, viability of flaps was evaluated by the presence of dye in the vessels after India ink injection. Following cannulation of the common carotid artery with a 24-gauge catheter, 5 ml of India ink were injected. The flap was harvested and stained with hematoxylin-eosin, and examined under light microscopy for the presence of India ink in blood vessels of the skin and subcutaneous tissue.
Histology
On post-transplant day 100, two animals in the allograft group were euthanized, and flaps were harvested and placed in 10 % buffered formalin. The skin sections were stained with H&E for evaluation.
Immunologic Evaluation
Flow cytometry [5] was used to evaluate the presence of donor-specific chimerism for donor MHC class I (RT1n) antigen, in the peripheral blood of Lewis EG flap transplant recipients during observation time at post-transplant days 7, 21, 63, 100, and 150. Donor chimerism was assessed with combinations of conjugated mouse anti-rat mAb RT1n−FITC (for MHC class I of donor cells RT1n, clone MCA156; Serotec, UK) with mAb specific for T-cells: CD4−PE (clone OX-35), CD8a−PE (clone OX-8), B-cell CD45RA−PE (clone OX-33) and monocytes, granulocytes CD11b/c −PE (clone OX-42) (BD Bioscience Pharmingen). After incubation, samples were lysed and fixed with 1 % PFA solution. Negative control panels were tested, and included isotype-matched antibodies (IgG1 −FITC/IgG2 −PE) and phosphate-buffered saline samples. Analysis was performed on 1 × 104 cells using a FACS scan (BD Bioscience Pharmingen) and Cell Quest software.
Results
Anatomic Study Group
At postoperative day 7, all skin islands were viable for EG flaps, based on SEV pedicles (Fig. 28.2). These results demonstrated that SEVs provide total EG flap viability. The area of skin islands of EG flap was measured to be 23.78 cm2.
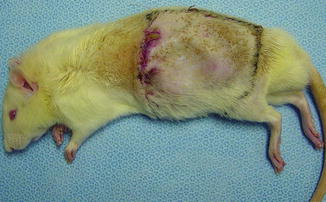

Fig. 28.2
Total viable skin of pedicled flap at day 7 after reinsert
EG Transplant Groups: Direct Observation
During the postoperative follow-up period (at post-transplant day 100), the skin islands of all flaps in the isograft and allograft transplant groups were completely viable (Fig. 28.3a–c).
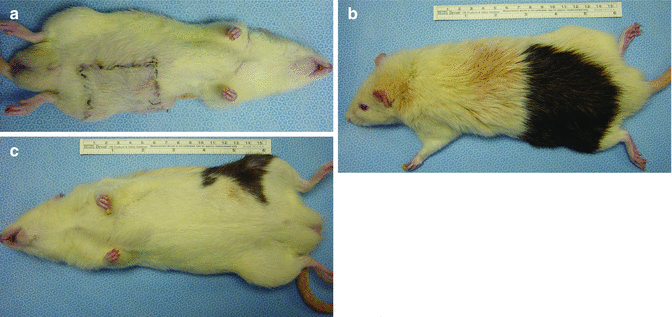

Fig. 28.3
(a) Viable skin island of isograft [(Lewis (RT11)→ Lewis (RT11)] (a) and allograft [(LBN (RT 1+n)→ (Lewis (RT11)] (b, c) at post-transplant day 100
Microangiography
Microangiography revealed the vascular supply in the flap, provided by the pedicle (Fig. 28.4a, b).
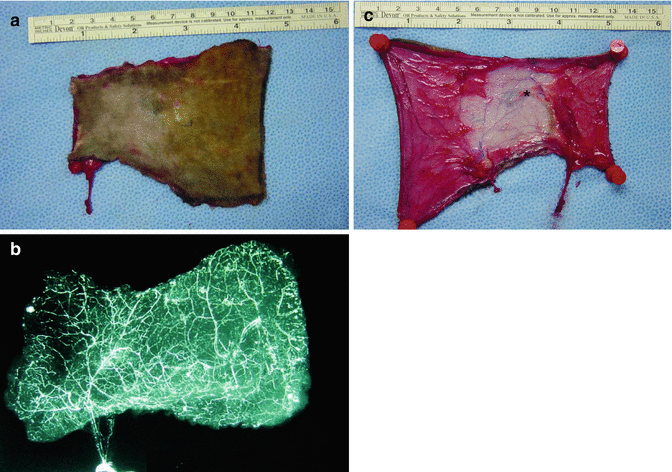

Fig. 28.4
(a, b) Microangiographic analysis demonstrates the course of the pedicle. (c) Vessel connections area between superficial epigastric and iliolumbar arteries
India Ink Injection Studies
Dye studies with India ink demonstrated its uptake by vessels in the skin, confirming perfusion via the pedicle (Fig. 28.5).
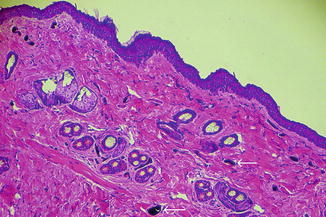

Fig. 28.5
Histological examination of allograft skin (LBN) (RT 1+n) indicates perfusion by presence of ink in vessels (arrows) (Hematoxylin-eosin stain; original magnification ×400)
Histology
Histologic examination of EG flap allotransplants at post-transplant day 100 did not reveal signs of rejection such as blood cell extravasation, edema, necrosis, and pigment incontinence. Normal skin and subcutaneous tissue architecture were protected; viable epidermis and dermis layers of the skin, as well as intact subcutaneous tissue, were also evident (Fig. 28.6).
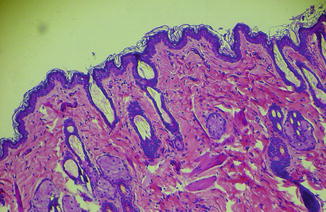

Fig. 28.6
Histological section. Skin and subcutaneous fat tissue component of flap on postoperative day 150 demonstrates viability of each segment (Hematoxylin-eosin stain; original magnification ×400)
Chimerism Level in the Peripheral Blood of Vascularized Skin Allograft Recipients
Chimerism in the peripheral blood was evaluated by the presence of donor-origin cells specific for T-lymphocytes (RT1n/CD4, RT1n/CD8), B-lymphocytes (RT1n/CD45RA), and granulocytes/monocytes (RT1n/CD11b/c), respectively (Fig. 28.7a




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








