Introduction
A severe burn elicits a stress response that initially assists survival. There is an elevation in circulating concentrations of catecholamines, such as epinephrine and norepinephrine, alongside a concurrent increase in inflammatory cytokine production. Cardiac index dramatically increases. This stress response is also associated with a significant increase in energy expenditure. In addition to cardiac and immune function, altered protein as well as lipid and glucose metabolism are key factors contributing to burn-induced hypermetabolism. The hyperinflammatory-state postburn promotes muscle protein catabolism and chronic skeletal muscle catabolism that is not readily overcome through nutritional support. If this response continues unchecked, as occurs in severely burned patients, chronic hypermetabolism and catabolism hinder patient recovery and reintegration into society. Thus the mechanisms underlying the development and persistence of the postburn stress response, as well as interventions to ameliorate these responses, remain areas of intense research interest.
Cardiovascular adaptations
One of the most profound and well-documented components of the burn-induced hypermetabolic response is cardiac dysfunction. Immediately following injury, patients experience burn shock accompanied by reduced heart rate, cardiac output, and contractility. By 2 to 3 days after injury, the cardiovascular system rebounds, and heart rate and cardiac work increase significantly above normal levels. , Postburn heart rates approach 160% of those in nonburned, healthy patients. Systolic and diastolic dysfunction and increased myocardial energy demand also occur. These alterations in cardiac function are associated with longer stays in the intensive care unit (ICU) in addition to increased morbidity, as quantified by number of surgical interventions. Furthermore, tachycardia and energy expenditure remained elevated for up to 3 years after the external wounds were healed, underscoring the longevity of the cardiovascular response to severe burns. ,
Skeletal muscle catabolism and body compositional changes
Postburn pathophysiologic skeletal muscle catabolism and loss of lean body mass (LBM, or body cell mass in Franny Moore’s work) significantly prolong rehabilitation. Muscle wasting occurs due to an imbalance in the ratio of protein synthesis to protein breakdown. , Catabolism of LBM correlates with increased morbidity in burn victims. After LBM loss of 10%, marked delays in wound healing and higher infection rates proportionally increase as the percentage of LBM loss increases. Acutely, the net LBM losses from muscle wasting lead to prolonged mechanical ventilation, inhibition of cough reflexes, and a delay in mobilization and contribute to increased morbidity in these patients. Chronically, these losses reduce strength and the potential for full rehabilitation. Similar to other components of the hypermetabolic response to burns, burn-related muscle cachexia persists for several years following injury. Furthermore, persistent protein catabolism has been hypothesized to account for the growth delay that frequently occurs in our pediatric burn patients.
LBM wasting is hypothesized to be due to a redistribution of protein as well as use of skeletal muscle as a fuel source. Protein balance studies (whole body and cross leg) show persistent muscle breakdown for nearly 1 year postburn. Patients with severe burns can experience an average nitrogen loss of 20 to 25 g/m total body surface area (TBSA) per day, a rate at which lethal muscle cachexia becomes imminent in less than 1 month if left untreated. These losses are likely due to a combination of postburn hypermetabolism and bed rest, and recent investigations indicate the loss of LBM more severely affects the upper extremities than the lower extremities. Because a significant portion of insulin-stimulated glucose uptake occurs in the skeletal muscle, significant LBM loss may also contribute to postburn insulin resistance. Flakoll et al. showed that increased plasma glucose levels stimulated whole-body proteolysis in the absence of changes in leucine oxidation or nonoxidative disposal. While LBM catabolism is elevated, a decrease in anabolic capacity occurs, further reducing LBM. Satellite cells, the muscle stem cells that regenerate skeletal muscle, are impacted by burn injury. Within the skeletal muscle tissue, although there is an increase in satellite cell proliferation, there is a concurrent increase in apoptosis, leading to a net reduction in satellite cells. The end result of the increase in net protein breakdown alongside a decrease in satellite cells appears to be a reduction in total LBM.
In addition to the alterations in skeletal muscle protein synthesis, breakdown, and regeneration, oxygen consumption is also greatly increased after burn injury. However, the molecular mechanism underlying these alterations is not clearly defined. Data suggest burn-induced derangements of skeletal muscle mitochondrial function and posit that through recruitment of mitochondrial proton leak, altered skeletal muscle energetics may be a key contributor to burn-induced hypermetabolism. Indeed, greater uncoupled mitochondrial respiration in skeletal muscle of severely burned patients persists for more than 1 year postburn, suggesting greater heat production through mitochondrial proton leak in muscle may contribute to chronic hypermetabolism postburn. At a whole-body level, proton leak (mitochondrial respiration that is uncoupled from adenosine triphosphate production) may account for nearly one-third of the hypermetabolic response to burns, suggesting that mitochondrial energy transduction may be a novel therapeutic target to alleviate hypermetabolism. ,
Insulin resistance and hyperglycemia
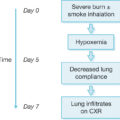
Hyperglycemia is another common metabolic derangement in response to burn injury that occurs in both pediatric and adult burn populations and persists well after the initial discharge from the ICU. , , Glucose flux is more complex than increased or decreased blood levels, as reviewed with Pruitt and Willmore’s extensive investigations into this phenomenon. , Insulin resistance and hyperglycemia contribute to poor wound healing and muscle catabolism. Elevated cortisol and catecholamine levels increase the delivery of glucose to vital organs, thus inhibiting insulin’s anabolic functions. Catecholamines impair glucose disposal and contribute to peripheral insulin resistance by inhibiting both insulin release and glucose uptake ( Fig. 23.1 ). , The availability of gluconeogenic substrates, such as glycerol lactate and alanine, is increased by lipolysis of adipose tissue, glycogenolysis, and proteolysis of skeletal muscle postburn, respectively, which support hepatic glucose production, particularly if nutritional support is lacking (see Fig. 23.1 ). , Moreover, elevated blood glucose levels fail to suppress hepatic glucose release, exacerbating hyperglycemia in burn patients. This is further complicated by catecholamine-mediated glycogen breakdown. Impaired mitochondrial function in the liver and the skeletal muscle has been associated with altered lipolysis and insulin signaling postburn by dampening insulin’s inhibition of glucose production in the liver and altering glucose uptake into skeletal muscle. , Glucagon and proinflammatory cytokines, such as interleukin 6, also play a role in modulating glycogenolysis, gluconeogenesis, and insulin signal transduction, resulting in further augmentation of hyperglycemia and insulin resistance.
The effects of metabolic dysfunction after severe burn injury.
(From Williams FN, Jeschke MG, Chinkes DL, et al. Modulation of the hypermetabolic response to trauma: Temperature, nutrition, and drugs. J Am Coll Surg. 2009;208[4]:489-502.)
Alterations in lipid metabolism and fat composition
Catecholamine-induced lipolysis increases plasma free fatty acid concentrations, which contributes to organ steatosis and insulin resistance in patients with severe burns. Kraft et al. showed that increased plasma triglyceride levels in severely burned children correlated with poorer outcomes, including impaired organ function, thus confirming an earlier report linking elevated triglycerides with morbidity. , Loss of peripheral subcutaneous fat may also play a role in the development and persistence of insulin resistance after a severe burn. Recently, browning of white adipose tissue (the adoption of a thermogenic brown phenotype by white adipocytes) and the implications this potentially has for patients with diabetes and/or metabolic syndrome has become the topic of intense research. White adipose tissue isolated from severely burned patients underwent an apparent beiging/browning, where the histologic development of smaller adipocytes with more mitochondria in addition to elevated expression of uncoupling protein 1 (UCP-1) were observed. This response has since been confirmed by others, affirming that increased adrenergic and inflammatory stress in white adipose tissue postburn is associated with an apparent browning response. Browning of white adipose tissue has been proposed to play a role in burn-induced hypermetabolism, where adipose tissue becomes a more thermogenic tissue in response to severe burn trauma. However, evidence for the induction of functional UCP-1 in the white adipose tissue of burn patients remains elusive. The emphasis of future research should be placed on developing both nonpharmacologic and pharmacologic therapeutic approaches to better understand and modulate the stress response in adipose tissue following severe burns.
Nonpharmacologic modulation of the hypermetabolic response
Environmental support
In addition to acting as a protective barrier, the skin plays a critical role in insulation and thermoregulation. Burn patients with extensive burn wounds and skin loss also evaporatively lose, in an uncontrolled manner, significant body water, which is also accompanied by dramatic and rapid heat loss (thermodynamic heat of vaporization of water). To adapt to heat loss through the wound, core and skin temperatures become elevated in burn patients. Resting energy expenditure can increase more than twofold postburn, but can be reduced by approximately 30% by increasing the temperature of the room and using occlusive wound dressings. Thus environmental modulation is a key primary treatment goal that is too often underutilized. It is the prototypic low-hanging fruit of metabolism modulation after burn injury.
Early wound excision and closure
The practice of early excision of necrotic tissue and burn wound closure has improved mortality and morbidity rates. Early burn wound closure provides rapid control of evaporative heat loss and infectious portal, which attends the burn wound; it is also associated with diminished incidence of excessive scars and joint contractures, which expedites patient rehabilitation. The use of various skin substitutes to cover wounds can reduce hypermetabolism by restoring the protective barrier required for thermoregulation and prevention of heat loss and water evaporation. Blood loss is a major determinant of morbidity and mortality, and a variety of techniques need to be used to control bleeding, including the local application of fibrin or thrombin sprays, epinephrine-soaked pads (1:40,000), topically administered epinephrine (1:10,000–1:20,000), or electrocautery of blood vessels. Additional use of tourniquets and preexcisional tumescence with epinephrine and saline may also help limit blood loss. Thus novel skin substitutes and therapies to improve wound healing and prevent blood loss are important potential avenues to modulating burn hypermetabolism and the larger burn problem.
Nutritional support
Adequate nutrition is a critical issue for severely burned patients because patients treated solely with oral supplementation may still lose one-quarter of their preadmission weight within the first several weeks. The marked postburn changes in resting energy expenditure determine the caloric needs of each respective patient. Insufficient nutrition exacerbates muscle wasting and prolongs wound healing. However, high-fat nutritional intake is associated with obesity and hepatic steatosis after burn injury, negatively impacting morbidity and mortality. , Caloric enteral intake guided by energy expenditure measurements via indirect calorimetry is currently recommended to provide sufficient energy while avoiding overfeeding, which may exacerbate hypermetabolism. , Over the past 40 years, several formulas have been derived to calculate patient-specific caloric requirements, including the Curreri formula (25 kcal/kg/day + 40 kcal/% TBSA burn/day). Recently, the European Society for Clinical Nutrition and Metabolism and the American Society for Parenteral and Enteral Nutrition released guidelines recommending that enteral feeding begin as soon as possible after admission with 1.5 to 2 g/kg and 3 g/kg protein for severely burned adults and children, respectively. , Many ICUs deliver a substantial percentage of calories as fat (25%) because essential fatty acid deficiency frequently accompanies long-term nutritional supplementation. This approach reduces carbohydrate requirements and burn-induced glucose intolerance, and therefore dietary compositions of 30% to 50% fat are now the standard of care when treating critically ill patients. Unfortunately, excessive fat administration in burn patients is associated with hepatic steatosis, hypoxemia, hyperlipidemia, higher infection rates, and higher postoperative mortality rates. , Hepatic triglyceride levels are higher, thereby limiting the utility of exogenous lipids as a postburn energy source. , , , The incidence of fatty liver is reduced in patients receiving Vivonex TEN or equivalent elemental formulas compared to those fed milk. Additional benefits attributed to Vivonex TEN include improved survival, significantly lower incidences of sepsis, and reduced length of ICU stay per %TBSA. A high-carbohydrate enteral diet was associated with a reduction in muscle wasting compared to a high-fat, low-carbohydrate formula. We thus advocate nutritional regimens for burn patients that reduce fat intake to no more than 3% of the total energy provided. Consistent high-carbohydrate, high-protein enteral supplementation can also support endogenous insulin release, which in turn can support skeletal muscle anabolism in severely burned patients. The attendant and expected hyperglycemia also permits safe insulin-drip administration and titration (lower risk of hypoglycemia).
Rehabilitative exercise
Despite nutritional interventions, cachexia and wound contractures are common comorbidities of major burns. While undergoing the hormone and inflammatory stress responses, patients with large burns are immobile for extended periods, further prolonging rehabilitation and recovery. The utilization of exercise therapy attenuates loss of muscle mass and function and significantly improves cardiopulmonary function, although sustained postburn rehabilitative exercise training has not yet been shown to completely restore cardiopulmonary function. A regimented exercise program improved pulmonary and skeletal muscle function in both pediatric and adult burn patients. A 12-week exercise program beginning immediately following discharge from the ICU also significantly increased percent predicted peak heart rate and maximal oxygen consumption. Progressive resistance exercises maintain or even increase LBM, facilitate the formation of muscle proteins by incorporating amino acids, enhance muscle strength, and increase walking distances by 50%. , Celis et al. reported fewer corrective surgeries in burned children after resistance exercise training. Similarly, Paratz et al. reported that severely burned adults who participated in an exercise program required fewer contracture releases. In almost all children with large burn injuries, severe, life-altering contractures are common. A program of progressive rehabilitative exercise training, including ~20 minutes of exercise sufficient to achieve 60% to 80% of the patient’s predicted peak heart rate (220 minus age in years), 5 days a week for 12 weeks, demonstrated a dramatic (60%) reduction in the need for burn scar contracture release surgeries.
Recent studies have shown that exercise training in adults improves psychosocial outcomes, such as quality of life, in addition to the expected improvements in physical function. This finding was corroborated by Rosenberg et al. in pediatric patients. The combination of exercise training and pharmacologic therapies to attenuate hypermetabolism, such as propranolol or oxandrolone, is also being investigated. Peak oxygen consumption was significantly higher in patients receiving both propranolol and exercise training. The combination of oxandrolone and exercise resulted in a significant increase in LBM. Finally, Wurzer et al. reported that the benefits of a 12-week post-ICU discharge exercise program were no longer apparent at 2 years postburn, indicating that patients should continue to exercise to maintain the aforementioned benefits.
Pharmacologic modulation of burn-induced hypermetabolism and muscle cachexia
Recombinant human growth hormone
As demonstrated by our group, during the acute hospitalization period, daily intramuscular injection of recombinant human growth hormone (rhGH; 0.2 mg/kg) reduces the hepatic acute-phase response, improves muscle protein kinetics, maintains muscular growth, blunts hypermetabolism, reduces cardiac output, and decreases donor-site healing times by approximately 1.5 days. Pediatric patients receiving rhGH for 1 year had significantly higher body weight and LBM at the end of the treatment period compared to their placebo-treated counterparts. Additionally, rhGH-treated patients had improved growth as measured by bone mineral content and height percentiles at both 1 and 2 years after injury. Similar improvements with rhGH treatment, such as increased LBM and muscle strength, have been reported in burned adults.
rhGH mediates its effects through its secondary mediator insulin-like growth factor 1 (IGF-1). In patients receiving rhGH, serum IGF-1 and IGF-binding protein 3 (IGFBP-3) increased 100% over the levels measured in healthy controls. Nevertheless, as demonstrated in a study by Takala et al., in 532 nonburned critically ill patients, increased rhGH (0.10 ± 0.02 mg/kg) correlated with increased morbidity and mortality rates and was associated with hyperglycemia and insulin resistance. This may be reflective of an age-specific effect, however, because in severely burned children, mortality rates were not affected by either short- or long-term rhGH administration. , However, rhGH-treated burn patients did display an increased incidence of hyperglycemic episodes and increased plasma levels of free fatty acids and triglycerides.
Insulin-like growth factor 1
Because positive effects of rhGH on the postburn hypermetabolic response are predominantly mediated by the secondary mediator IGF-1, it is not surprising that administration of rhIGF-1 would have similar results. Indeed, administration of equimolar amounts of rhIGF-1 along with its binding protein, IGFBP-3, attenuated muscle catabolism, restored gut mucosal integrity, enhanced immune function, and returned serum concentrations of constitutive proteins to nonburned levels. However, the administration of IGFBP-3 was associated with increased neuropathies in this patient population and is not currently recommended for clinical use.
Oxandrolone
The use of oxandrolone, a nonaromatizable analog of testosterone possessing only 5% of testosterone’s virilizing androgenic effects, enhances anabolism of muscle protein by improving the efficiency of protein synthesis in severely burned children. Oxandrolone administration decreases loss of body weight and improves healing of the donor site wound. In a large clinical trial by our group, 0.1 mg/kg oxandrolone administered twice daily reduced length of acute hospitalization, sustained LBM, and improved liver protein synthesis. Severely burned pediatric patients receiving oxandrolone for 1 year experienced improved growth, decreased cardiac work, and improved muscle strength. Oxandrolone treatment also improved lung function at rest and during exercise in this patient population. These improvements were maintained for up to 4 years after treatment had ended. The benefits of oxandrolone administration after burn injury were further enhanced when the treatment period was increased from 1 to 2 years. Oxandrolone and propranolol in combination were also shown to reduce postburn scaring and improve physical recovery. It is unspeakably disappointing that oxandrolone has been withdrawn from the US market at the manufacturer’s request. A concerted effort by organized medicine, led by the American Burn Association and the American College of Surgeons, to assist the US Food and Drug Administration in understanding the importance of this affordable and impactful medication in improving countless burn recoveries is here proposed.
Propranolol
The catecholamines norepinephrine and epinephrine activate cardiac β-adrenergic receptors to increase heart rate and cardiac work in response to severe burn trauma. Propranolol is a nonspecific β-adrenergic antagonist that prevents catecholamine activation of β-adrenergic receptor-mediated signal transduction. The administration of propranolol (titrated to decrease heart rate by 15%–20%) diminishes cardiac work and reduces hepatic steatosis, as shown in several studies. , , In the hypermetabolic burn patient, these efficiencies are not accompanied by decreased peripheral perfusion, highlighting the apparent excess of sustained postburn hypermetabolism. Also, administration of propranolol leads to reduced skeletal muscle catabolism and increased LBM postburn, as demonstrated using stable isotope studies and body composition analysis. In the postburn environment of heightened protein breakdown and peripheral lipolysis, propranolol enhances protein synthesis and improves postburn morbidity. , Similar to oxandrolone, long-term propranolol administration was associated with decreased cardiac work, resting energy expenditure, and other key markers of the hypermetabolic, hypercatabolic response to burn injury. Furthermore, patients treated with propranolol for 12 months showed improved LBM accretion and reduced bone mass. While the majority of investigation into the use of propranolol postburn has been in pediatric patients, we have shown that, in adult patients, propranolol administration can reduce blood loss as well as improve wound healing. Thus β-adrenergic blockade with propranolol may represent an efficacious anticatabolic therapy for severe burns.
There are several published studies that investigated the use of rhGH in severely burned patients who were also receiving propranolol. The premise of these studies was that the coadministration of rhGH and propranolol would have an additive effect to reduce postburn hypermetabolism and catabolism. In a crossover study of six burned children, heart rate and the rate of release of free fatty acids were significantly reduced with coadministration of propranolol and rhGH. In a later study, there was no evidence of an additive effect of combining rhGH and propranolol, although propranolol reduced heart rate and energy expenditure and improved anabolism. This study also found no evidence of an anabolic effect of rhGH alone. A prospective randomized controlled trial of rhGH and propranolol coadministration showed that the addition of propranolol reduced rhGH side effects by reducing peripheral lipolysis and inflammation. These data indicate that the beneficial effects of these drugs have different mechanisms and do not display characteristics of a synergistic or additive relationship.
Coadministration of oxandrolone and propranolol decreased duration of growth arrest and increased growth rate following the resumption of growth in pediatric burn patients. Thus oxandrolone, either alone or with propranolol coadministration, is a viable and promising therapeutic for attenuating postburn hypermetabolism and catabolism. As noted, scarring and psychosocial recovery were improved in oxandrolone plus propranolol cotreated children.
Insulin
Insulin is the prototypic anabolic hormone. Like other components of the stress response to burns, hyperglycemia and insulin resistance can persist for up to 3 years postinjury and may have further long-term health implications for burn survivors. , , The most common in-hospital treatment for postburn hyperglycemia is insulin administration. In addition to its well-known effects on gluconeogenesis, proteolysis, and fatty acid synthesis, insulin also promotes antiinflammatory signaling pathways. Pediatric burn patients treated with insulin had significantly better outcomes as evidenced by faster wound healing and reduced muscle wasting. Despite these improvements, insulin administration in this patient population must be performed with care. Studies in critically ill patients using either an intensive insulin dosing regimen or a continuous hyperinsulinemic, euglycemic clamp showed that high insulin doses increased episodes of severe hypoglycemia. However, these episodes do not materialize out of thin air. Real-time monitoring of blood glucose concentration at the bedside is essential in safely clamping glucose levels by continuous insulin infusion, suggesting failure of adequate glucose monitoring and provision likely underly hypoglycemic events. For perspective, insulin drips are routinely and safely administered in critical care units around the world. From a critical care standpoint, it can be challenging to maintain a continuous hyperinsulinemic, euglycemic clamp in severe burn patients because of their dependence on enteral feeding in the early stages of recovery. Burn patients also undergo frequent procedures that are (often erroneously) accompanied by the cessation of enteral nutrition, such as operations and dressing changes. These periods of feeding cessation can increase the risk of hypoglycemia. The obvious solution is to provide a sufficient intravenous glucose rate to prevent hypoglycemia (often higher than expected in burn patients with stopped enteral feeds) and to titrate the intravenous insulin drip to maintain blood sugar below 180 mg/dL, just like how hyperglycemia is treated in every high-preforming (nonburn) critical care unit worldwide. The widespread use of continuous glucose monitoring in the care of diabetic patients presents a new means of providing real-time glucose data and potentially a way of predicting episodes of hypo- and hyperglycemia that should be explored in the context of glucose control in acute burn care.
Metformin
Metformin is a biguanide drug that corrects hyperglycemia by stimulating peripheral glucose disposal and blunting hepatic glucose production. Moreover, unlike insulin, metformin is not associated with hypoglycemic events. In severe burn patients, metformin not only improved glucose levels but also improved muscle protein synthesis. , A recent study in adult burn patients confirmed the benefits of metformin treatment on glucose levels and insulin resistance. Similar to insulin, metformin reduced the postburn inflammatory response. Insulin sensitivity was also enhanced with metformin administration. One contraindication of metformin administration in burn patients is the development of lactic acidosis. There has been one case report of lactic acidosis in a burn patient who was receiving metformin clinically for type 2 diabetes mellitus prior to injury. However, in a cohort of 18 metformin-treated severe burn adult patients, lactic acidosis was not observed. Studies are ongoing to evaluate the incidence of lactic acidosis in burn patients and whether metformin will continue to be a valid hyperglycemia treatment in this patient population. Preliminary data indicate that metformin is safe and effective in severe burn patients.
Alternative therapeutic options
There have been a variety of drugs investigated to attenuate the postburn hypermetabolic, hypercatabolic response. Many of these drugs work to resolve hyperglycemia, such as glucagon-like peptide 1 (GLP-1) and peroxisome proliferator-activated receptor γ (PPAR-γ) agonists. An analog of GLP-1, exenatide, decreased burn-induced hyperglycemia but required adjunctive insulin therapy to be effective. Fenofibrate, a PPAR-γ agonist, not only improved plasma glucose levels but also insulin signaling in the muscle and liver, with the end result of increased insulin sensitivity. Fenofibrate also improved skeletal muscle mitochondrial enzyme activity and respiratory function as well as whole-body and muscle fat oxidation. , Ketoconazole is an antifungal agent that has been shown to reduce cortisol production, and it was hypothesized that cortisol reduction would diminish postburn hypermetabolism and inflammation. However, despite effectively reducing urine cortisol levels, ketoconazole did not significantly reduce any other aspect of the hypermetabolic response.
Conclusion
The physiologically exhaustive metabolic derangements accompanying severe burns contribute to morbidity and mortality in this patient population. Outcomes for these patients have been significantly improved by advances in nonpharmacologic and pharmacologic therapies ( Table 23.1 ). However therapeutic strategies to abate the persistent hypermetabolism and hyperglycemia remain challenging. Environmental and nutritional support, accompanied by early burn wound excision and grafting, represents one of the greatest improvements in the past 2 decades, improving both morbidity and mortality. Currently, the most effective therapy to reduce the burn-induced hypermetabolic and hypercatabolic responses is β-adrenergic blockade via propranolol administration; rhGH, IGF-1, and oxandrolone (see Table 23.1 ) have also been successfully used in the attenuation of the hypermetabolic and hypercatabolic responses. While intensive insulin therapy can improve mortality and morbidity, there is a need for additional strategies, such as metformin, that lack the increased risk of hypoglycemic events associated with insulin treatment. Nonetheless, further work is needed to elucidate the ideal glucose ranges and safety of the aforementioned therapies in this unique patient population.
Table 23.1
Summary of the Effects of Various Pharmacologic Interventions on Key Components of the Hypermetabolic Response to Burn Injury
| Drug | Cardiovascular | Skeletal Muscle | Insulin Resistance | Lipid Metabolism and Fat Composition |
|---|---|---|---|---|
| rhGH | ↓︎ Cardiac output |
|
↑︎ Hyperglycemia | Unknown |
| IGF-1 | Unknown | ↓︎ Muscle catabolism | ↑︎ Insulin sensitivity hypoglycemia | Unknown |
| Oxandrolone |
|
|
No difference | ↓︎ Free fatty acids |
| Insulin | ↑︎ Resting energy expenditure | ↓︎ Loss of lean body mass | ↓︎ Hyperglycemia |
|
| Metformin | ↑︎ Resting energy expenditure | ↑︎ Protein synthesis |
|
Antilipolytic |
| Fenofibrate | Unknown | ↑︎ Mitochondrial enzyme activity |
|
↑︎ Fat oxidation |
| GLP-1 | No difference | Unknown | ↓︎ Hyperglycemia | Unknown |
| Propranolol |
|
|
↑︎ Insulin sensitivity |
|
| Ketoconazole | No difference | No difference | No difference | No difference |
| rhGH + propranolol |
|
|
↑︎ Insulin sensitivity | ↓︎ Free fatty acids |
| Oxandrolone + propranolol | Unknown |
|
Unknown | Unknown |
G LP-1 , Glucagon-like peptide 1; IGF-1 , insulin-like growth factor 1; rhGH , recombinant human growth hormone.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree