Introduction
Worldwide, lower extremity trauma is associated with significant cost, as well as considerable morbidity and even mortality. In the United States, open tibial shaft fractures are most common in young adult males and typically result from motor vehicle collisions (MVCs). Quantifying the cost of such injuries is challenging. To improve understanding of the financial and medical implications of limb-threatening lower extremity trauma and its management, the Lower Extremity Assessment Project (LEAP) was of paramount importance. For both limb salvage and amputation patients, total lifetime costs were found to be remarkably high, though salvage was shown to offer higher utility and to cost less compared to amputation (by at least 93,606–154,636 USD for a 40-year-old patient). Lower extremity trauma is both common and costly, and an ongoing evolution in management has been instrumental to improving outcomes. It has become increasingly evident that a multidisciplinary approach is critical. Care by emergency medicine, general surgery, and vascular surgery teams is acutely required, followed by careful planning and coordinated efforts from the orthopedic surgery and plastic surgery teams. This “orthoplastic approach” can provide optimal and definitive care of limb-threatening lower extremity trauma. Importantly, the role of the plastic surgeon in the “orthoplastic approach” can involve reconstructive efforts for both limb salvage and limb amputation.
This chapter aims to review the history of lower extremity reconstruction, injury classification, early assessment and initial management, patient selection for limb salvage versus amputation, principles of skeletal fixation, principles of soft tissue reconstruction, and postoperative considerations.
History
Lower extremity trauma management has undergone significant progress over many centuries, periodically catapulted forward around times of war.
Amputation of the lower extremity is thought to have occurred as a form a punishment or in religious ceremony as far back as Neolithic times, based on archeological evidence. As a form of medical practice, however, amputation is first mentioned in the Hippocratic treatise On Joints (460–370 BC). The amputation of gangrenous extremities was described with surgical intent, recommending amputation be done distal to the “boundary of blackening,” thus limiting pain and blood loss. Later, around 25 BC–AD 50, Celsus promoted the concept of thorough debridement to healthy tissue followed by bone shortening with very deliberate management of the soft tissue at the stump. He described that “the face of the bone … is smoothed down, and the skin drawn over it: this must be sufficiently loosened in an operation of this sort to cover the bone all over as completely as possible.”
Further developments were described by Archigenes and Heliodorus (AD 100), who designed flaps for complete soft tissue closure after amputation of digits, though not specifically for the lower extremity.
The introduction of gunpowder to warfare in 1338 resulted in a drastic shift in injury patterns, greatly increasing the number of amputation cases that presented to military surgeons.
A paramount figure in surgical advancement, Ambroise Paré (1509–1590) rejected the widely held notion that gunpowder was poisonous and thus discouraged the common practice of dressing the wounds with caustic hot oil. Paré also refined the amputation technique by using vessel ligation rather than nonspecific cauterization. Introducing a pivotal concept, Paré was the first to consider amputation in a reconstructive manner. For instance, he considered the functional implications of the amputation location, recommending amputation five fingerbreadths below the knee to accommodate a wooden leg prosthesis, and he fashioned the first artificial limbs.
Acute management of lower extremity trauma shifted dramatically in the early 1800s. Through extensive experience managing combat injuries, Larrey of France and Guthrie of Great Britain noted that early or primary amputation (within the first 24 hours) of the lower extremity reduced mortality, hastened stump healing, and reduced infection and overall blood loss. , Indications for primary amputation of the lower extremity included compound fractures resulting from gunshot wounds, all fractures of joints, all gunshot wounds of joints, significant soft tissue loss (especially with critical nerve injury), and loss of major blood supply of an extremity.
The advent of ether and chloroform anesthesia in the 1840s permitted surgeons to prioritize technique over speed. Surgical hygiene also saw a significant advancement with Joseph Lister in 1867. Lister drew from Louis Pasteur’s research and posited that, in order to prevent infection, “septic organisms” must be eliminated from the wound and from anything coming into contact with the wound. His method of antisepsis involved spraying carbolic acid in the air overlying the surgical field and applying a carbolic acid-soaked dressing to the wound. Carl Thiersch and Richard von Volkmann of Germany adopted these antisepsis methods and, with them, demonstrated lower mortality rates, faster healing, and fewer complications. This contributed to the gradual acceptance of later developments, such as the use of steam sterilization technique, sterile gowns, and face masks.
At the time of the First World War (1914–1918), more powerful and accurate weapons were employed which caused significant mutilation and resulted in half a million amputations. High concentrations of bacteria in the battlefield soil resulted in frequent infections. Works from Ollier and Orr were integrated by Carrel into his practice of thorough debridement, plaster immobilization, antisepsis with 0.5% sodium hypochlorite (Dakin’s) solution, and delayed wound closure.
Subsequently, during the Spanish Civil War (1936–1939), Trueta synthesized the recommendations of preceding surgeons by treating open fractures with wound debridement, fracture reduction, packing the wound with sterile gauze, plaster cast application, and tetanus prophylaxis administration.
Through the Second World War (1939–1945) and the Korean conflict (1950–1953), indications for amputation remained largely unchanged from the time of Larrey and Guthrie. Improvements implemented over that period of time notably improved outcomes, with the mortality from lower extremity trauma dropping from 8% during the First World War to 4.5% in the Second World War. By the time of the Vietnam War, through the 1960s and early 1970s, compound fractures and gunshot injuries of the joints were no longer considered indications for amputation. Without many options for large soft tissue transfer, extensive soft tissue injury remained an indication for amputation, along with extensive bone trauma and nerve injury.
In 1975, Taylor harnessed the developing fields of bone reconstruction, vascular surgery, and microsurgery to perform a free vascularized bone graft. Distraction osteogenesis, pioneered by Codivilla and Ilizarov in the 20th century, was a means to provide stable fixation and bone stock without the donor site morbidity of a bone graft.
Soft tissue reconstruction progressed dramatically following the arrival of anesthesia in the 1840s. A surgical innovator, Hamilton performed the first cross-leg flap for lower extremity reconstruction in 1854 and is also credited with furthering the basic understanding of skin grafts that arose centuries prior. Daniel and Taylor applied emerging microsurgical techniques and performed the first free soft tissue transfer in 1973, using a free groin flap to the lower leg. Free tissue transfer for lower extremity reconstruction was a critical addition to the reconstructive armamentarium and has bourgeoned into a reliable method for limb salvage and outcome optimization.
Initial Management
Out of necessity, lessons learned during wartimes of the 19th and 20th centuries helped develop efficient methods of evacuation, stabilization, and early critical treatments of injuries in the military. An orthopedic surgeon, Jim Styner, helped to bridge this concept to the civilian world by devising a methodical, concise approach to trauma patient care with the Advanced Trauma Life Support (ATLS) program. The first ATLS course, held in 1978, championed the multidisciplinary approach, and encouraged process standardization to improve outcomes. Today, initial management of the lower extremity trauma patient should adhere to the ATLS protocol, focusing on respiratory and cardiovascular stabilization and neurological evaluation before specifically addressing the lower extremities. A stable airway should be ensured and breathing assessed. A circulatory exam should be performed, examining peripheral perfusion and acutely managing hemorrhage with pressure, splint application, or pelvic binders. Ideally, a neurological exam is performed prior to analgesia or sedation, with particular focus on peripheral motor and sensory nerve function of the lower extremity. Complete exposure of the patient with removal of clothing and dressings is critical and may reveal unexpected soft tissue injury. Any indicated imaging and laboratory tests should be ordered, and adequate intravenous access and a Foley catheter placed.
On secondary survey, a focused exam of the lower extremity is performed to consider the following:
- •
Skin : The area, size, and degree of soft tissue damage should be documented. The degree of contamination of an open fracture will guide administration of prophylactic antibiotics and tetanus immunization ( Table 43.1 ).
TABLE 43.1
Tetanus Prophylaxis Immunization Schedule
From Valenziano CP, Chattar-Cora D, O’Neill A, Hubli EH, Cudjoe EA. Efficacy of primary wound cultures in long bone open extremity fractures: are they of any value? Arch Orthop Trauma Surg . 2002;122(5):259–261.
Tetanus Immunization History
Clean,
Minor Wounds
Contaminated,
Complex Wounds
Td or Tdap
TIG
Td or Tdap
TIG
Unknown or <3 doses
Yes
No
Yes
Yes
≥3 doses
No a
No
No b
No
Td, Tetanus–diphtheria vaccine; Tdap, tetanus–diphtheria and acellular pertussis vaccine; TIG, tetanus immune globulin.
a Yes, if >10 years since the last tetanus booster.
- •
Nerve function : Motor and sensory function should be examined thoroughly throughout both lower extremities. Special attention should be given to the tibial nerve, where trauma can cause loss of plantarflexion and sensory loss to the plantar foot. The finding of an insensate plantar surface has historically been a key reason to selecting amputation rather than limb salvage. However, in a LEAP study, 55% of patients presenting with an insensate foot who underwent limb reconstruction ultimately regained sensation at 2 years after injury, indicating that an insensate foot is not an accurate indicator of the need for amputation.
- •
Vascular status : Peripheral pulses should be palpated and evaluated with Doppler. CT angiography is the imaging of choice to diagnose vascular injury, as they have been shown to have both high sensitivity and specificity in detecting traumatic arterial injury.
- •
Musculoskeletal : Muscle functions and musculotendinous integrity should be assessed. Compartments should be palpated and, in conjunction with the neurovascular exam, a determination should be made if compartment syndrome is suspected. Standard radiographs should be obtained to include a joint above and below the injury site.
Injury Classification
After the secondary trauma survey and review of the lower extremity imaging results, an assessment of the injury pattern can be described and classified according to various systems. The Gustilo and the Byrd classifications are used most commonly ( Table 43.2 ). Other systems include the Mangled Extremity Severity Score (MESS), which aims to predict which limbs should be salvaged rather than amputated. The MESS and other lower extremity injury-severity scores have failed to demonstrate clinical utility and should be used cautiously in determining if a lower extremity trauma patient is best served with an amputation or limb salvage.
| System | Grade | Soft Tissue | Bone |
|---|---|---|---|
| Gustilo | I | Wound <1 cm | Simple fracture No comminution |
| II | Wound >1 cm Minimal soft tissue damage | Moderate comminution | |
| III | Extensive soft tissue damage (ex: degloving, contusion, flaps) | Comminuted fracture | |
| IIIA | III with adequate soft tissue coverage | Comminuted fracture | |
| IIIB | III with extensive soft tissue loss Inadequate soft tissue coverage | Periosteal stripping Exposed bone | |
| IIIC | III with arterial injury requiring repair | Any open fracture pattern | |
| Byrd | I | Wound <2 cm Clean | Low energy Spiral or oblique fracture |
| II | Wound >2 cm Moderate adjacent skin/muscle contusion No devitalized muscle | Moderate energy Comminuted or displaced fracture | |
| III | Extensive skin loss Devitalized muscle | High energy Significantly displaced fracture Severe comminution Segmental fracture or bone defect | |
| IV | Crush, degloving, or associated vascular injury requiring repair | As III but extremely high energy |
Orthoplastic Approach
For patients with moderate-severe trauma injury, the transfer to a trauma center is warranted, with in-hospital mortality rate being significantly lower at trauma centers compared to non-trauma centers (7.6% vs. 9.5%), as well as a lower one-year mortality rate (10.4% vs. 13.8%). While the specific reason for the improved outcome of lower extremity trauma patients at trauma centers is uncertain, the availability of the multidisciplinary team is likely a significant factor.
To further optimize care and outcomes for lower extremity trauma patients, a combined approach by orthopedic and plastic surgery teams is preferred. Success in limb reconstruction requires the surgeon to abandon the tissue-specific perspective and to adopt a holistic approach, considering the extremity in totality. Indeed, decreased rates of complications and revision surgery were noted in patients with Gustilo grade IIIB open tibial fractures when treated at trauma centers with an orthoplastic team instead of a tertiary center without an orthoplastic team.
Compartment Syndrome
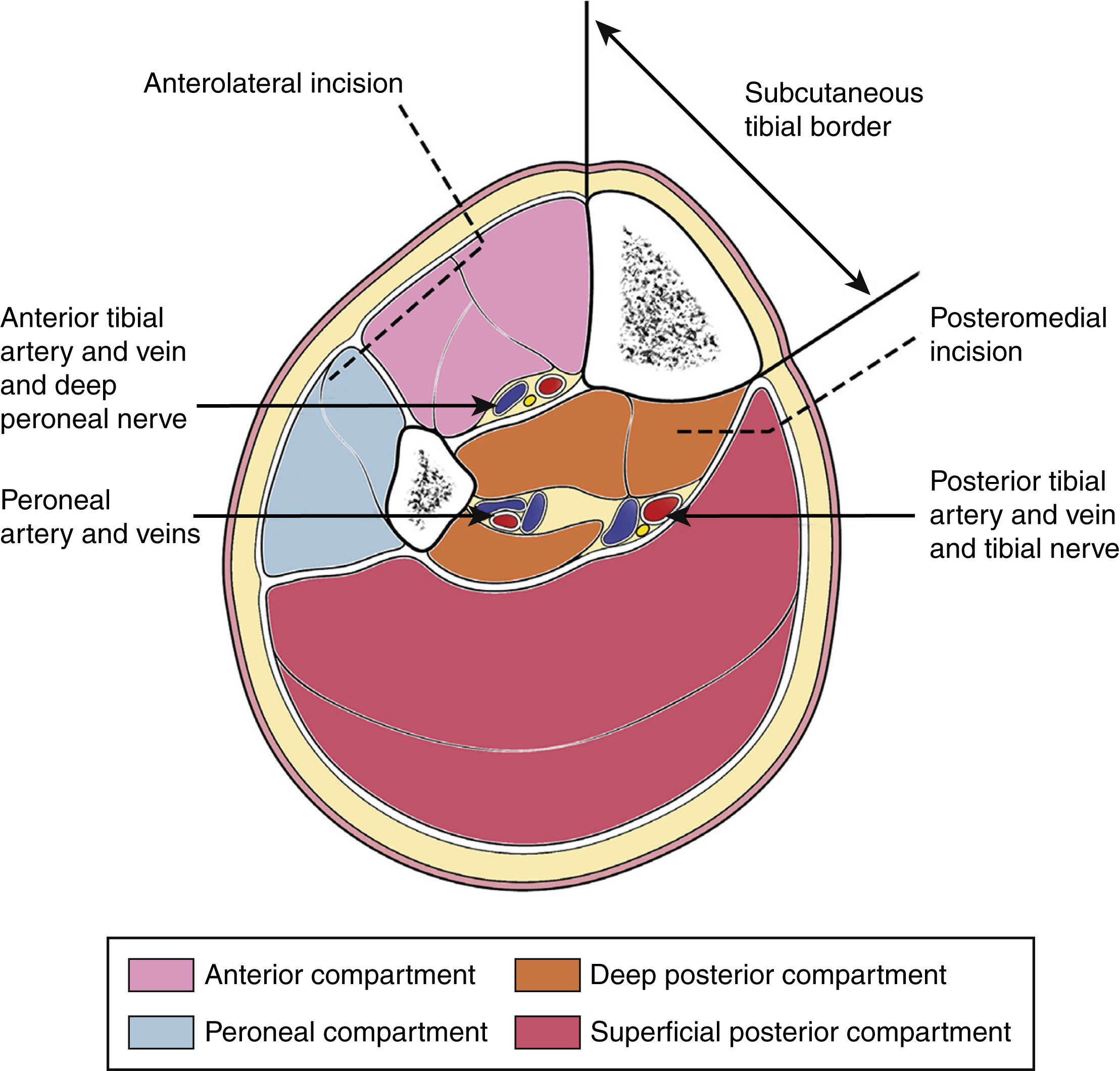
The five “Ps” (pain, pallor, paralysis, paresthesia, poikilothermy) suggest compartment syndrome, with paresthesia and disproportionate pain on passive stretch being the earlier signs. Traditionally, intracompartmental pressure (ICP) of 30 mmHg or less was an indication for fasciotomy. Newer evidence suggests that the differential pressure (diastolic minus compartment pressure) over 30 mmHg is a more sensitive indicator of compartment syndrome requiring fasciotomy. Fasciotomy incisions should be placed precisely to avoid compromising perforators for possible local fasciocutaneous flap and to avoid undue tibial exposure ( Figs. 43.1 and 43.2 ).

Vascular Injury
Evidence of arterial injury on CT angiography can include sudden narrowing of an artery, loss of arterial opacification, pseudoaneurysm formation, active contrast material extravasation, and arteriovenous fistula formation. An ischemic limb should have circulation restored ideally within 3–4 hours of injury to avoid irreversible muscle damage. Preoperative angiography should not delay revascularization attempts. When vascular disruption is present in combination with bone injury, the order of repair is debatable. On one hand, fracture reduction and skeletal fixation will require manipulation that can potentially jeopardize an adjacent, delicate vascular repair. On the other hand, prompt reversal of ischemia is critical to avoid muscle damage and to ensure limb salvage. No definitive evidence exists to indicate that surgical sequence has any impact on the rate of subsequent lower extremity amputation.
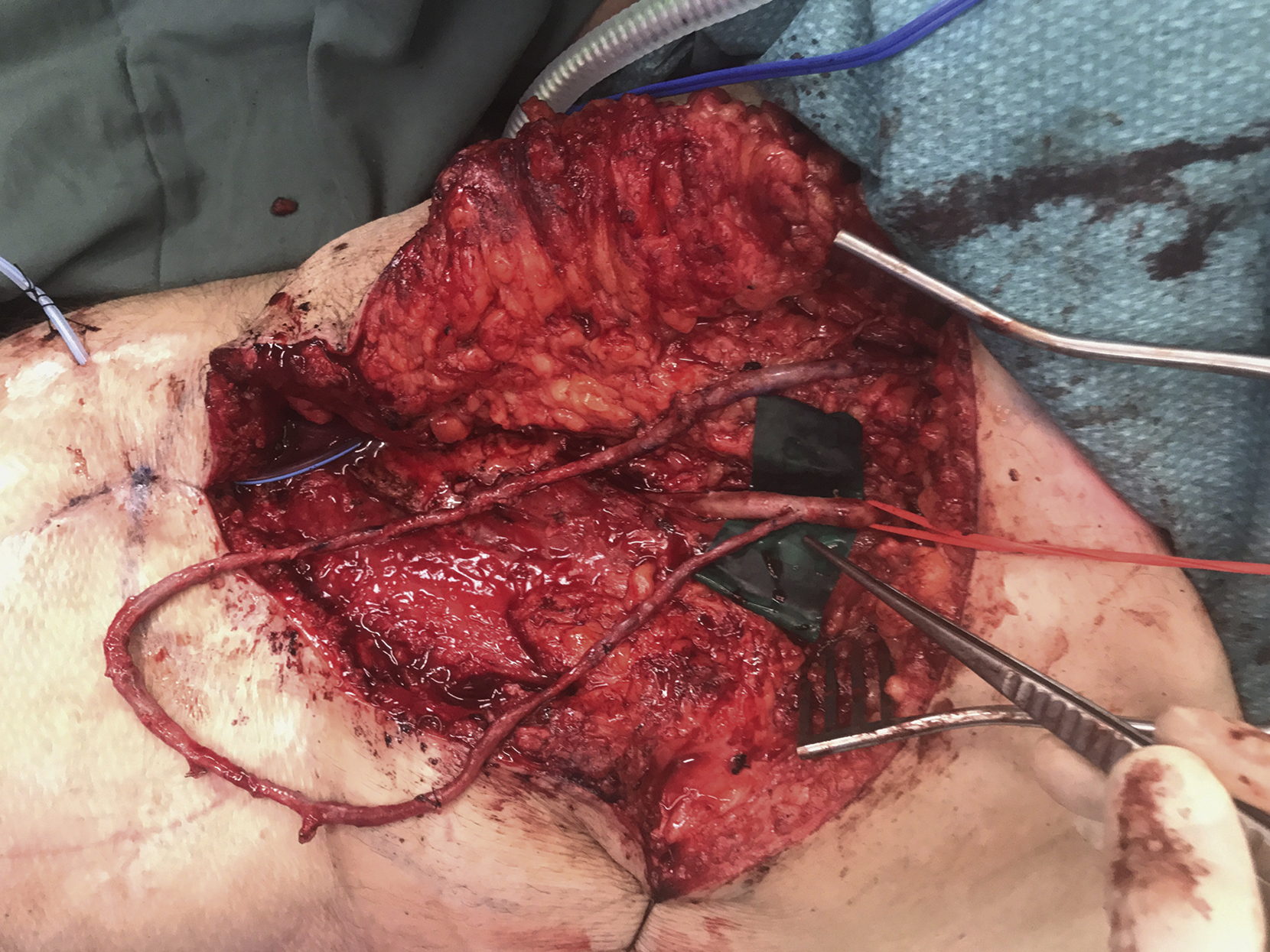
Free flap reconstructive options can be limited by the availability of native recipient arteries and veins in the traumatized lower extremity. To circumvent the shortage of peripheral recipient vessels, a temporary arteriovenous (AV) fistula can be created. The fistula is designed with a long, reversed vein graft anastomosed to a large artery at one end and a large vein at the other end. The vein graft is preferably harvested from the ipsilateral limb to minimize donor site morbidity. Alternatively, the Corlett loop technique, originally described by Mulholland et al in the neck, can be applied to the lower extremity. The ipsilateral lesser saphenous vein can be utilized “in situ” to create the fistula by preserving the proximal end where it merges into the popliteal vein, thereby avoiding a second microvascular anastomosis. The distal end of the vein is transposed to be anastomosed to an artery. Anastomosis of the vein can be performed to a major artery such as the popliteal artery in an “end-to-side” fashion or to a sural artery branch in an “end-to-end” fashion. The fistula loop should be positioned adjacent to the defect to be reconstructed ( Fig. 43.3 ). Ideally, the fistula location permits the microvascular anastomosis to be performed outside the zone of injury. Division of the fistula loop and anastomosis to the flap vessels can be performed as a single-stage surgery or in a delayed fashion. If this is done as a single stage, it is recommended to perfuse the AV fistula for one hour intraoperatively prior to division of the loop and anastomosis to the flap. A single-stage technique is beneficial as early thrombotic occlusion of the fistula can theoretically be identified immediately and anastomotic revision performed.