Left Lobe and Left Lateral Segment Transplantation in Children
Michael J. Englesbe
Amit K. Mathur
John C. Magee
DEFINITION
For children with end stage liver disease, liver transplantation is the only potential life-saving therapy. Children can receive a whole or partial allograft. The most common partial allograft done in children is a left lateral segment (segments II and III) transplant. A left lobe (segments II, III, and IV) liver transplant is another commonly used graft. The source of these organs can either be a living or deceased donor.
PATIENT HISTORY AND PHYSICAL FINDINGS
Priority for transplantation is driven by severity of illness. Children with acute liver failure receive priority. Objective measures of risk of death without a transplant (Pediatric End-Stage Liver Disease [PELD] score or Model for End-Stage Liver Disease [MELD] score) are used to determine allocation of organs.
Children with chronic liver disease should be listed for transplantation if they develop signs of decompensation, including growth failure, malnutrition, ascites, encephalopathy, frailty, or coagulopathy.
A complete assessment of the patient’s suitability for transplantation includes a discussion of the risks and benefits with the patient’s guardians. In addition, careful counseling regarding the rigors of posttransplant care is mandatory.
A thorough history and physical exam should be performed. Frequently, the specific etiology of liver disease (acute or chronic) cannot be firmly established. This circumstance is acceptable, assuming that specific contraindications to transplantation have been excluded. Physical exam should focus on the size of the spleen, liver, and presence of previous surgical incisions.
The majority of pediatric liver transplant candidates have had previous abdominal surgery, frequently for the treatment of biliary atresia. Careful review of previous operations is required prior to transplant surgery.
Contraindications include severe infection, extrahepatic malignancy, and anatomic considerations that would preclude adequate inflow or outflow of the allograft.
IMAGING AND OTHER DIAGNOSTIC STUDIES
Pretransplant assessment should include blood tests and serologies as well as an echocardiogram, chest x-ray, and duplex ultrasound of the liver.
If screening ultrasound suggests abnormal vascular anatomy or flow, further workup is required. Importantly, a recanalized, thrombosed portal vein may be mistakenly appreciated as “normal” on ultrasound.
Arterial and venous phase magnetic resonance imaging (MRI) or computed tomography (CT) scan (both require intravenous contrast) provide excellent anatomic assessment. Careful review of cross-sectional imaging by the transplant surgeon is mandatory to assure that the patient is a candidate for a partial allograft.
SURGICAL MANAGEMENT
Preoperative Planning
Diagnosis of abnormal arterial anatomy or flow will inform the surgeon to prepare for an aortic conduit at the time of transplant (see Chapters 29 and 30). Further, this finding may preclude a living donor allograft because arterial conduit may not be available.
Diagnosis of abnormal mesenteric or portal venous anatomy or flow (such as portal vein thrombosis) will allow the transplant surgeon to prepare for alternative methods for achieving venous inflow to the liver, including portal venous endovenotomy (see Chapter 25). If there is concern regarding adequate mesenteric venous inflow, a mesenteric angiogram with venous phase assessment or a direct mesenteric venogram (accessed via the intrahepatic portal vein or splenic vein) may be necessary. Abnormal mesenteric venous anatomy may preclude living donor transplantation because venous conduit may not be available.
Diagnosis of abnormal inferior vena cava (IVC) or hepatic venous anatomy may preclude a segmental allograft. Generally, a patent IVC is required.
Any patient with malignancy as the indication for transplantation (most commonly hepatoblastoma) requires a complete radiographic evaluation for operative planning. The possibility of metastatic disease must be considered.
Close collaboration with an experienced anesthesia team is critical for optimal care of the patient. Continuous communication between the surgical and anesthesia staff both preoperatively and throughout the transplant is required. The goal for a pediatric liver transplant is to extubate the patient upon completion of the operation. This requires judicious resuscitation as well as perfect operative technique. Over-resuscitation results in excess bleeding and congestion of the allograft. Under-resuscitation leads to poor perfusion of the allograft and other vital organs.
Selecting the Correct Allograft
In general, children have generous access to precious donor organs. Within this context, decisions regarding organ selection should carefully weigh the risks of waitlist mortality and the benefits of transplantation with the available allograft.
Patients requiring extensive vascular reconstruction, particularly involving the IVC, are best suited to receive a whole graft.
Selection of an appropriately sized allograft is absolutely critical for successful left lobe or left lateral segment liver transplant. Right lobe allografts, with their large anterior to posterior diameter, are not used in pediatric liver transplantation.
Anatomic considerations will determine the size of the liver that the recipient can accommodate. Patients with significant ascites can accommodate a larger allograft. Several convenient liver weight calculators are available.1,2,3
When the allograft size is greater than 4% of the body weight, it can be technically difficult to complete the anastomoses and to close the abdomen. Conversely, when the allograft size is less than 1.5% of the body weight in small children and less than 1% in larger children, allograft small-for-size syndrome can occur. If the allograft is split on the back table, direct visual comparison of the recipient abdomen and the allograft can help determine whether the left lobe or the left lateral segment can be used.
Positioning
The patient is placed in the supine position on the operating table with arms at sides for smaller children.
An arterial line and central line should be placed.
TECHNIQUES
INCISION AND RETRACTION
A bilateral subcostal incision is made.
Most children have had previous abdominal surgery and have portal hypertension. The first goal of the operation is to complete adequate dissection to enable placement of a fixed retractor. This usually includes adhesiolysis from the anterior and inferior aspects of the liver. Blunt dissection will cause excessive bleeding, thus electrocautery or Argon beam photocoagulation devices are the preferred methods for this portion of the operation. A needle tip electrocautery device is a helpful tool for this delicate work.
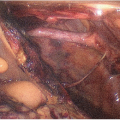
It is critical that both the surgeon and the assistant are able to use both hands during the entire operation. This demands exceptional retraction. For smaller children, a standard Bookwalter™ retractor provides excellent retraction (FIG 1). In older children, subcostal retraction is best achieved by an Omni™ or Thompson™ retractor.
The retractor should be placed in order to facilitate exceptional exposure of the suprahepatic vena cava. The subcostal retractor should only retract the ribs superiorly; retraction on the diaphragm will cause unnecessary compression of the lungs and heart.
Inferiorly, the retractor should pull the spleen, stomach, duodenum, and colon out of the visual field. Packing these viscera away with multiple packs underneath retractor blades is helpful. A retractor blade across the duodenum just underneath the porta hepatis provides critical retraction for the portal dissection.
PORTAL DISSECTION
Electrocautery and gentle dissection are the primary methods for this phase of the operation.
Frequently, recipients will have had a Kasai procedure. Preserve the Roux limb so that it can be used for the biliary reconstruction. The Roux limb is taken down from the portal plate and shortened with a gastrointestinal anastomosis (GIA) stapler, excluding the enterotomy from the previous biliary-enteric anastomosis.
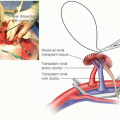
Dissection and isolation of the hepatic artery is the next step of the operation. The left hepatic artery is usually the easiest to identify first (FIG 2). The sutures on the ligated end of left hepatic artery are left long to facilitate identification later in the operation. The surgeon should follow the left hepatic artery to the bifurcation of the proper hepatic artery and then the right (and middle, if present) hepatic arteries. These vessels should also be ligated. The proper hepatic artery should then be
followed to the level of the gastroduodenal artery. The gastroduodenal artery should be left in continuity. Additional dissection may be required to facilitate clamping of the common hepatic artery during the transplant phase of the operation.

FIG 2 • The left hepatic artery is reliably identified on the superficial and inferior aspect of the porta hepatis. The portal dissection should begin here.
If a replaced right or left hepatic artery is present, retain as much length of these vessels as possible. Unlike a replaced right hepatic artery, a replaced left hepatic artery is rarely big enough to provide inflow to the liver.
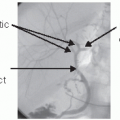
Next, dissect and isolate the portal vein (FIG 3). The portal vein can be extremely thin in children and treacherous to repair if injured during this phase of the operation. It should be left intact and will be divided later in the operation after the IVC dissection is complete. Within this context, it is critical that a conservative dissection of the portal vein is completed if there are dense adhesions from previous surgical procedures. Additional dissection toward the liver or pancreas can be completed later in the operation, just prior to clamping the portal vein.
RETROPERITONEAL DISSECTION
The liver is further mobilized by taking down the posterior attachments of the liver to expose the retrohepatic IVC.
At this point, excessive resuscitation significantly complicates the IVC dissection and leads to increased blood loss. Ideally, the child should have a central venous pressure less than 5 mmHg.
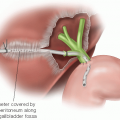
The dissection of the IVC off of the caudate lobe of the liver is best approached by alternating exposure from the right and left sides (FIG 4). Branches going to the liver are tied on the IVC side and can be clipped or cauterized on the liver side (FIG 5). Broad-based, accessory hepatic veins should be clamped using a small vascular clamp on the IVC and a single well-placed clip should then be applied to the liver side of this vein. The vein should then be divided with a knife and oversewn on top of the vascular clamp with small monofilament suture.
Commonly, the caudate lobe wraps circumferentially around the IVC, making this dissection treacherous. If making safe progress becomes extremely difficult or if there is significant bleeding from the IVC during this dissection, the IVC can be clamped above and below the liver. Children usually tolerate IVC clamping well, although careful communication with anesthesia colleagues is critical during this phase of the operation. Additional resuscitation of the patient may be necessary
to make sure that the patient tolerates the preload reduction associated with clamping.
If the IVC is clamped, scissors can be used to divide small hepatic veins as the caudate lobe is dissected from the IVC. Small branches in the IVC can then be oversewn without bleeding. It is easy to narrow the IVC in small children, so as little IVC wall as possible should be used when oversewing these branches.
COMPLETION OF THE HEPATECTOMY
If the IVC was clamped during the caudate dissection, progress should be made more quickly.
At this point, it is usually advantageous to complete the portal vein dissection so that there is ample length for clamping and later anastomosis. The portal vein clamp is then carefully placed to assure that it is flat (FIG 6), and the portal vein is divided as close to the liver is possible.
There is minimal role for a portacaval shunt during these cases. Most children tolerate clamping of the portal vein without significant mesenteric congestion. In addition, complete clamping of the IVC is usually necessary in children to allow for adequate visualization for the hepatic vein anastomosis, thus the portacaval shunt would not be effective. If mesenteric congestion does occur, it can significantly limit exposure for the transplant operation.
The liver is then isolated on the hepatic veins (FIG 7). If not already done, the IVC above and below the hepatic veins should be clamped. The hepatic veins should be divided, leaving as much length as possible. This usually requires dividing the veins within the hepatic parenchyma (FIG 8).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree