Chapter 44 Molecular Biology of Tendon Healing
This chapter does not appear in the print edition.
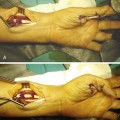
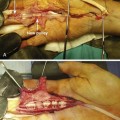
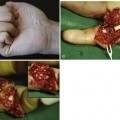
In the 1970s and 1980s, a number of elegant experimental investigations demonstrated that cells in the intrasynovial tendon segment can proliferate and participate in the healing process, establishing the concept of “intrinsic tendon healing.”1–7 Since 1990s, investigations into tendon healing biology have been directed toward elucidation of molecular events underlying the healing process and development of molecular therapeutic methods to enhance the healing or to decrease adhesion formations. These investigations came from several major laboratories around the world, under investigators such as Abrahamsson,8–10 Chang,11–14 Gelberman, Boyer, and Thomopoulos,15–20 McGrouther,21–24 Mass,25,26 Tang,27–34 and Wolfe.35
Molecular Biology of Tendon Healing
Roles of Individual Growth Factors
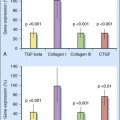
A variety of molecules are involved in tendon healing.36 The roles of six growth factors have been studied associated with tendon healing in vivo or tenocyte proliferation in vitro:
Direct injection of VEGF increased the healing strength of Achilles tendon in a rat model. However, the effectiveness of direct use of VEGF in synovial tendons has not been tested. We found that AAV2-VEGF treatment significantly increased the healing strength of the chicken flexor tendons at postoperative weeks 3, 4, and 6.
No studies have directly investigated expression profile of BMPs during intrasynovial tendon healing process. However, studies were performed to determine relation of BMPs with the healing in rat Achilles tendon. Eliasson and colleagues45
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree