Key points
- •
Although most patients seeking hair restoration have androgenetic alopecia, there are several other potential etiologies of hair-loss disorders. It is important to exclude other causes of hair loss resembling androgenetic alopecia before considering cosmetic treatment.
- •
The comprehensive initial assessment of a patient seeking cosmetic hair restoration includes obtaining a full medical history, assessing risk factors for alopecia, performing a global and dermoscopic examination of the hair and scalp, as well as laboratory and histopathology assessments, when indicated.
- •
Effective tools that allow progressive assessment of response to cosmetic hair loss treatment include scalp photography and videodermoscopy.
Background, definitions, and history
The search for hair loss remedies has been ongoing for centuries. Ancient Egyptians documented the use of a concoction of ibex, crocodile, and snake fat to stimulate hair growth, and the pursuit of hair-loss treatments continues to present day (level of evidence: 5). Since the late twentieth century, several promising pharmacotherapeutic and procedural modalities for cosmetic hair restoration have become available. Although most patients seeking cosmetic hair restoration have androgenetic alopecia (AGA), there are several other potential etiologies for hair-loss disorders, and it is important to exclude other causes of hair loss resembling AGA before considering these treatment options. In this chapter, we will review the differential diagnosis of hair loss and the notable history and examination findings for each diagnosis.
The hair cycle
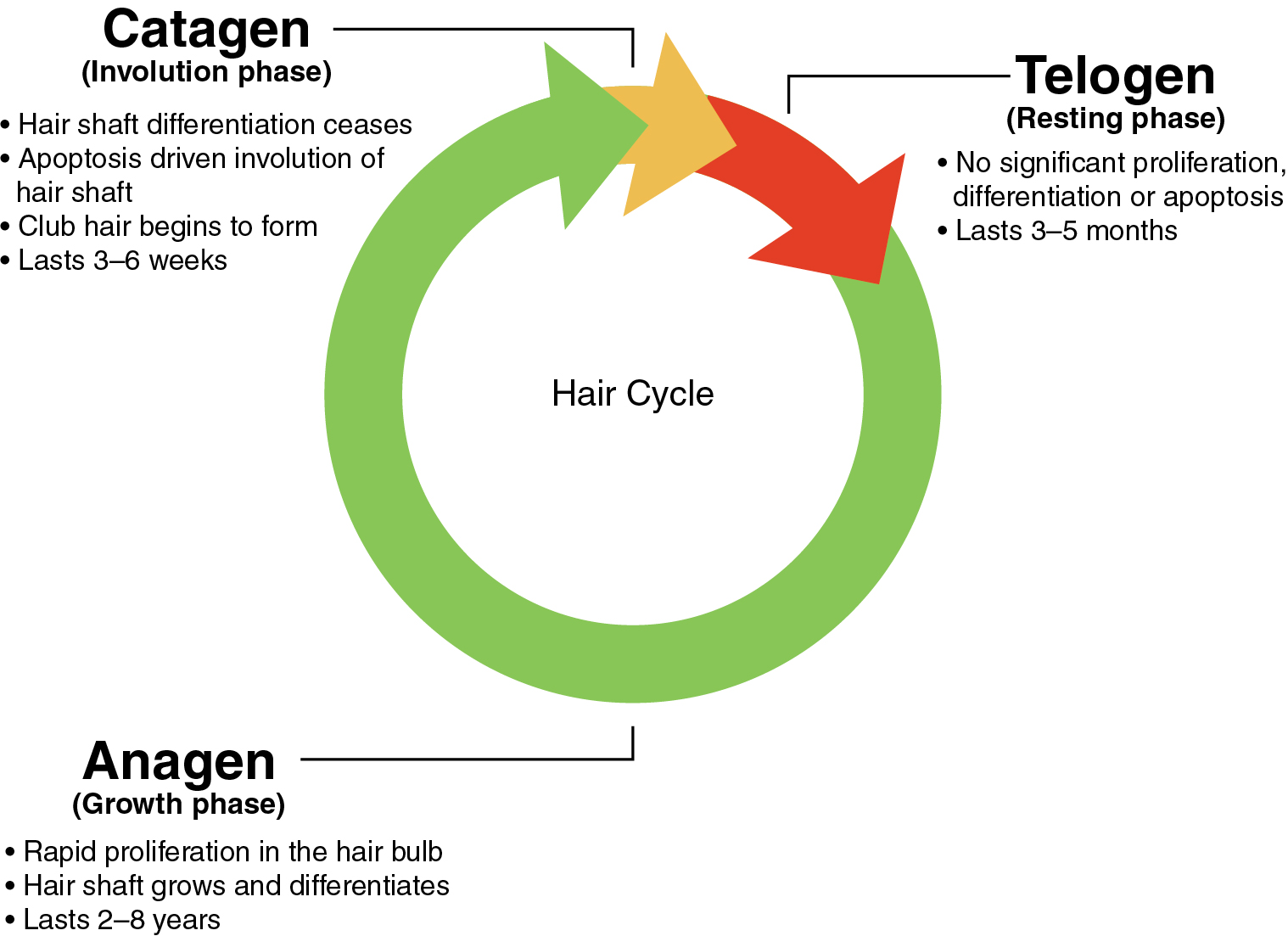
On a normal scalp, each hair follicle cycles independently; at any given moment, a percentage of hairs are growing, while others are resting and/or shedding. The majority of hair follicles are in the anagen or growth phase (86%–95%), a few are in the catagen or involution phase (1%), and a minority are in the telogen or resting phase (about 10%) (level of evidence: 5). The anagen phase lasts for about 2 to 8 years, followed by a catagen phase lasting 3 to 6 weeks. The cycle concludes with the telogen phase, lasting for approximately 3 to 5 months. An overly simplified but easily recalled summary of typical phase durations is “3 years, 3 weeks, 3 months.” During the telogen phase, the hair shaft matures and is eventually shed from the follicle, after which a new anagen phase may commence. As long as the percentages of follicles in each phase are stable, the density and total number of scalp hairs remain stable (level of evidence: 5). Any disturbance of the hair cycle may contribute to hair loss. The timing of this cycle also has implications for the expected timing of treatment response, often on the order of months ( Fig. 1.1 ).
Differential diagnosis of hair loss
Hair loss, or alopecia, is typically divided into two main categories: scarring and nonscarring. In scarring alopecia, the hair follicle may be irreversibly destroyed and replaced by fibrous scar tissue, leading to permanent hair loss, with minimal potential for hair regrowth. Scarring alopecia is less common overall, usually found in a minority of cases in specialized hair clinics (level of evidence: 5). In recent decades, however, there has been a rise in the prevalence of some scarring alopecias, for reasons that are poorly understood (level of evidence: 2b). This makes the identification of any features of scarring alopecia a paramount consideration in the evaluation of patients seeking cosmetic hair restoration.
Although AGA is the most common cause of alopecia in adult patients, other nonscarring or scarring alopecia diagnoses should be considered and ruled out before pursuing cosmetic hair restoration. The most common mimickers of AGA are outlined later and will be discussed in greater detail in subsequent chapters ( Fig. 1.2 ). In general, scarring alopecias, unlike AGA, may have symptoms, (classically itch or pain) and signs such as erythema, scaling, follicular papules, or areas of smooth follicular loss, leading to suspicion of scarring ( Pearl 1.1 ). Other nonscarring alopecias may differ in their distribution or pattern, timing, or severity.
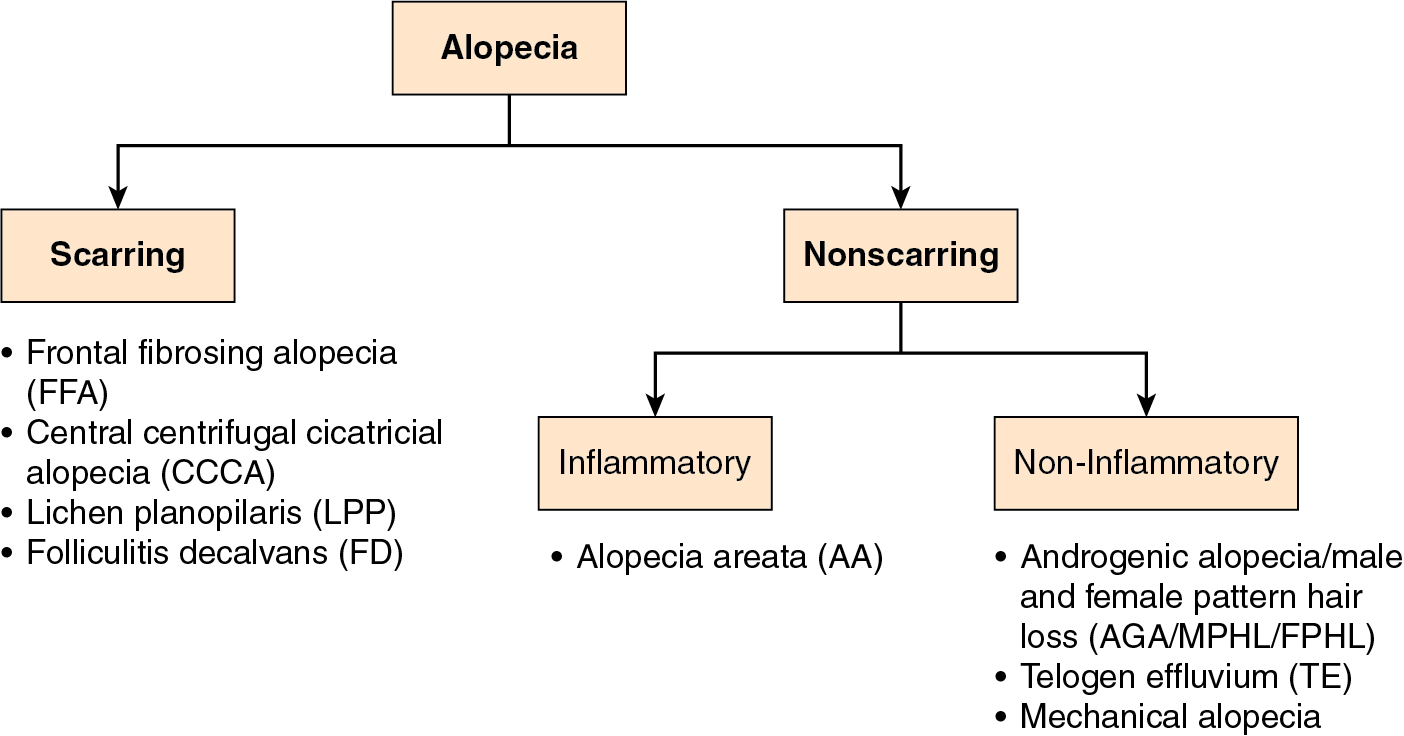
In general, scarring alopecias, unlike androgenetic alopecia, may have symptoms (classically itch or pain) and signs such as erythema, scaling, follicular papules, or areas of smooth follicular loss suspicious for scarring.
Nonscarring alopecia
Androgenetic alopecia
AGA, also known as male pattern or female pattern hair loss (MPHL/FPHL), is a nonscarring alopecia affecting a large proportion of adults over their lifetime. AGA affects approximately 50% of men by age of 50, and 50% of women by age 60 (level of evidence: 4). AGA is a genetically determined, polygenic trait. It is most prevalent in Caucasian men, who are four times more likely to develop premature balding compared with African American men.
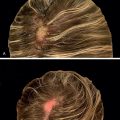
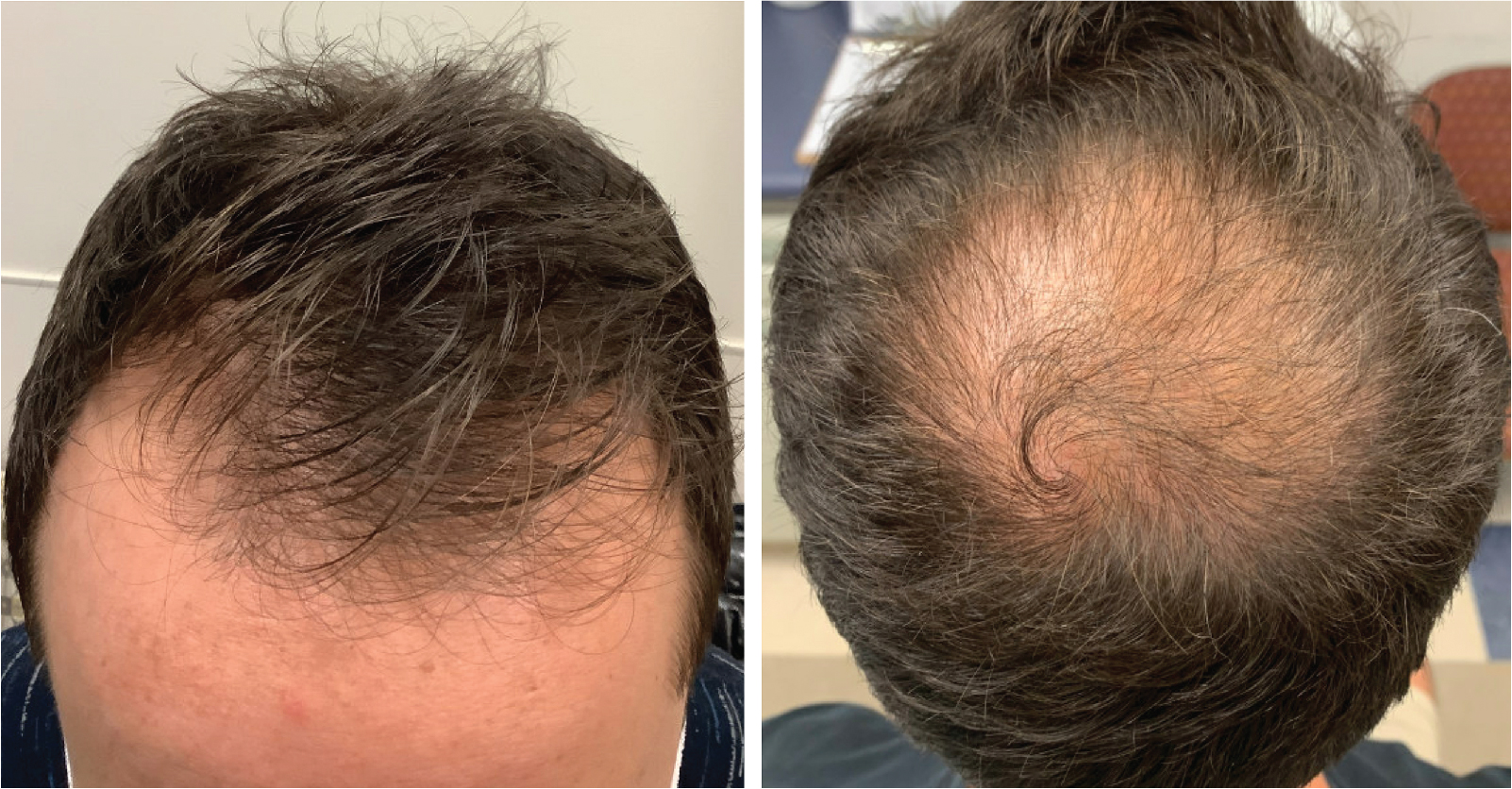
The pathophysiology of AGA involves progressive shrinkage of the hair follicles and shafts, called miniaturization ( Fig. 1.3 ). Hairs transition over time from a normal, terminal hair to a vellus-type hair, with a potentially dramatic reduction in hair-shaft diameter. There is also a concomitant shortening of the anagen phase, resulting in shorter hair lengths. Androgen (male hormone) effects appear to drive this process over time, forming the basis for antiandrogen therapies for AGA. For reasons not yet wholly elucidated, miniaturization is typically much more prominent on the frontal and vertex scalp, with occipital sparing, leading to the typical distribution patterns seen in AGA in men and women. This difference in miniaturization over the scalp provides the basis for follicular unit transplantation, whereby nonminiaturized, typically occipital-scalp hairs are transplanted to the thinned frontotemporal areas, where they retain their terminal status and may provide significant scalp coverage.

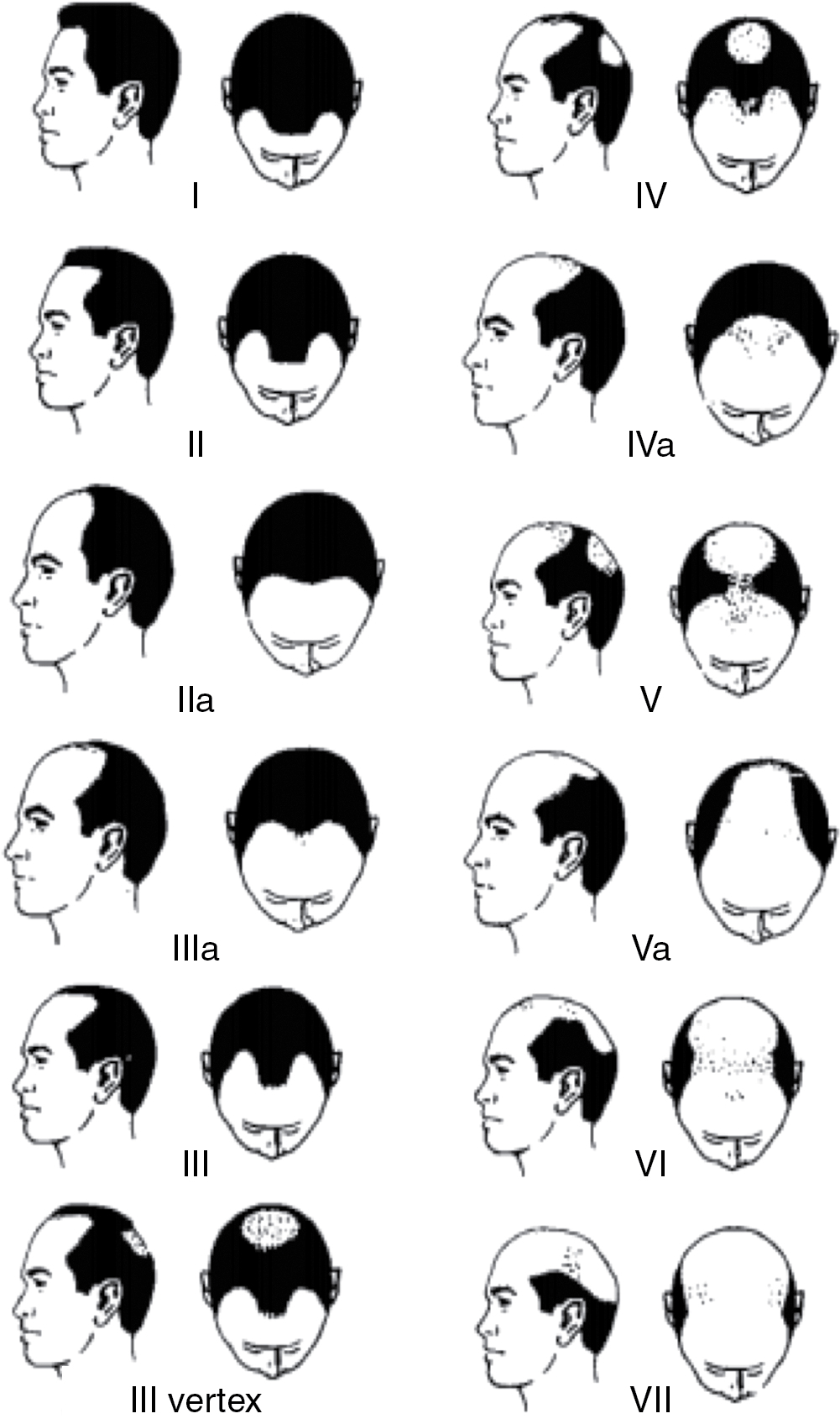
Individuals with MPHL experience varying degrees of hair loss in the bitemporal, frontal, mid-scalp, and vertex regions ( Fig. 1.4 ). The Hamilton-Norwood scale is the leading classification system used for grading MPHL, and it has been widely reproduced for use in clinical practice ( Fig. 1.5 ) (level of evidence: 3b).
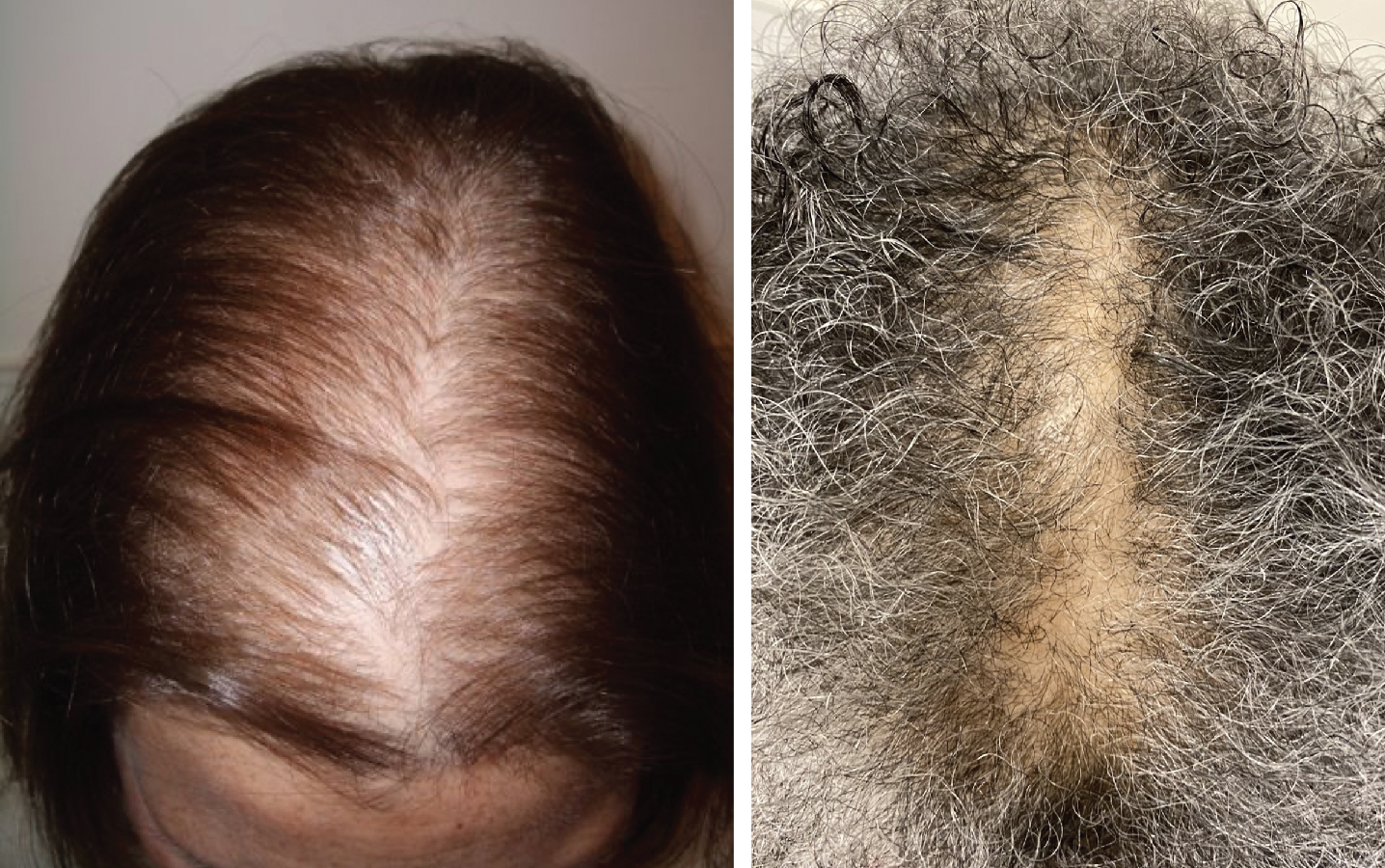
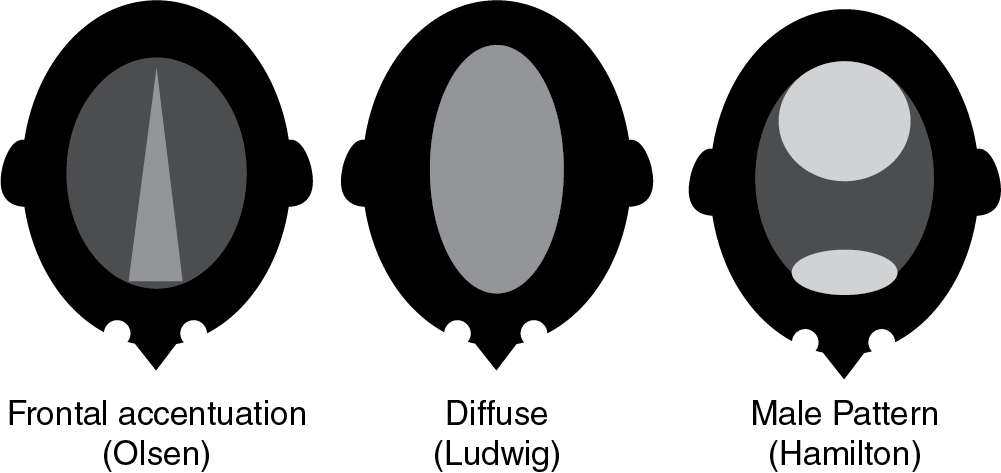
Women with FPHL typically experience diffuse hair loss in patterns described by Olsen, Ludwig, and Hamilton ( Figs. 1.6 and 1.7 ) (level of evidence: 5). The Olsen pattern describes accentuation of hair thinning in the anterior frontal scalp, giving the alopecic area a triangular-shaped appearance. The Ludwig pattern describes diffuse thinning of the crown region with frontal hairline preservation. Lastly, the Hamilton pattern has the classic distribution of MPHL, and an examination will reveal notable thinning in the lateral frontal part of the superior scalp and vertex ( Pearl 1.2 ).
In androgenetic alopecia, miniaturization is typically prominent on the frontal and vertex scalp, with occipital sparing, leading to the distribution patterns seen in androgenetic alopecia in men and women.
Telogen effluvium
Telogen effluvium (TE) is a form of nonscarring alopecia characterized by diffuse, often acute hair shedding. Common triggering events include medical stress (e.g., severe illness, surgical procedures), emotional trauma (e.g., divorce, bankruptcy, bereavement), parturition, discontinuation of oral contraceptives, and significant weight loss or a crash diet in the months prior to hair loss (level of evidence: 5). In TE, hair loss may also be a manifestation of a general medical problem. For example, patients may present with diffuse hair loss as a symptom of thyroid disorders, anemia or iron deficiency, connective tissue disease, or as a side effect of medication (with a wide range of potentially implicated drugs). Obtaining a full medical history is essential for evaluating the potential contribution of underlying medical conditions to hair loss.
Patients with TE will often complain of increased hair shedding leading to shower drain clogging or increased hair notable on a comb/brush and pillow. They will commonly present samples of shed hair for validation ( Fig. 1.8 ). TE and AGA frequently coexist, but the diffuse pattern and acuity or abruptness of onset, with the primary complaint of shedding rather than increased scalp visibility, may be clues in support of the TE diagnosis.

In many cases, TE is said to “unmask” AGA, whereby patients notice their underlying MPHL/FPHL more prominently in the setting of a recent or ongoing TE episode. Thus in many cases, treatments for AGA may be instituted at the same time TE triggers are addressed ( Pearl 1.3 ).
Telogen effluvium and androgenetic alopecia often coexist, but the diffuse pattern, acuity, or abruptness of onset, with the primary complaint of shedding rather than increased scalp visibility, may be clues to the diagnosis of telogen effluvium.
Alopecia areata
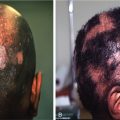
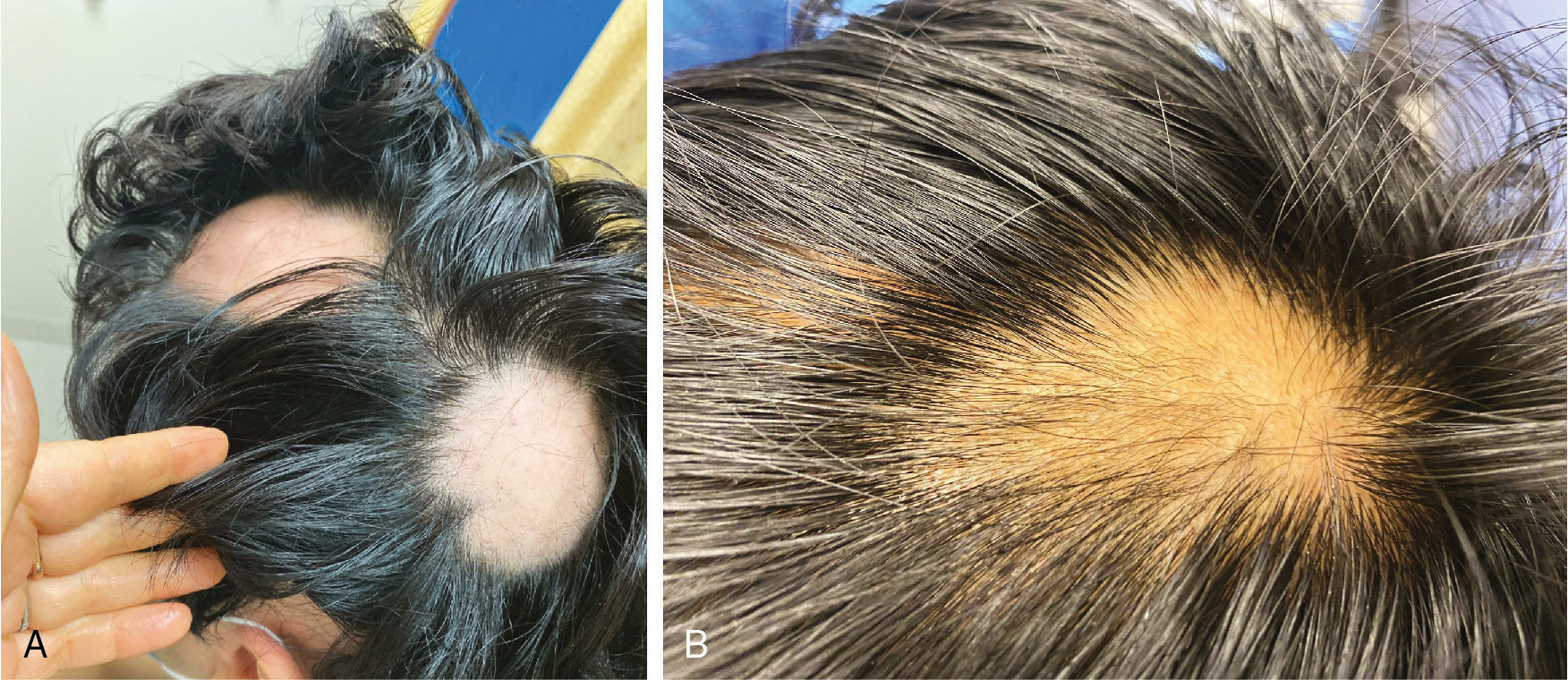
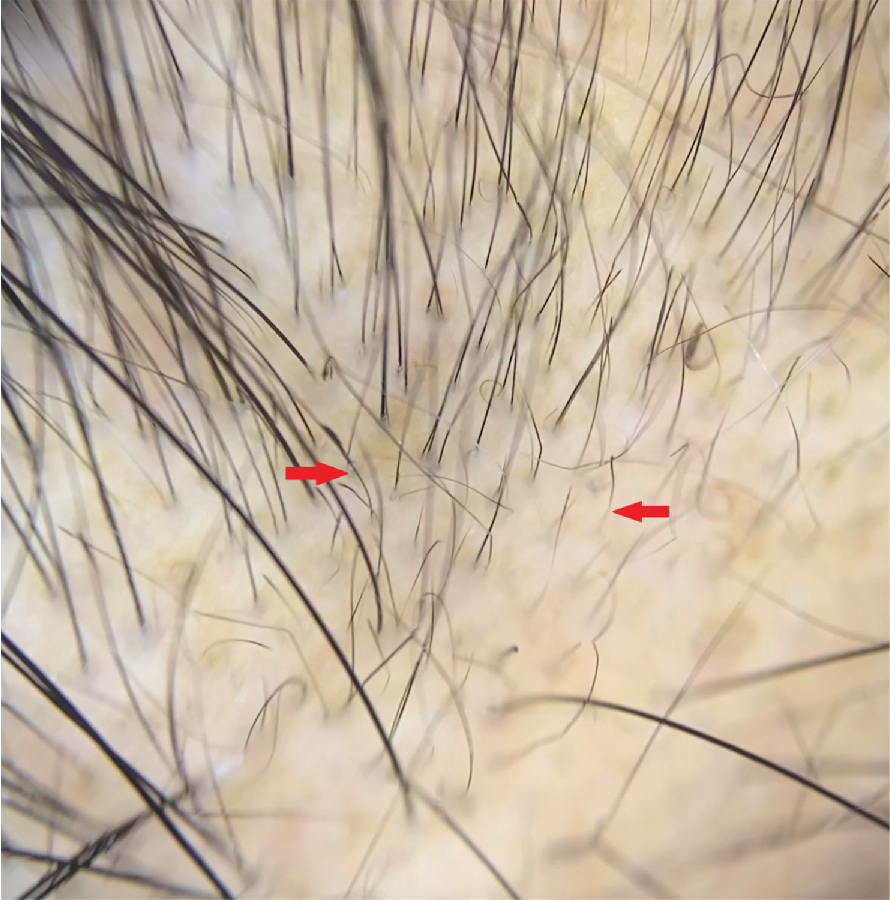
Alopecia areata (AA) is a nonscarring, autoimmune, inflammatory hair-loss disorder that may affect the scalp or any hair-bearing area. Hair loss in AA can take many forms, ranging from the classic presentation, with abrupt development of well-demarcated focal patches ( Fig. 1.9 ), to diffuse or total hair loss (alopecia totalis or universalis) (level of evidence: 5). Family history of AA or related autoimmune conditions can provide clues for this diagnosis. Extensive facial-hair loss (including eyebrows, eyelashes, or beard hair) should raise suspicion for this condition. Dermoscopic examination may reveal the classic “exclamation mark,” or proximally tapered short hairs of active AA ( Fig. 1.10 ).
Mechanical alopecia
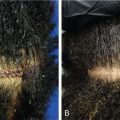
The hair loss seen in this type of alopecia is caused by the exertion of excessive pulling forces, leading to mechanical damage of hair follicles. This initiates as a nonscarring process, but with chronicity, it may become scarring. Traction alopecia, related to tight hairstyles, classically causes alopecia at the frontotemporal hairline, with a “fringe sign” of remaining short hairs at the anterior aspect ( Fig. 1.11 ) (level of evidence: 5).
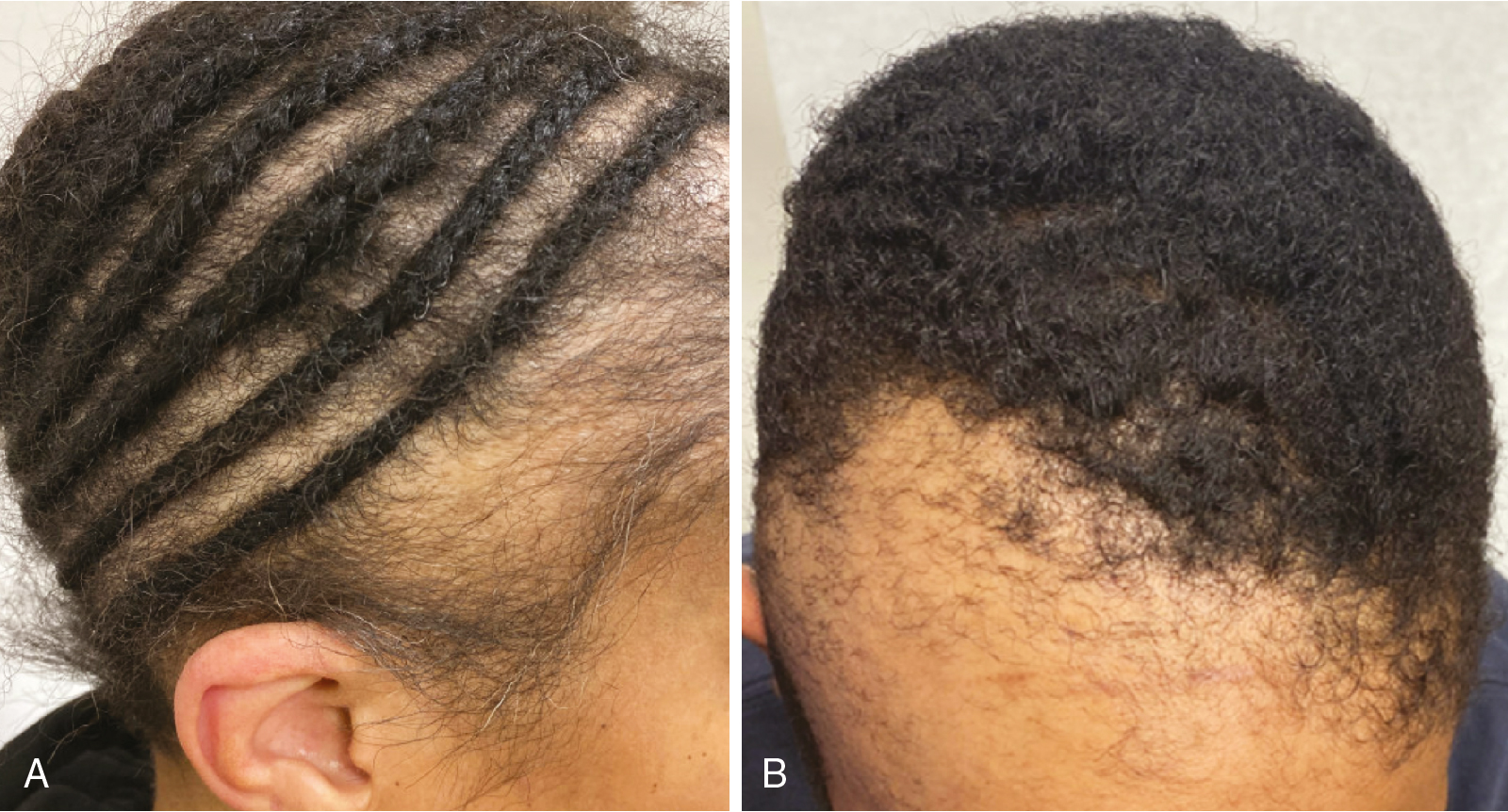
Patients with trichotillomania have an abnormal urge to pull hair, which can lead to irregular patches of alopecia, often geometric in shape. Nonscalp hair (brows, lashes, pubic) is commonly affected by trichotillomania in adults.
Scarring alopecia
Lichen planopilaris
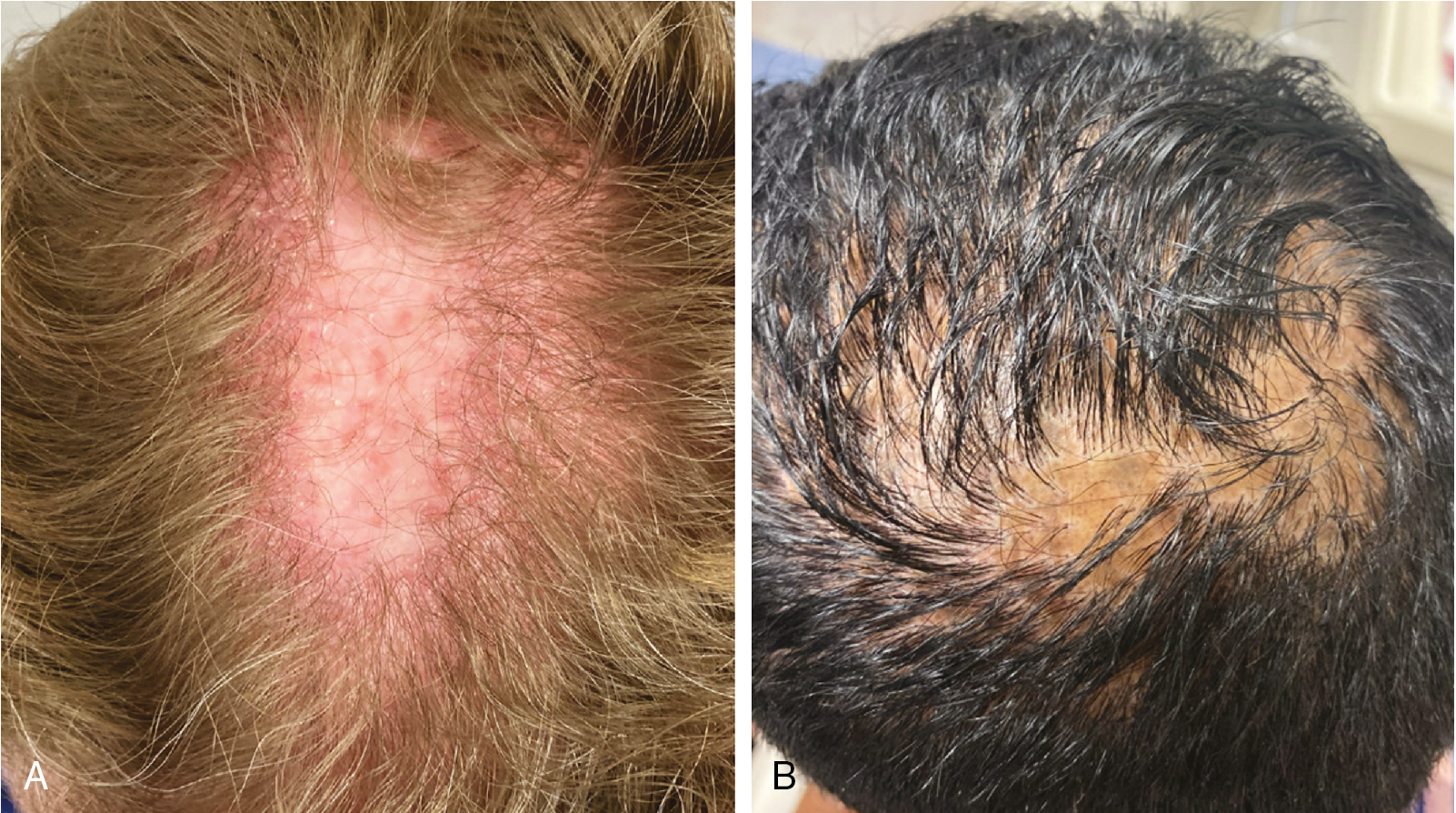
Lichen planopilaris (LPP) is an immune mediated scarring alopecia that is more common in women than in men. Patients present with patchy alopecic areas, and their examination reveals follicular keratotic plugs and/or perifollicular scaling along with perifollicular erythema ( Fig. 1.12 ) (level of evidence: 5). Common presenting symptoms in the acute phase of LPP include shedding, itching, scaling, burning, and tenderness. The classic form of LPP involves the vertex, although any region of the scalp may be affected.
Frontal fibrosing alopecia
Frontal fibrosing alopecia (FFA) may be considered a subset of LPP with a distinct clinical pattern. FFA is a scarring alopecia characterized by progressive symmetric frontotemporal hairline recession and marked decrease or complete loss of eyebrows ( Fig. 1.13 ) (level of evidence: 5). Close examination of patients with this condition may reveal mild skin atrophy and perifollicular erythema at the scalp margin. The classic “lonely hair sign” (level of evidence: 5) refers to remaining terminal hairs at the frontal hairline margin ( Fig. 1.14 ). This condition was initially described in postmenopausal Caucasian women but has increasingly been observed in younger women of all ethnic backgrounds and in male patients. The prevalence of FFA has increased dramatically since the 1990s, for unclear reasons (level of evidence: 4). Environmental triggers (such as skin contactants) have been theorized but not proven. This condition should be considered in patients presenting with extensive eyebrow loss and in male patients with loss of beard hair ( Pearl 1.4 ).
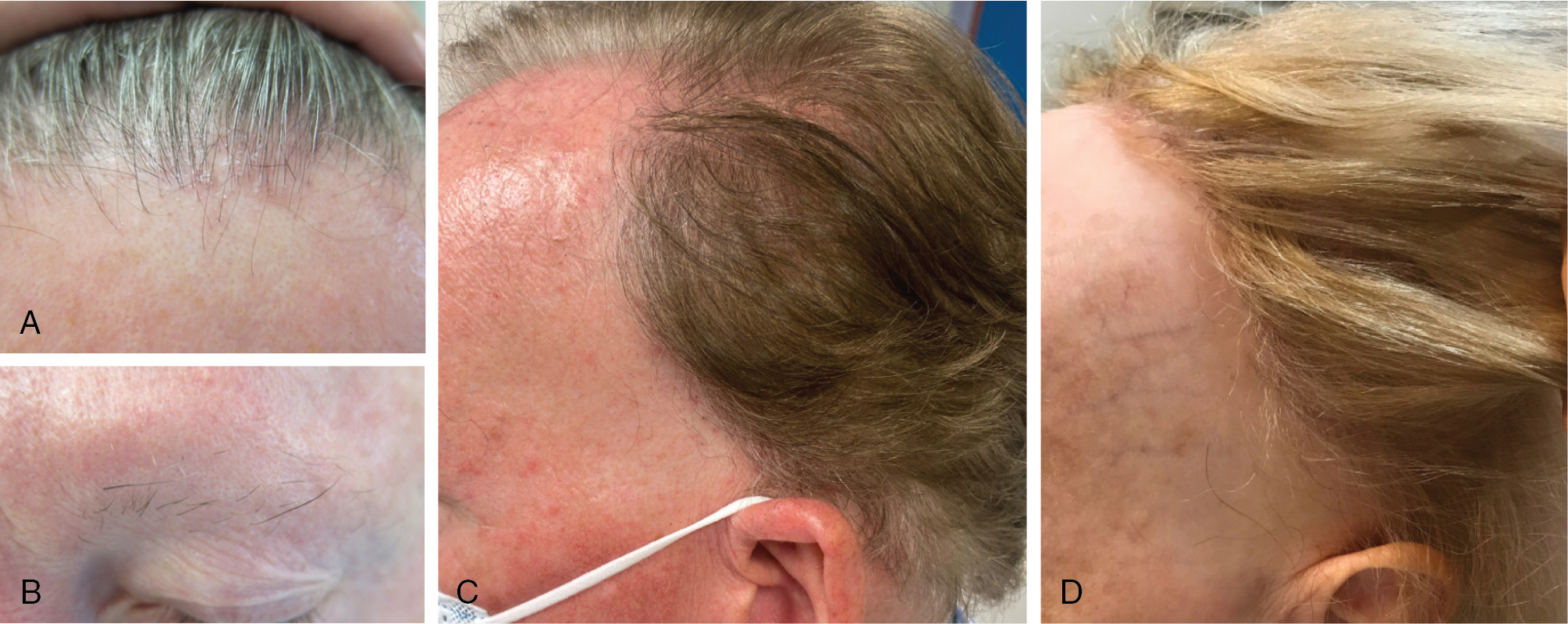
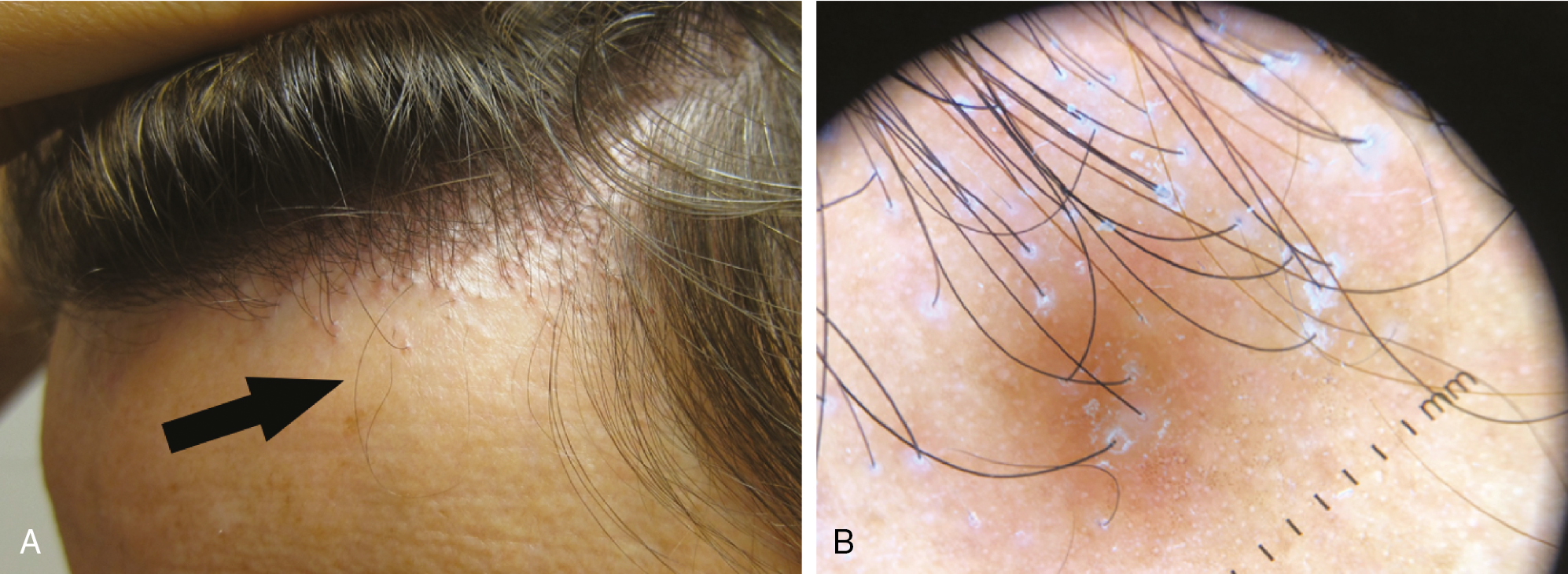
Frontal fibrosing alopecia should be considered in patients who present with prominent loss of eyebrows or beard hair.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree