Tumor necrosis factor α (TNF-α) is an inflammatory cytokine that is released by a variety of cell types, including immune effector cells and tissue-specific cells. TNF-α has been shown to promote inflammation via the activation and induction of cytokines interleukin 1 (IL-1), IL-6, and IL-8 and by the upregulation of adhesion molecules on endothelial cells leading to increased leukocyte extravasation. Theoretically, the blockade of TNF should have widespread potential in the treatment of numerous inflammatory diseases. Currently, 3 TNF-α inhibitors available in the United States are approved for psoriasis and psoriatic arthritis: infliximab, etanercept, and adalimumab. Numerous case reports and case series have been published in recent years reporting the off-label uses of these drugs in various inflammatory skin diseases. This review summarizes the most recent reports on 20 such conditions.
Tumor necrosis factor α (TNF-α) is an inflammatory cytokine that is released by a variety of cell types, including immune effector cells, such as macrophages, monocytes, lymphocytes, and neutrophils, as well as tissue-specific cells, including keratinocytes and dendritic cells. TNF-α has been shown to promote inflammation via the activation and induction of cytokines interleukin 1 (IL-1), IL-6, and IL-8 and by the upregulation of adhesion molecules on endothelial cells leading to increased leukocyte extravasation. Theoretically, the blockade of TNF should have widespread potential in the treatment of numerous inflammatory diseases. Currently, 3 TNF-α inhibitors available in the United States are approved for psoriasis and psoriatic arthritis: infliximab, etanercept, and adalimumab. Numerous case reports and case series have been published in recent years reporting the off-label uses of these drugs in various inflammatory skin diseases. This review summarizes the most recent reports on 20 such conditions.
Infliximab (Remicade)
Inflizimab is a chimeric IgG 1 monoclonal antibody against TNF-α that is comprised of the human constant (Fc) region of human IgG and the murine variable (Fab) region that binds to soluble and membrane-bound TNF-α, therefore preventing binding of TNF-α to its receptor. In addition, it also fixes complement and causes apoptosis of cells with cell surface TNF. It is currently approved by the US Food and Drug Administration for the treatment of psoriasis, psoriatic arthritis, Crohn disease and associated fistulas, rheumatoid arthritis (RA), ulcerative colitis, and ankylosing spondylitis. It is contraindicated in patients with a murine protein sensitivity and should be avoided in patients with known or recent malignancies as well as congestive heart failure and multiple sclerosis. The most common side effect is infusion-related reactions, which occur in 16% of patients and have been linked to the presence of antichimeric antibodies. Symptoms include fever, chills, urticaria, chest pain, hypotension, hypertension, and shortness of breath. This infusion reaction risk can be decreased through the concomitant use of methotrexate, azathioprine, or corticosteroids, preventing the formation of antichimeric antibodies. Infections such as pneumonia and sepsis as well as opportunistic infections such as histoplasmosis have been reported, and because of the ability of the drug to inhibit granuloma formation, mycobacterial infection or reactivation is of concern. Therefore a baseline purified protein derivative (PPD) is necessary, and many physicians opt to obtain a chest radiograph before imitation of treatment. There has been a proven increased risk of lymphoproliferative diseases and malignancies as well as onset or flares of multiple sclerosis. Clinical systemic lupus erythematosus (SLE)-like syndromes have been reported with the formation of antinuclear and antinuclear-DNA antibodies. This process has been shown to be reversible on cessation of the agent. Dermatologic conditions such as the paradoxic new onset of psoriasis, cutaneous small vessel vasculitis, and interstitial granulomatous dermatitis have all been reported. Infliximab is pregnancy category B.
Etanercept (Enbrel)
Etanercept is a receptor fusion protein consisting of the extracellular domain of the TNF-α receptor fused with the Fc portion of human IgG, which binds to and inhibits soluble and, to a lesser degree, cell membrane-bound TNF-α. It has 2 p75-binding sites, which confers it a higher affinity for TNF-α compared with the natural receptor. It does not fix complement, cause antibody-dependent cytotoxicity, or trigger T-cell apoptosis compared with infliximab. Etanercept is given as a self-administered subcutaneous injection at a dosage of 25 mg twice weekly for RA, ankylosing spondylitis, and psoriatic arthritis or 50 mg twice weekly for 3 months with a subsequent decrease to 50 mg weekly for psoriasis. Entanercept is contraindicated in patients with sepsis or with known hypersensitivity to the medication and should be avoided in patients with a history of an active infection, malignancy, multiple sclerosis, and unstable cardiac disease. Injection site reactions were the most common adverse events in initial clinical trials and occurred in up to 40% of patients. In trials there was an increased rate of upper respiratory infections. Also, there is a debate on the oncogenic potential of etanercept with regard to lymphoma occurrence; some reports show a 3-fold increase. There is an increase in antinuclear antibody formation, with a small series of patients developing signs and symptoms of SLE; this is readily reversible. The rate of development of antietanercept antibodies has been less than 10% and has not been observed to lead to decreased efficacy. Etanercept is pregnancy category B, with no evidence of harm to the fetus in animal studies. However, the drug has not been specifically studied in pregnant women nor is it known if it is secreted in breast milk. Salmon and Alpert have suggested that etanerept and other TNF antagonists be avoided in the first trimester.
Etanercept (Enbrel)
Etanercept is a receptor fusion protein consisting of the extracellular domain of the TNF-α receptor fused with the Fc portion of human IgG, which binds to and inhibits soluble and, to a lesser degree, cell membrane-bound TNF-α. It has 2 p75-binding sites, which confers it a higher affinity for TNF-α compared with the natural receptor. It does not fix complement, cause antibody-dependent cytotoxicity, or trigger T-cell apoptosis compared with infliximab. Etanercept is given as a self-administered subcutaneous injection at a dosage of 25 mg twice weekly for RA, ankylosing spondylitis, and psoriatic arthritis or 50 mg twice weekly for 3 months with a subsequent decrease to 50 mg weekly for psoriasis. Entanercept is contraindicated in patients with sepsis or with known hypersensitivity to the medication and should be avoided in patients with a history of an active infection, malignancy, multiple sclerosis, and unstable cardiac disease. Injection site reactions were the most common adverse events in initial clinical trials and occurred in up to 40% of patients. In trials there was an increased rate of upper respiratory infections. Also, there is a debate on the oncogenic potential of etanercept with regard to lymphoma occurrence; some reports show a 3-fold increase. There is an increase in antinuclear antibody formation, with a small series of patients developing signs and symptoms of SLE; this is readily reversible. The rate of development of antietanercept antibodies has been less than 10% and has not been observed to lead to decreased efficacy. Etanercept is pregnancy category B, with no evidence of harm to the fetus in animal studies. However, the drug has not been specifically studied in pregnant women nor is it known if it is secreted in breast milk. Salmon and Alpert have suggested that etanerept and other TNF antagonists be avoided in the first trimester.
Adalimumab (Humira)
Adalimumab is a human recombinant IgG 1 monoclonal antibody against human TNF-α. It binds to soluble and membrane-bound TNF-α, with apoptosis of cells with membrane-bound TNF occurring. Like infliximab, it fixes complement and causes lysis of cells expressing membrane-bound TNF-α. It was initially approved for the treatment of RA and then for psoriatic arthritis, ankylosing spondylitis, and Crohn disease, and is administered as a 40-mg dose subcutaneously or intravenously every other week or weekly. An initial loading dose of 80 mg is used to increase rapidity of response onset. It is contraindicated in patients with known sensitivity reactions and should be avoided in patients with active infections or malignancies as well as multiple sclerosis or congestive heart failure. Live vaccines should be avoided. Mycobacterial infections are a concern and therefore all patients need a baseline PPD. Injection site reactions are the most common adverse event and occur in 20% of patients in clinical trials. In clinical trials for RA, antiadalimumab antibodies were seen in 12% of patients receiving adalimumab as monotherapy and 1% of patients receiving concomitant methotrexate. Antinuclear antibodies seem to be increased in patients being treated with adalimumab, with some patients experiencing SLE-like symptoms. New onset psoriasis as well as cutaneous small vessel vasculitis have been known to complicate therapy. Adalimumab is pregnancy category B and its lactation profile is unknown.
Off-label uses of TNF-α antagonists
Sarcoidosis
Sarcoidosis is a multiorgan system idiopathic granulomatous disease that affects the lungs, skin, bone, and other organs. TNF-α is believed to play a key role in the pathogenesis of sarcoidosis. In pulmonary sarcoidosis, increased production of TNF-α by alveolar macrophages is seen and TNF-α plays an essential role in the process of granuloma formation. Genetic polymorphisms in the TNF-α promoter are also associated with specific clinical subtypes of sarcoidosis. More than 35 cases of sarcoidosis, including cutaneous, pulmonary, hepatic, and gastrointestinal types, have been reported to be treated with infliximab. Meyerle and Shorr reported the use of infliximab for sarcoidosis. A typical dosing regimen was intravenous infliximab at a dose of 3 to 5 mg/kg as an induction at baseline, week 2, and week 6, followed by maintenance therapy every 4 to 8 weeks thereafter. An improvement was often noted after the second or third infusion and many patients were able to significantly reduce their concomitant corticosteroid requirements.
Using etancercept in the treatment of pulmonary sarcoidosis, Utz and colleagues reported 5 of 17 treatment successes, not as promising as infliximab. Khanna and colleagues reported a patient with lupus pernio and arthritis who was treated with etanercept, 25 mg twice weekly, with improvement in her skin and joint disease within 2 months. The patient’s concomitant prednisone and hydroxychloroquine were discontinued by 3 months. Complete remission was reported to be maintained for 18 months on a regimen of etanercept, 25 mg twice weekly, and methotrexate, 5 mg once weekly.
Phillips and colleagues reported the successful treatment of cutaneous sarcoidosis with adalimumab at 40 mg weekly added to the initial treatment of hydroxychloroquine and prednisone. Remission was seen by 9 weeks. Heffernan and Smith reported a case of recalcitrant cutaneous sarcoidosis that showed extensive improvement after adding adalimumab 40 mg once weekly to hydroxychloroquine and pentoxifylline, with results seen at 5 weeks and continued improvement at 10 weeks.
Granuloma annulare
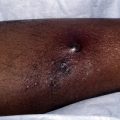
Granuloma annulare (GA) is a relatively common skin disease that affects the dermis and subcutaneous tissue. Although most cases resolve spontaneously and without epidermal sequelae, the disease process often results in loss of elastic tissue and leaves behind atrophic lesions. The cause of GA is unknown but it has been speculated to occur after certain inciting factors, such as trauma, insect bites, sun exposure, or psoralen and ultraviolet A therapy. It is postulated that a delayed type hypersensitivity reaction to an unknown antigen is the inciting event, with a subsequent Th1, interferon γ (IFN-γ) producing, lymphocyte reaction causing matrix degradation. Multiple different clinical subtypes of GA exist and they include localized, generalized, patch-type or macular, subcutaneous, perforating, GA in human immunodeficiency virus (HIV) disease, as well as GA associated with malignant disease. Spontaneous resolution of GA occurs within 2 years in 50% of cases, but there is a 40% recurrence rate. Often patients report resolution status following lesional skin biopsy, and because most lesions are asymptomatic no treatment is required. However, generalized cases represent a therapeutic hurdle. The use of TNF-α antagonists has been reported in the literature, although no randomized, double-blinded, placebo-controlled studies have been performed. One case study reports the use of infliximab for the treatment of generalized GA recalcitrant to previous treatments. The patient received infliximab 5 mg/kg intravenously at 0, 2, and 6 weeks and then every 4 weeks thereafter for 4 additional months. A significant improvement was noted by week 2, and by week 6 most lesions had resolved. The patient remained disease free at 1 year. There has also been a case report of the successful use of etanercept for the treatment of generalized GA. The dosing regime was 50 mg biweekly, for a total of 12 months. By 12 weeks of treatment, the patient had cleared mostly all lesions, with only erythema remaining. The patient was still disease free 12 weeks after discontinuing treatment.
Necrobiosis lipoidica diabeticorum
Necrobiosis lipodica diabeticorum (NLD) is characterized by firm depressed yellowish waxy lesions that start as erythematous papules then later develop into sclerodermalike lesions. Sixty percent of patients with NLD have diabetes mellitus, and 20% have glucose intolerance or a family history of diabetes. It is postulated that immunologically mediated vascular disease is the primary cause of the altered collagen found in NLD and that the microangiopathic vessel changes seen in diabetic patients could also contribute to the collagen degeneration and dermal inflammation. There have been no double-blind placebo-controlled studies to assess treatment efficacy in NLD; first-line therapy includes topical corticosteroids. There have been a few case reports of NLD treated with TNF-α antagonists. Because TNF-α is important in granuloma formation, these antagonists are believed to act in blocking this formation. Cummins and colleagues published a case report of a patient with generalized NLD that was recalcitrant to prednisone and dapsone. The patient was then treated with surgical split thickness autografting followed by prednisone 0.5 mg/kg/d and etanercept at 25 mg twice weekly that was commenced 6 days after surgery. The etanercept was continued for 16 months and the prednisone for 12 months. The patient’s lesions cleared and remained disease free at 2 years of follow-up. Another patient with refractory NLD with a single plaque on her shin was treated with intralesional etanercept at 25 mg weekly. The injections were dermal spaced at 1 cm each on the surface of the lesion. At 1 month an initial improvement was noted, with continuing resolution during the next 8 months. A third case report described a patient with an isolated ulcerated lesion of NLD that was unresponsive to multiple treatments. The patient was treated with an infliximab infusion at 5 mg/kg. By the second infusion improvement of the lesion was noted; however, the patient developed military tuberculosis. This case serves to reinforce the importance of pretreatment screening for tuberculosis exposure. Although infliximab was discontinued, this patient’s NLD continued to improve thereafter.
Pyoderma gangrenosum
Pyoderma gangrenosum (PG) is an uncommon and recurrent ulcerative disease that is neither infectious nor gangrenous. There are 4 clinical forms: ulcerative, bullous, pustular, and superficial. Initial lesions often start out as a pustule on an erythematous base, or an erythematous nodule or bulla, whereas the classic well-formed lesion is an ulcer with a necrotic undermined border. Histologically, in early PG there is a neutrophilic vascular reaction, which can be folliculocentric, and in untreated lesions a neutrophilic infiltrate often with leukocytoclasia can be seen. Although the disease is idiopathic in nature, it is hypothesized that an underlying immunologic aberration is the cause because of the association of PG with systemic autoimmune processes. Approximately 50% of patients with PG have an associated disease with inflammatory bowel disease, Crohn disease, and ulcerative colitis, being the most commonly associated. It is estimated that between 1.5% and 5% of patients afflicted with inflammatory bowel disease develop PG. Other associated conditions include leukemia, acute and chronic myelogenous leukemia, myeloma, monoclonal gammopathy of the IgA type, polycythemia vera, myeloid metaplasia, chronic active hepatitis, hepatitis C, HIV, SLE, pregnancy, PAPA (pyogenic sterile arthritis, PG, and acne) syndrome, and Takaysu arteritis.
The treatment options for PG are numerous, and the therapeutic approach depends on the number, size, and expansion of lesions, as well as the associated underlying disease. The goal is to reduce the inflammatory process of the wound and to control the underlying disease process. The standard treatment of PG is local or local and combined systemic corticosteroid therapy with or without additional systemic therapy. TNF-α inhibitors have been used with success in PG, especially those associated with inflammatory bowel disease. The published treatment regime for infliximab is 5 mg/kg intravenously at weeks 0, 2, and 6. For adalimumab, it is 80 mg subcutaneously as an initial dose then 40 mg subcutaneously weekly or every other week, and for etanercept the recommended dosing is 50 to 100 mg subcutaneously weekly either in 1 or 2 doses.
The largest case series of patients with PG and inflammatory bowel disease treated with infliximab included 13 patients. In this report, 3 patients had a complete response with infliximab induction alone, whereas 10 patients responded to infliximab induction and maintenance infusion therapy every 4 to 12 weeks. All patients who were also treated with corticosteroids were able to discontinue them after infliximab induction. Eleven patients in this study, however, had to remain on azathioprine and 6-mercaptopurine.
Ljung and colleagues reported a study of 8 patients with PG and associated Crohn disease treated with infliximab; 3 patients experienced completed resolution of the PG and 2 patients experienced partial remission. Many other case reports have been published documenting the treatment of PG with infliximab. Most patients noticed an improvement after the first infusion, with remission occurring within 3 months. A prospective trial of infliximab by Kaufman and colleagues for the use of extraintestinal manifestations of Crohn disease included 4 patients with PG. The mean duration of their PG was 5.5 years. All 4 responded to infliximab, with partial or complete resolution of the PG after 2 or 4 infusions; no adverse events were noted.
McGowan and colleagues published a case report of successful treatment of widespread PG unassociated with inflammatory bowel disease with etancercept. The patient experienced worsening PG while being treated with systemic corticosteroids. She was then started on etanercept 25 mg twice weekly along with prednisone 30 mg orally daily. At 1 month, complete resolution of the PG was noted. However, when the prednisone was tapered to 2.5 mg daily a recurrence was noted. Hydrocortisone intramuscularly was used, and prednisone 60 mg daily was started. Etanercept was increased to 100 mg weekly, which resulted in rapid clearance of the lesions, and the patient remained disease free at 4 months on etancercept and a low concurrent dose of prednisone (5 mg daily).
Hubbard and colleagues reported a more complicated case of PG in a patient with skin ulcerations and abscesses in the spleen and psoas muscle that were believed to be related to the PG. Infliximab was started and the patient was noted to have a rapid improvement of the skin lesions and abscesses. However, the patient suffered an anaphylactoid reaction during the fourth infliximab infusion. Infliximab was discontinued and the PG and abscesses returned. At this time, etanercept was initiated at 25 mg 3 times weekly, although at 3 weeks of treatment no improvement was seen. Etanercept was discontinued and treatment with adalimumab at 40 mg weekly was initiated. On adalimumab the patient experienced rapid resolution of the skin and extracutaneous lesions. Goldenberg and Jorizzo as well as Pastor and colleagues published case reports of the successful use of etanercept for PG.
Sweet syndrome
Sweet syndrome, or acute febrile neutrophilic dermatosis, is an uncommon disease with an unknown pathogenesis. A hypersensitivity reaction is proposed suggested by the association with infections, autoimmune diseases, inflammatory bowel diseases, and malignancies. It can also be drug induced or pregnancy related. Up to 50% of patients have idiopathic disease. Initial lesions are tender, erythematous plaques or papules that may enlarge and coalesce. There is associated edema of the lesions and some patients may develop vesiculation and bulla formation. Constitutional signs and symptoms such as fever and malaise are noted. Histologically, a dense perivascular neutrophilic infiltrate with edema is seen, and leukocytoclasia with minimal to no evidence of vasculitis is seen. Sweet syndrome follows a benign course that may last for weeks to months and then involute spontaneously. The most effective therapy is oral prednisone at 0.5 to 1.0 mg/kg/d for 4 to 6 weeks. Increased TNF-α level was reported in lesions of Sweet syndrome, and a study of successful treatment with etanercept of 2 patients with Sweet syndrome with RA has been reported. Etanercept 50 mg twice weekly was used in 1 patient and 25 mg twice weekly in the other patient. Improvement was noted in both patients; however, it is possible that improvement of the patients’ underlying RA may have also contributed.
Subcorneal pustular dermatosis
Subcorneal pustular dermatosis (SPD), or Sneddon-Wilkinson disease, is a chronic sterile subcorneal pustular disease that occurs mainly in middle-aged women. The pustules are superficial and annular or serpiginous in configuration especially on the abdomen, axillae, and groin. Some cases are associated with a monoclonal gammopathy, usually IgA. Histologically, the pustules form below the stratum corneum, with possible spongioform pustules in the upper epidermis. Dapsone, at doses of 50 to 200 mg/d, is effective for most cases. Grob and colleagues reported increased TNF-α levels in the serum and blister fluid of a patient with SPD, and so TNF-α inhibitors are believed to be effective in SPD by disrupting leukocyte adhesion and migration and preventing the accumulation of neutrophils. A report of a dapsone-intolerant patient with SPD was published by Voigtlander and colleagues. This patient was successfully treated with infliximab at 5 mg/kg in addition to acitretin at 0.4 mg/kg/d. Two days after the first infusion of infliximab the patient experienced complete resolution of pustules. The patient had a recurrence of symptoms at 14 days and received a second infusion of infliximab with subsequent resolution. At 6 months the patient remained free of disease on acitretin 0.16 mg/kg/d with the addition of systemic steroids for 1 flare.
Vasculitis
The vasculitides are a heterogeneous group of disorders similar in their inflammatory destruction of blood vessels. Numerous case reports have been published on the use of TNF antagonists in the treatment of diseases such as Churg-Strauss syndrome, Takayasu arteritis, giant cell arteritis, Kawasaki disease, and leukocytoclastic vasculitis.
The mainstay of treatment of most patients with systemic vasculitis is corticosteroids and concurrent immunosuppressant drugs. As discussed earlier, the anti-TNF agents are particularly useful in the treatment of granulomatous illness, such as sarcoidosis. For this reason, Wegener granulomatosis (WG) has been targeted as a potential indication for TNF blockade. Several open-label trials have shown infliximab to be beneficial in the treatment of WG. Josselin and colleagues conducted a long-term follow-up of 15 patients treated with infliximab for several vasculitides, including 10 patients with WG, 1 with microscopic polyangiitis, 3 with RA, and 1 with cryoglobulinemia. By day 45 of treatment, 11 patients had entered remission, and 4 others had some degree of therapeutic response. Despite the continuation of immunosuppression, 10 of the 15 patients ultimately relapsed. Investigators conclude that in treatment-refractory vasculitis, the TNF antagonists may serve as a potential salvage treatment. Etanercept has also been tried but results have been far less promising, pointing to a potential key to understanding the pathogenesis of this illness. The risk for using TNF blockers in these patients has largely been related to the increased rates of infection.
Bullous pemphigoid and mucous membrane pemphigoid
Bullous pemphigoid (BP) is an acute or chronic autoimmune-mediated skin disease characterized by the formation of bullae. In patients with BP, autoantibodies are formed against glycoproteins of the hemidesmosome. Mucous membrane pemphigoid (MMP), also known as cicatricial pemphigoid, shares many common features with BP, but almost always involves the mucosal membranes and is less commonly associated with skin involvement. This disorder carries the risk of serious complications, such as blindness, as a result of scarring of ocular mucous membranes.
Patients with BP have been shown to have increased TNF levels in their serum and in blister fluid, and these levels tend to correlate with severity of disease. Patients with ocular MMP were also found to have increased levels of TNF-α in their serum compared with healthy controls. For this reason, TNF antagonists have been used as a potential treatment of this illness.
There has been 1 report of a patient with concurrent BP and psoriasis treated with etanercept after initial treatment with prednisone. This technique allowed the successful tapering of the prednisone without a flare of either illness. Two cases have been reported in which 4 treatment-refractory patients with MMP were treated with 25 mg subcutaneous etanercept twice weekly in addition to their prior regime. Three patients had oral mucosal involvement and 1 had severe, recalcitrant, ocular disease. In all cases, patients achieved clinical remission. Recommendations are that this class of drugs may be a useful alternative in patients who may otherwise require, aggressive systemic treatments.
SLE
SLE is a multisystem chronic inflammatory, connective tissue disease that affects the joints, skin, lungs, kidneys, nervous system, and other organs. Immunologic abnormalities, particularly the production of antinuclear antibodies, are the prominent feature of this disease. SLE leads to tissue destruction via activation of the complement cascade and deposition of complement in tissues. TNF, an early proinflammatory cytokine, is implicated as a key player in this disease process. Thus, theoretically, medications aimed at blocking its effects should interrupt the complement cascade and decrease clinical symptoms. However, treatment of patients with SLE with TNF antagonists remains controversial. There have been numerous reported cases of increased production of autoantibodies and several cases of drug-induced lupus secondary to this class of medications. Most cases of increased autoantibody levels have been seen in clinical trials of patients with RA and Crohn disease. However, in many circumstances this potential risk may be outweighed by the beneficial effects of treatment. In a Finnish study of 53 patients treated with TNF antagonists, researchers found a response rate greater than 50% in 74% of patients treated with etanercept and 60% of patients treated with adalimumab. Only 1 patient in this study was noted to show features of drug-induced lupus erythematosus.
Increases in anti–double-stranded DNA (dsDNA) and in anticardiolipin were noted in several patients in a 2004 study on the effect of infliximab in conjunction with azathioprine or methotrexate. However, 6 patients with SLE were noted to have a decrease in symptoms of arthritis and proteinuria. The increases in autoantibody levels were not associated with an increase in disease activity in these patients. Another report described the successful management with etanercept of a patient with subacute cutaneous lupus erythematosis and RA. No increase of autoantibodies was noted in this patient. An additional report described the improvement of a patient with subacute cutaneous lupus erythematosis treated with etanercept; however, no comment was made on autoantibody levels.
Recently, Aringer and colleagues reported on 13 patients with refractory lupus nephritis treated with infliximab. They found that an induction regime of 4 infusions combined with immunosuppressive therapy led to significant improvement in symptoms and may potentially induce long-term remission. However, prolonged use of TNF antagonists may be associated with an increased risk of potentially life-threatening illness, such as Legionella pneumonia and cerebral lymphoma. Another study found significant improvement in patients who had infliximab added to standard treatment regimens compared with controls.
In 2008, Costa and colleagues performed a retrospective review of 33 patients who met criteria for drug-induced lupus erythematosis (DILE): 21 caused by infliximab, 10 caused by etanercept, and 2 caused by adalimumab. These investigators found that the patients with DILE secondary to anti-TNF agents differed from patients with classic DILE secondary to traditional agents. In classic DILE there is significantly less cutaneous involvement and the disease is not usually associated with antibodies to dsDNA. In this review, however, 72% of patients with presumed TNF blocker-induced DILE had cutaneous involvement and 90% had positive dsDNA antibodies, suggesting the possibility of a unique reaction. Further complicating this picture is the difficulty in distinguishing a DILE reaction from the underlying rheumatologic disorder.
Based on the reports mentioned earlier, it is possible that although autoantibody levels may increase during treatment with the TNF blockers, this increase does not seem to be related to severity of disease or clinical progression of symptoms, and may be a separate process altogether. Currently, 2 trials are recruiting participants to study the safety profiles of etanercept and infliximab in the treatment of patients with SLE ( http://clinicaltrials.gov ). Further studies are needed to fully evaluate the efficacy and safety of these drugs in SLE.
Scleroderma
Scleroderma is a chronic autoimmune disease characterized by the progressive fibrosis of skin and internal organs, including lungs and gastrointestinal tract. Clinically, these patients display marked variability in the extent and severity of their disease. Although the cause of scleroderma is largely unknown, it is believed to be perpetuated by inflammation following vascular alterations and fibrosis. TNF-α is believed to play a role in increased fibroblast chemotaxis and decreased collagen production. In mouse models of bleomycin-induced scleroderma, etanercept has been shown to lead to a significant reduction in dermal sclerosis, collagen accumulation, and the number of infiltrating myofibroblastic cells.
In 2000, etanercept was studied in a trial of 10 patients with systemic sclerosis. Patients were treated with 25-mg subcutaneous injections twice weekly for 6 months. Four of the 10 patients had improvement of their Rodnan skin score and 3 of 4 patients with digital ulcers reported improvement. Other measures of disease, such as pulmonary function tests, oral aperture, and hand extension, remained stable. One patient had clinical progression of his disease, but no adverse effects were noted.
More recently, in a retrospective analysis of 18 patients at the Johns Hopkins Scleroderma Center treated with etanercept for active inflammatory joint involvement, investigators found that 15 of the 18 patients experienced a significant decrease in signs of inflammation and synovitis. There was no reported worsening of skin involvement during treatment. In addition, they reported no complications, such as opportunistic infections, hospitalizations or death, related to the treatment.
A 2008 study by Denton and colleagues included 16 patients with diffuse cutaneous systemic sclerosis treated with 5 infusions of infliximab 5 mg/kg. After 26 weeks there was no statistically significant change in Rodnan skin score. Nonetheless, clinical stabilization of disease was reported, as well as a decrease in 2 laboratory markers of collagen synthesis. One-third of these patients were reported to have formed anti-infliximab antibodies that resulted in subsequent infusion reactions. A recent case report of a patient with systemic sclerosis, complicated by lung fibrosis and pulmonary hypertension, reported stabilization of lung function and improvement in quality of life during treatment with infliximab 5 mg/kg and methotrexate 10 mg/wk.
There have also been several reports on the adverse effects of TNF antagonists in the treatment of scleroderma. Drug-induced lupus has been reported in association with the use of etanercept and infliximab. Both patients had resolution of clinical symptoms and normalization of autoantibody levels following cessation of treatment. Another report discussed a patient with scleroderma who developed pancytopenia and a fungal infection following treatment with infliximab. These adverse events highlight the potential concern in using TNF blockers and emphasize the need for more research on the safety and efficacy of these drugs in the treatment of scleroderma.
Dermatomyositis
Dermatomyositis (DM) is a connective tissue disease characterized by inflammation of the muscles and skin. The cause is largely unknown. Regardless of the definite cause, multiple studies have implicated TNF as playing a significant role in the pathogenesis of DM. In addition, there have been reports of successful management of DM with etanercept and infliximab. In a study of 5 children with refractory juvenile DM, all patients showed significant clinical benefit following treatment with infliximab. An open-label trial of anti-TNF-α in combination with methotrexate in treatment-naive patients with DM and polymyositis yielded less promising results. Only 2 patients of 6 reached the primary end point at 26 weeks, with 3 dropping out of the study because of disease progression. Based on previous studies and reports, as well as their own data, investigators concluded that anti-TNF treatment is beneficial in some patients with DM and polymyositis, whether treatment naive or refractory, but that it remains difficult to predict which patients will respond to TNF blockade. Another case report of 2 patients with DM treated with infliximab reported a favorable response to treatment after 3 infusions. One patient was noted to be progressing well, whereas the other succumbed to aspiration pneumonia during the course of a fourth infusion while on concomitant high-dose prednisone treatment.
There has been 1 report of a patient with refractory DM who became septic after her third infusion with infliximab. The patient recovered with antibiotics, but 4 months later was diagnosed with non-Hodgkin lymphoma. Although it is more likely that the lymphoma is the result of the DM or of her long-standing use of immunosuppressives, given the time course, it is not possible to completely rule out infliximab as a potential contributor. For this reason, these investigators suggested using caution when prescribing TNF blockers for patients with syndromes known to have paraneoplastic associations.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree