Key points
- •
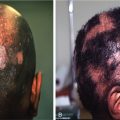
Androgenetic alopecia is the most common cause of alopecia in both men and women; however, approved therapeutic treatment options are limited.
- •
Patients should be counseled that treatment of androgenetic alopecia is directed toward prevention of further hair thinning and loss, as complete hair regrowth may not be achieved.
- •
Topical minoxidil is an Food and Drug Administration–approved, first line treatment for androgenetic alopecia in both men and women. Oral low-dose minoxidil is not Food and Drug Administration approved for the treatment of androgenetic alopecia but may be considered in patients who are unwilling or unable to use the topical formulation.
- •
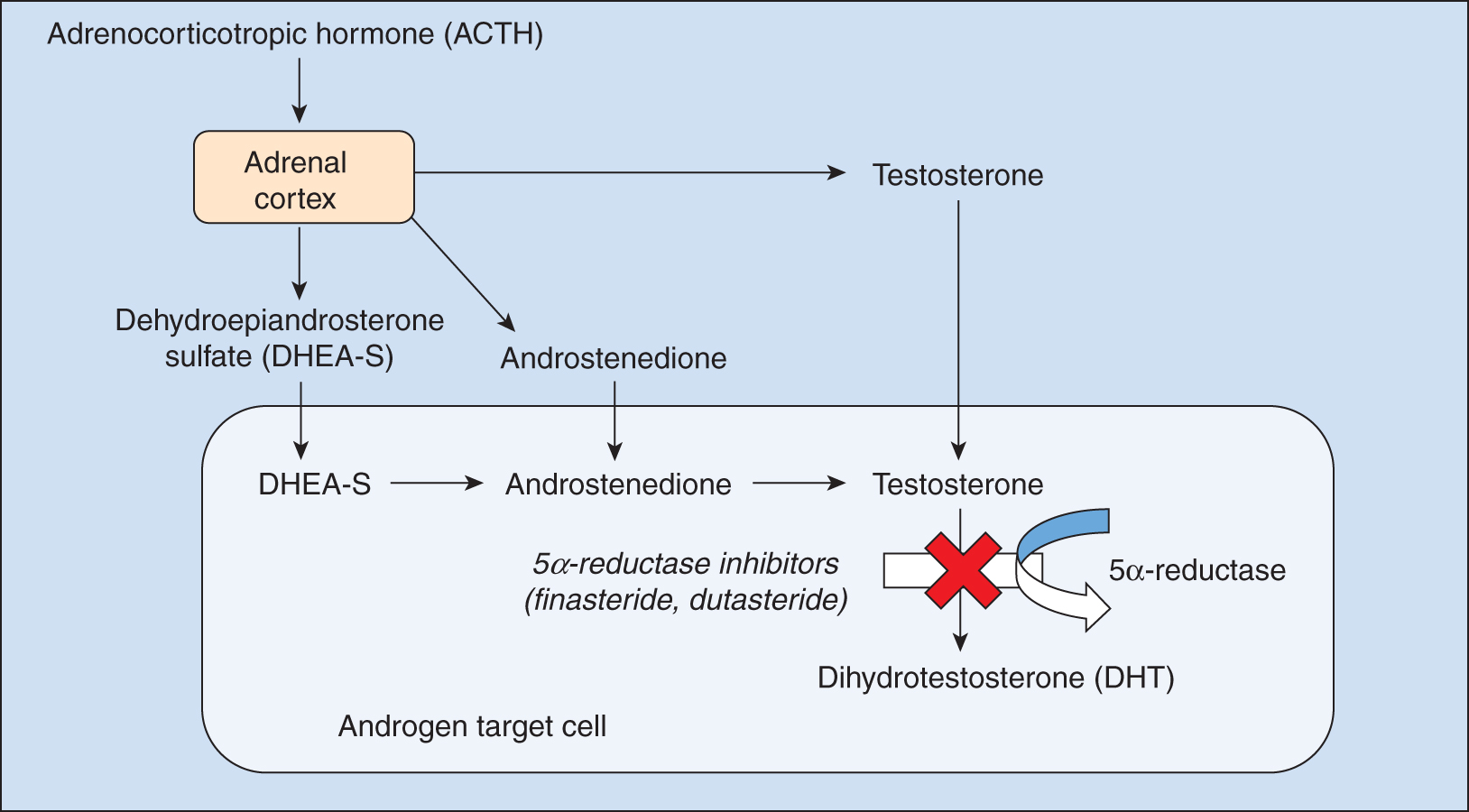
The enzyme 5α-reductase converts testosterone to dihydrotestosterone, thereby inducing hair follicle miniaturization in androgenetic alopecia. Inhibition of this pathway is the target of the 5α-reductase inhibitors finasteride and dutasteride.
- •
Oral finasteride is a type II 5α-reductase inhibitor that is Food and Drug Administration–approved and a first line treatment for androgenetic alopecia in men. Oral dutasteride is a type I and type II 5α-reductase inhibitor that is not Food and Drug Administration approved; however, it can be considered a second-line treatment for androgenetic alopecia in men.
- •
Topical 5α-reductase inhibitors are a promising area of investigation for the treatment of androgenetic alopecia.
Background, definitions, and history
Androgenetic alopecia (AGA) is the most common cause of alopecia in both men and women. Despite its high prevalence, approved therapeutic options for AGA are limited. Treatment of AGA is directed toward prevention of further hair thinning and loss, which can affect both quality of life and self-esteem (level of evidence: 1). , This chapter discusses the U.S. Food and Drug Administration (FDA)-approved treatment options for AGA, including topical minoxidil and oral finasteride. Non-FDA approved treatment options including oral dutasteride, oral minoxidil, and topical finasteride are also discussed.
Minoxidil (Rogaine [topical]), the first FDA-approved treatment for AGA in men and women, was originally developed as an oral hypertension medication. Its possible use in AGA was incidentally discovered as a result of its side effect of increased hair growth. The exact mechanism by which minoxidil promotes hair growth remains unclear, but it is thought to be partly caused by potassium channel opening. Minoxidil is an inactive metabolite transformed into its active metabolite, minoxidil sulfate, by the enzyme sulphotranspherase. Minoxidil sulfate then opens adenosine triphosphate (ATP)-sensitive potassium channels in cell membranes, leading to vasodilation. Other mechanisms by which minoxidil is thought to promote hair growth include stimulation of vascular endothelial growth factor and increased prostaglandin synthesis.
The 5α-reductase inhibitors finasteride (Propecia [oral]) and dutasteride (Avodart [oral]) were initially developed and approved for the treatment of benign prostatic hyperplasia. The enzyme 5α-reductase converts testosterone to the more potent dihydrotestosterone (DHT), which induces characteristic miniaturization of the hair follicles in AGA ( Fig. 8.1 ). There are type I and type II isoenzymes of 5α-reductase, and their expression varies depending on body location. Hair follicles express higher concentrations of the type I isoenzyme, suggesting a key role in androgen-regulated hair growth. , Finasteride is a type II 5α-reductase inhibitor that decreases serum DHT levels by about 65% (see Fig. 8.1 ). In contrast, dutasteride inhibits both type I and type II isoenzymes, further reducing serum DHT levels by 90% (see Fig. 8.1 ). ,
Indications and patient selection
Topical minoxidil is the first line treatment for AGA in both men and women (level of evidence: 1) ( Pearl 8.1 ). , , For men, the 2% solution was approved in 1988, the 5% solution in 1991, and the 5% foam in 2016. For women, the 2% solution was approved in 1991, and the 5% foam was approved in 2014. Topical minoxidil 2-5% solution or 5% foam twice daily is recommended for treatment of mild-to-moderate AGA in men ages 18 and older ( Table 8.1 ). Topical minoxidil 2% solution twice daily or 5% topical foam once daily is recommended for treatment of mild-to-moderate AGA in women ages 18 and older (see Table 8.1 ). Topical minoxidil is not advised for use in women who are pregnant or breastfeeding. ,
Topical minoxidil is first line for the treatment of androgenetic alopecia in both men and women.
| Medication | Dosage | Side Effects |
|---|---|---|
| Topical minoxidil | 2% solution twice daily, 5% solution twice daily (men), 5% foam twice daily (men), or once daily (women) | Transitory increase in hair shedding, scalp irritation and erythema, facial hypertrichosis |
| Topical finasteride | 0.25% solution once daily | Scalp irritation and erythema, testicular pain, headaches, presyncope |
Oral finasteride is FDA approved for treatment of AGA in men and is also considered a first-line therapy (level of evidence: 1) ( Pearl 8.2 ). , The recommended dose of finasteride is 1 mg once daily for men older than 18 years of age to prevent progression or to improve mild-to-moderate AGA ( Table 8.2 ) (level of evidence: 1). Oral dutasteride is not currently FDA approved for treatment of AGA but can be considered a second-line treatment in men older than 18 years of age who have failed previous treatment over 12 months with finasteride (level of evidence: 1). The recommended dose of dutasteride is 0.5 mg once daily (see Table 8.2 ). Finasteride and dutasteride are contraindicated in pregnancy and women of childbearing age.
The 5α-reductase inhibitors include finasteride and dutasteride. Finasteride is FDA-approved and a first-line treatment for androgenetic alopecia in men, whereas dutasteride is not FDA-approved and considered a second-line treatment.
| Medication | Dosage | Side Effects |
|---|---|---|
| Oral finasteride | 1 mg once daily | Decreased libido, erectile dysfunction, decreased ejaculation volume, depression, hypersensitivity reactions, breast tenderness, gynecomastia |
| Oral dutasteride | 0.5 mg once daily | |
| Oral minoxidil | 0.25–2.5 mg once daily | Hypertrichosis, hypotension, bradycardia, pedal edema, electrocardiogram changes |
Expected outcomes
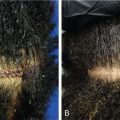
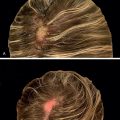
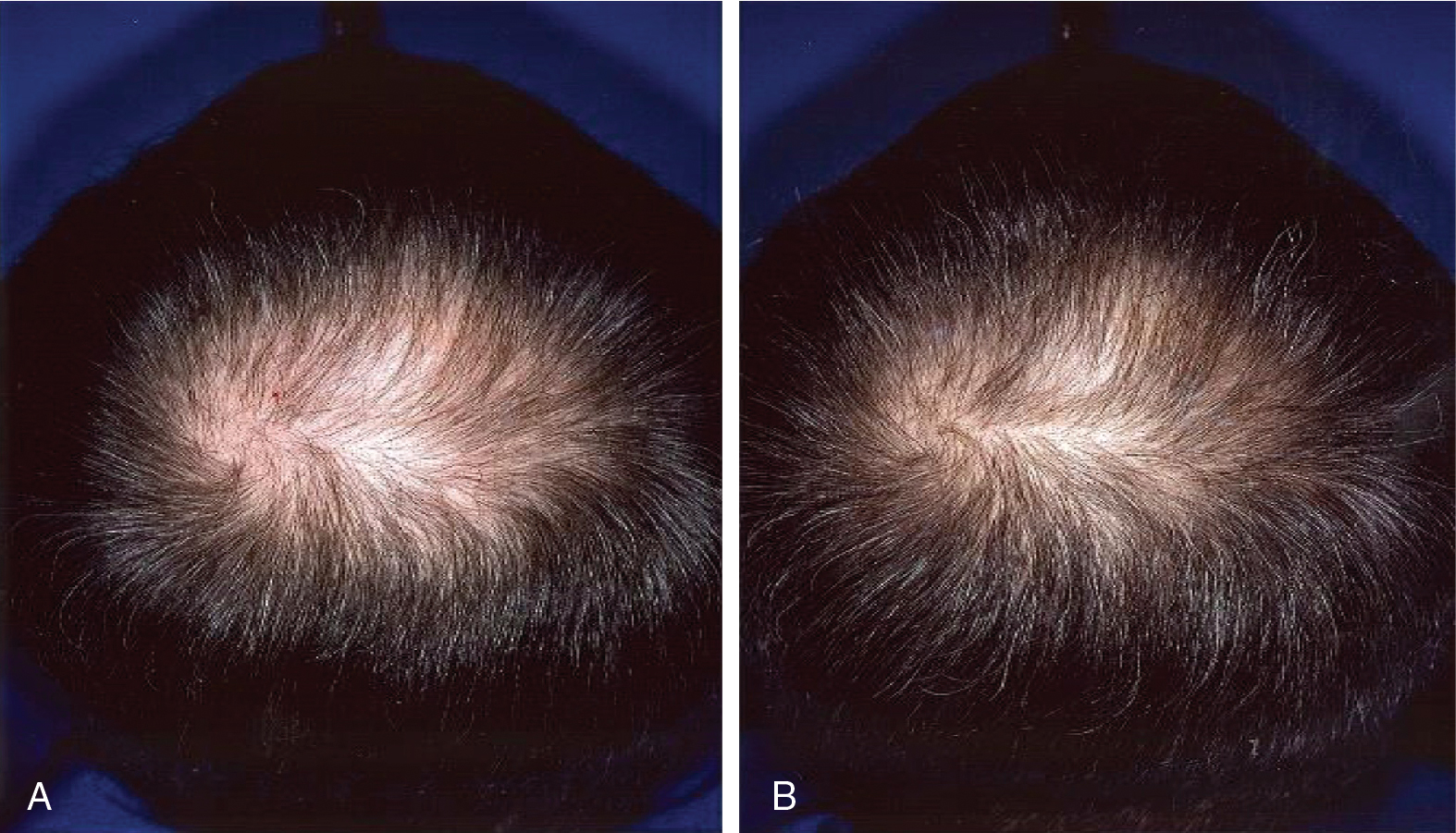
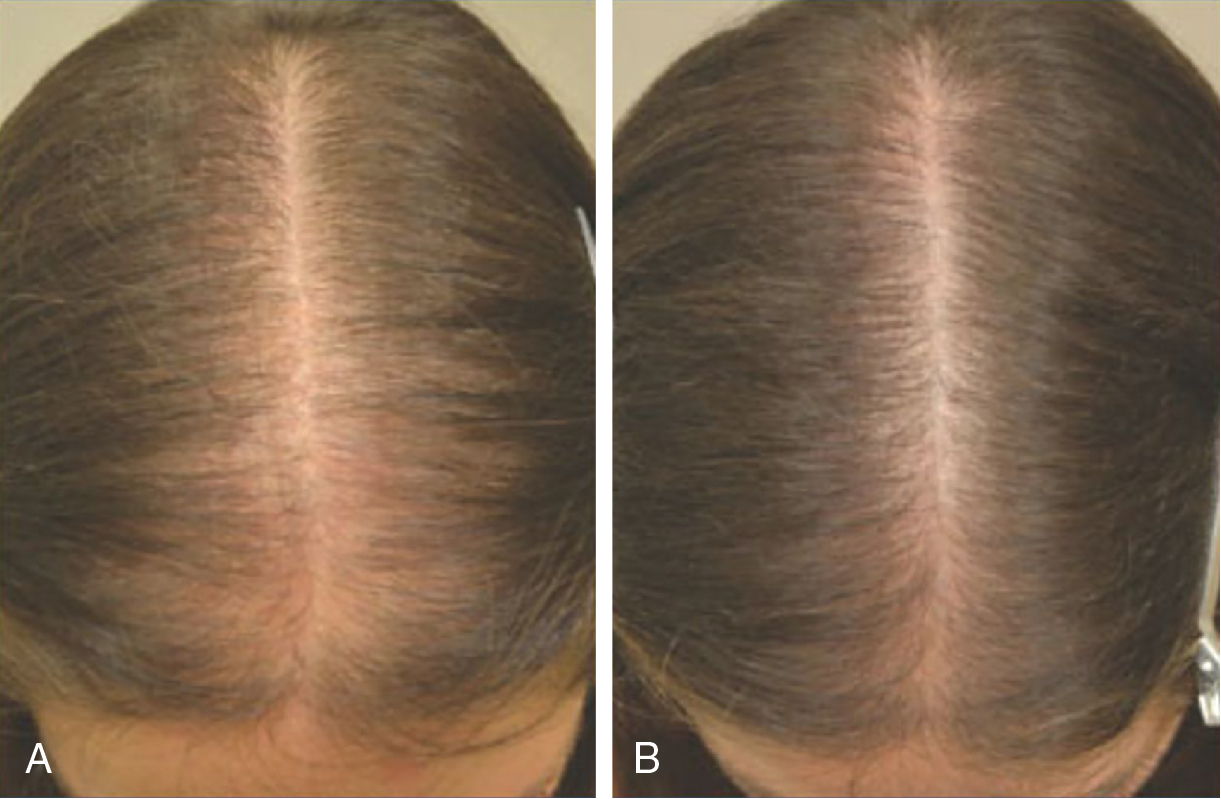
The efficacy of topical minoxidil in treating male and female AGA has been established by numerous double-blind, randomized, placebo-controlled trials and meta-analyses. Kanti et al. assessed the efficacy of topical minoxidil in 48 studies of AGA in men and 19 studies AGA in women. The majority of trials reported findings on the efficacy of topical minoxidil 2% solution, 5% solution, or 5% foam. In male AGA, randomized controlled trials confirmed minoxidil 5% solution and 5% foam are more effective for hair regrowth than the 2% solution ( Figs. 8.2 and 8.3 ) (level of evidence: 1b). , , In female AGA, once-daily 5% foam was similar in efficacy compared with the 2% solution twice daily (level of evidence: 1b) ( Fig. 8.4 ). , , In all studies, topical minoxidil led to significant increases in total hair count when compared with placebo. The mean changes from baseline total hair count were 5.4 hairs/cm 2 and 29.9 hairs/cm 2 at 4 and 6 months, followed by counts between 15.5 hairs/cm 2 and 83.3 hairs/cm 2 at 12 months (level of evidence: 1b). , Visible hair growth was most apparent at four to six months with continuous application of topical minoxidil. To maintain any beneficial effect, applications must continue indefinitely to prevent hair loss. , , If topical minoxidil treatment is stopped, clinical regression occurs within six months. ,
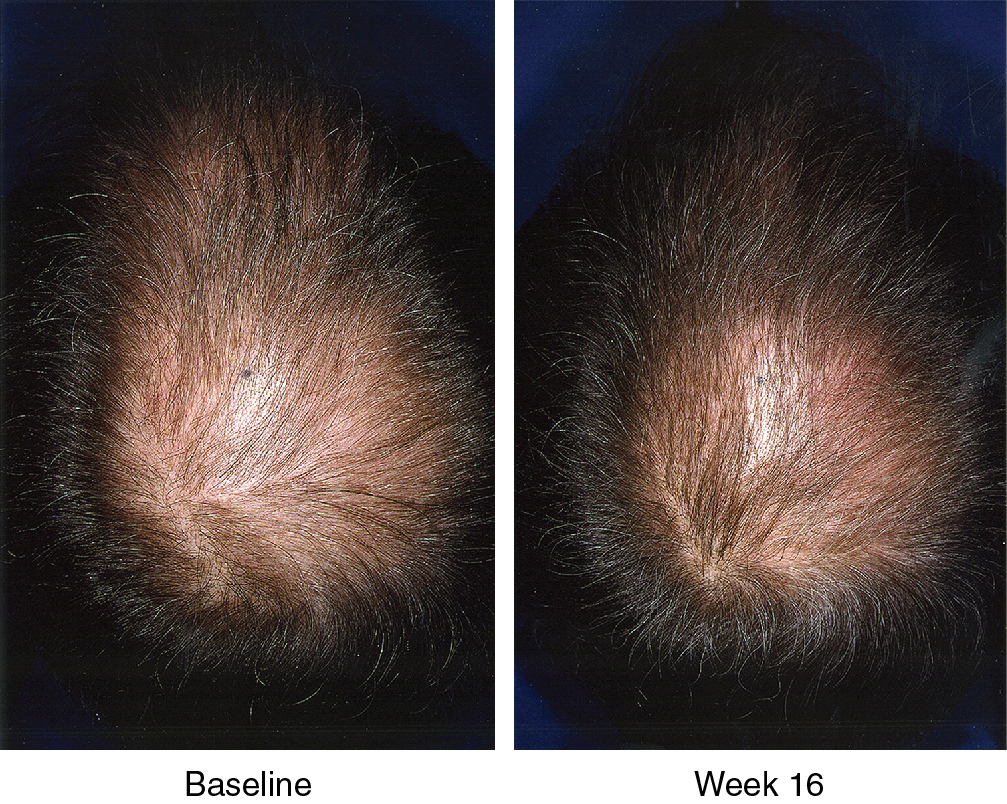
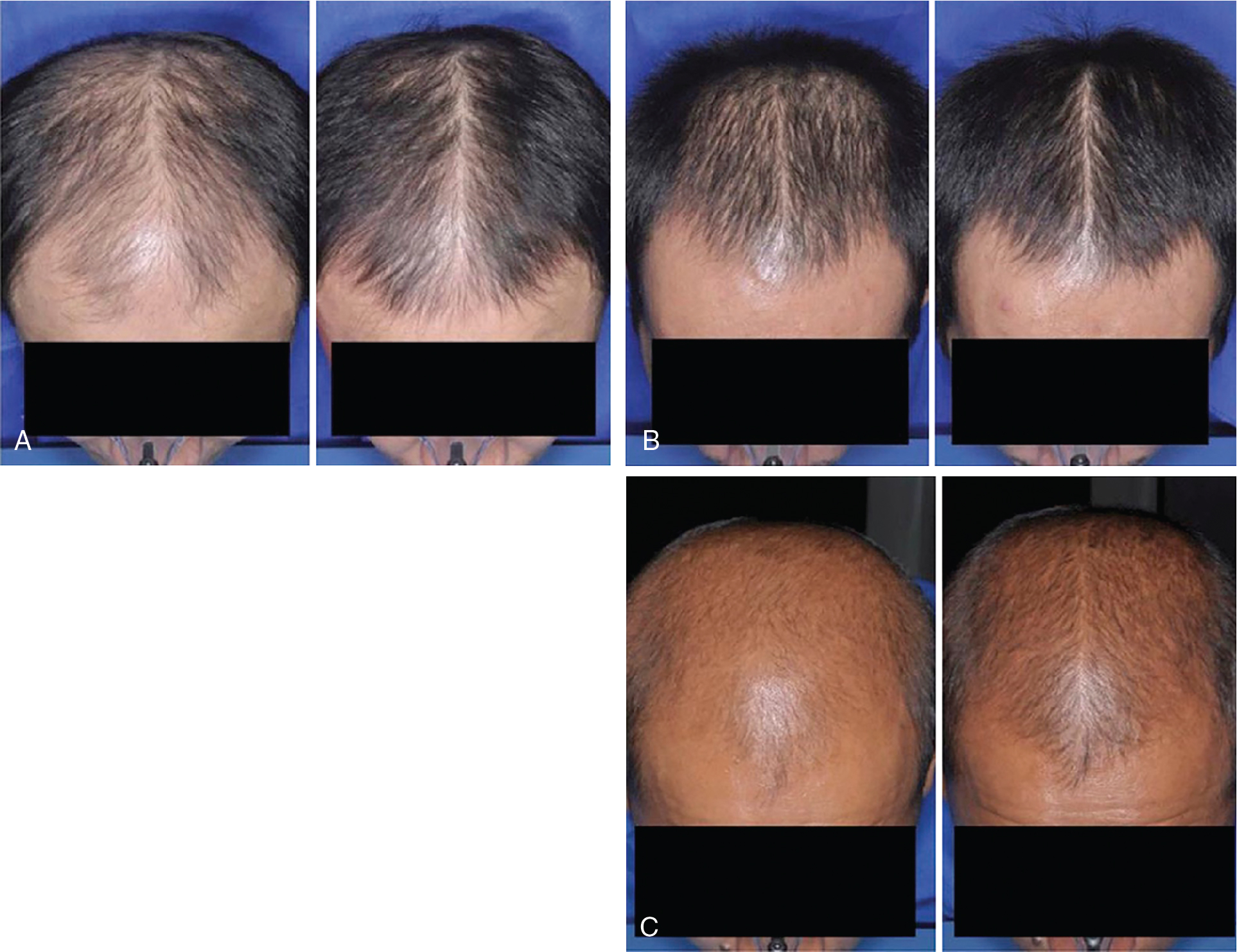
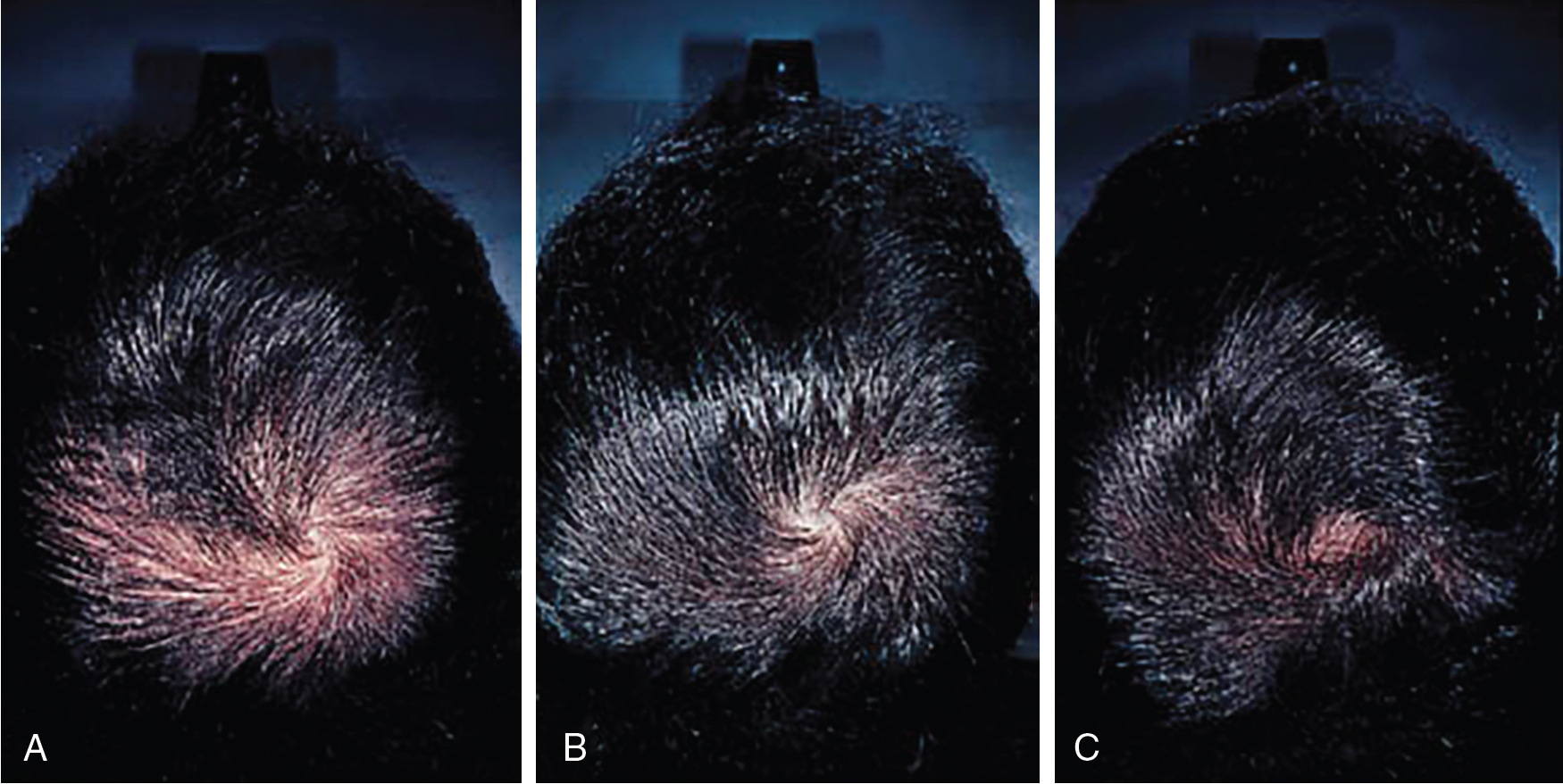
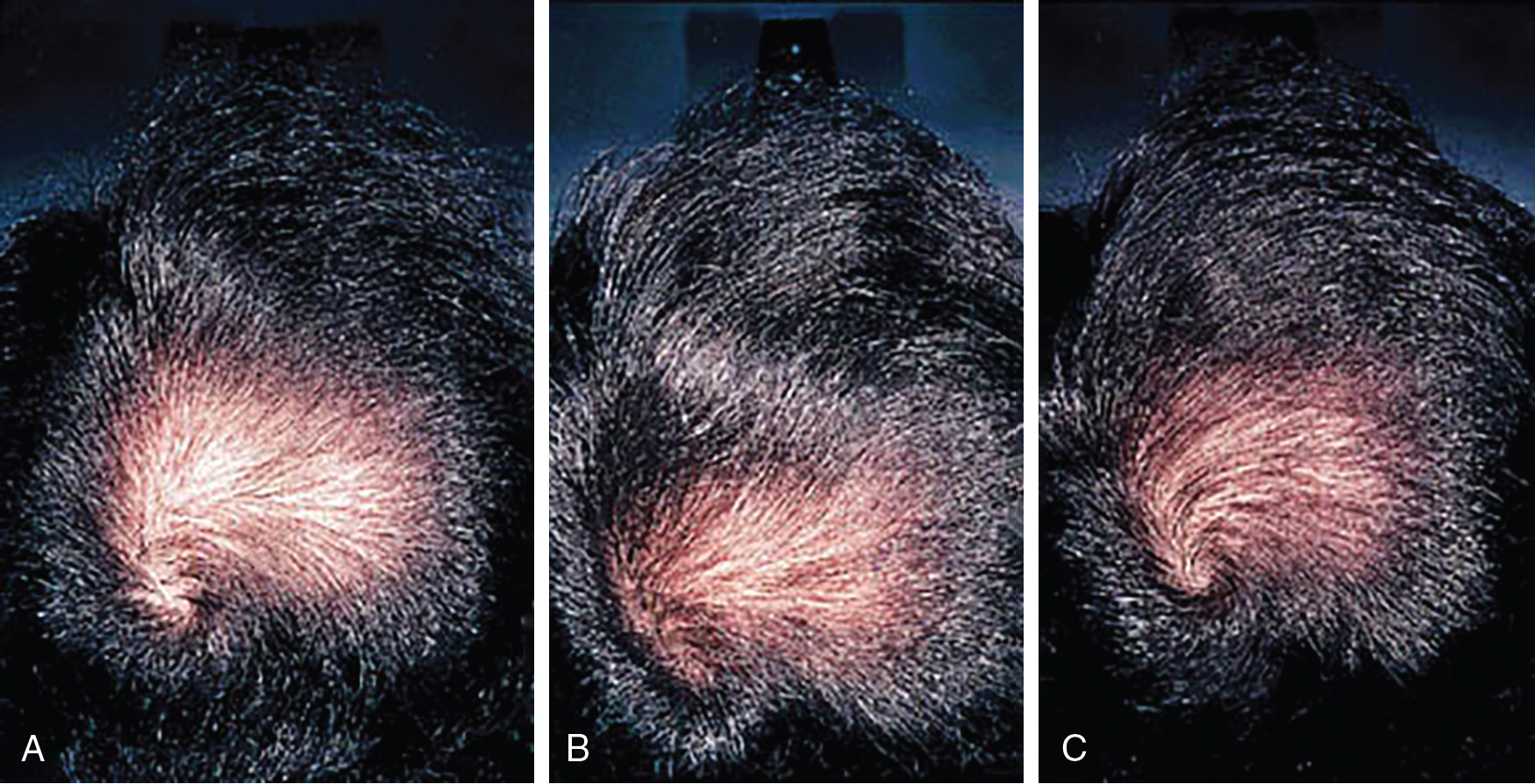
Systematic reviews of long-term use of finasteride 1 mg daily for treatment of male AGA report noticeable improvement in 30% of patients at 3 months of use (level of evidence: 1a). , Kanti et al. reviewed 25 studies assessing the efficacy of finasteride as a monotherapy for treatment of AGA in males. In all included trials, finasteride 1 mg daily led to a significant increase in total hair count compared to placebo. The mean change from baseline total hair count was 7.0 hairs/cm 2 in the frontal and centroparietal regions and 13.5 hairs/cm 2 in the vertex at 6 months (level of evidence: 1b). , , The mean increase from baseline total hair counts at 12 months was between 9.3 hairs/cm 2 and 9.6 hairs/cm 2 in the frontal and centroparietal region and 7.2 hairs/cm 2 and 36.1 hairs/cm 2 in the vertex (level of evidence: 1b). , In general, daily use for 3 months or more is necessary before benefit is observed ( Figs. 8.5 and 8.6 ). Continued use is recommended to sustain the benefit, as withdrawal of treatment leads to gradual hair loss and return to the pretreatment status within 1 year. , For greater efficacy, oral finasteride 1 mg may be combined with topical minoxidil.
In comparison with minoxidil and finasteride, fewer randomized controlled studies have analyzed the efficacy of dutasteride to treat AGA in males. In 2006, the first randomized controlled trial compared the effect of dutasteride 0.05, 0.1, 0.5, and 2.5 mg, finasteride 5 mg, and placebo in 416 men with AGA over 24 weeks (level of evidence: 1b). , Dutasteride 2.5 mg was shown to be superior over finasteride 5 mg in increasing hair count (level of evidence: 1b). , In 2014, a randomized controlled trial comparing dutasteride 0.02, 0.1, and 0.5 mg daily with finasteride 1 mg daily and placebo in 917 men over 24 weeks revealed that dutasteride 0.5 mg was statistically superior than finasteride 1 mg and placebo at increasing hair count after 24 weeks (level of evidence: 1b) ( Fig. 8.7 ). , Based on systematic reviews and meta-analyses, dutasteride may provide better efficacy compared to finasteride for treatment of male AGA (level of evidence: 1a). Further case series have shown efficacy of dutasteride in males with AGA who have failed prior treatment with oral finasteride over 12 months (level of evidence: 4). , ,