Since its approval in 1997 by the US Food and Drug Administration, rituximab has been approved for use in certain B-cell lymphomas and treatment-resistant rheumatoid arthritis. Over the past 10 years, many published reports have suggested rituximab’s efficacy in several inflammatory conditions in dermatology. This article includes a review of the mechanism of action, dosing, side-effect profile, and the current literature for various off-label uses of this CD20+ B-cell antagonist, rituximab.
Rituximab (Rituxan, Genentech, San Francisco, CA, USA and MabThera, Roche, Basel, Switzerland) is a chimeric murine-human monoclonal antibody (IgG1 k) directed against CD20 that induces depletion of B cells in vivo. CD20 is a transmembrane protein found on pre-B–cell and mature lymphocytes. Rituximab was initially developed for treatment of lymphoma, and is currently approved for CD20+ B-cell lymphoma and rheumatoid arthritis unresponsive to other therapies.
Although the US Food and Drug Administration (FDA)-approved indications are limited, numerous off-label uses have evolved. Aside from dermatologic indications, there are reports of efficacy in rheumatology, solid organ transplantation, renal disease, neuromuscular disorders, and endocrine disorders.
Rituximab’s site of action is at the CD20 receptor. CD20 is a B-cell specific antigen expressed on the surface of B lymphocytes throughout differentiation from the pre-B–cell to the mature B-cell–stage, but not on plasma cells or stem cells. Because plasma cells and hematopoietic precursors are spared, immunoglobulin levels do not fall dramatically and B cells typically begin to return to the circulation within 6 months of therapy. Rituximab’s cytotoxicity in vitro is mediated by three mechanisms including antibody-dependent cellular cytotoxicity, complement-mediated lysis, and direct disruption of signaling pathways and triggering of apoptosis. The contribution of each mechanism in vivo remains unclear, and different mechanisms may predominate in the treatment of different diseases. Additionally, studies in low-grade lymphomas and systemic lupus erythematosus (SLE) have shown that polymorphisms of FcγRIII (receptor for IgG1 found on natural killers cells and macrophages) effect the elimination of B lymphocytes with rituximab therapy. Anolik and colleagues found that subjects with the high affinity FcγRIIIa-158V phenotype had a higher degree of B-cell depletion than those homozygous for FcγRIIIa-158F.
The initial approved dosing regimen was 4 weekly infusions of 375 mg/m 2 . This dosage was rather arbitrarily based on two phase I studies looking at single 10 to 500mg/m 2 infusions and 4 weekly infusions of 125 to 500 mg/m 2 . The results did not show a dose-dependent increase in efficacy or increase in side effects. Alternatively, two 1000 mg intravenous infusions separated by 2 weeks are used for the standard dosage in patients with rheumatoid arthritis. To date, there is no consensus on the most efficacious dosing regimen, and further studies comparing various regimens are necessary.
The incidence of serious adverse effects with rituximab is low. Infusion reactions are the most common adverse event. In most cases these are mild and occur only with the first infusion. Patients are often premedicated with acetaminophen and diphenhydramine to prevent or blunt infusion reactions. In a recent study of rituximab for the treatment of rheumatoid arthritis (RA), infections occurred in 35% of subjects in the rituximab group as compared with 28% of the placebo group. Serious infections occurred in 2% of the rituximab group as compared with 1% of the placebo group. Human anti-chimeric antibodies (HACAs) develop in less than 1% of patients treated for lymphoma, though the incidence may be higher in patients treated for autoimmune disease. In a study of rituximab for SLE, 6 of 18 subjects developed detectable HACAs; however, no subjects had adverse events related to this development. HACAs also developed in 2 of 11 subjects in a recent study of rituximab for pemphigus vulgaris. In this report, there was an association between an increase in HACA levels and increased disease activity.
There have also been three cases of rapid onset cutaneous squamous cell carcinoma and two cases of rapid onset Merkel cell carcinoma. Late onset neutropenia has been reported in patients with lymphoma, pemphigus, and vasculitis treated with rituximab.
Recent reports describe two cases of progressive multifocal leukoencephalopathy (PML) in patients with SLE and one patient with rheumatoid arthritis treated with rituximab. A review of case reports from the US FDA, the manufacturer, and a literature review from 1997 to 2008 revealed 59 cases of PML in patients who were HIV-negative and treated with rituximab. Of these, 52 had lymphoproliferative disorders, three had RA, two had SLE, and one subject each of idiopathic autoimmune pancytopenia and immune thrombocytopenia. The median time to diagnosis of PML was 5.5 months after the last rituximab dose and the median time to death after PML diagnosis was 2 months with a fatality rate of 90%. The two subjects with SLE had previously been treated with corticosteroids and alkylating agents; whereas, two of the subjects with RA had other risk factors for PML, including chemotherapy and radiation therapy, or long-standing lymphopenia. The most recent report of PML in RA (October 2009) was in a patient who had been treated with leflunomide, hydroxychloroquine, and prednisone. This report was the first of PML in a patient with RA who had not received antecedent therapy with a tumor necrosis factor-alpha antagonist.
Off-label uses of rituximab
Rituximab results in the depletion of normal and malignant B cells, leading to investigation of its use in autoimmune disorders, including SLE, RA, autoimmune thrombocytopenia and hemolytic anemia, and autoimmune neuropathies. It has shown promise in the treatment of RA and SLE. Although there have been no randomized, controlled trials of rituximab in dermatologic disease, case reports describe its use in pemphigus vulgaris; pemphigus foliaceus; paraneoplastic pemphigus; epidermolysis bullosa acquisita; bullous pemphigoid; mucous membrane pemphigoid; cutaneous B-cell lymphoma; dermatomyositis; cutaneous lupus erythematosus; graft versus host disease (GVHD); Wegener’s granulomatosis (WG); microscopic polyangiitis; cryoglobulinemic vasculitis; and Churg-Strauss syndrome.
Pemphigus Vulgaris, Pemphigus Foliaceus, and Paraneoplastic Pemphigus
Rituximab treatment of pemphigus vulgaris (PV) was first attempted based on the success in other autoimmune disorders and the hypothesis that depletion of B cells would result in a decrease in production of the disease-causing autoantibodies. The correlation of decreases in PV autoantibody levels with clinical improvement in most of the patients in which these levels were reported would seem to support this theory. This suggests that most PV autoantibodies are produced by CD20+ B-cell clones susceptible to rituximab. Alternatively, it has been suggested that although plasma cells and memory B cells may produce PV autoantibodies, plasma cells, which are CD20- and thus resistant to the effects of rituximab, may predominantly produce the less pathogenic IgG1 desmoglein 3 antibodies, whereas the memory B cells produce the IgG4 antibodies responsible for disease. This suggestion might explain the clinical improvement in the face of persistently elevated PV antibody titers observed in one patient and the improvement preceding decreased titers in a second patient. Furthermore, in patients with SLE, clinical improvement after rituximab is regularly correlated with B-cell depletion, but not to any decrease in other serologic markers or autoantibody levels. It is postulated that disruption of antibody-independent activities of B cells, including presentation of autoantigens, co-stimulation of T cells, and regulation of leukocytes and dendritic cells, are central to rituximab’s effect on the disease. Thus, the critical effects of rituximab in PV may expand beyond decreasing autoantibody production to inhibiting B-cell dependent activation of T cells.
Recent work showed decreased titers of antidesmoglein-1 and antidesmoglein-3 antibodies in response to rituximab treatment. No changes in the titers of antibodies to pneumococcal capsule polysaccharide and tetanus toxoid were noted in one study group ; and an increase in anti-varicella zoster virus-IgG and anti-Epstein-Barr virus-IgG titers were seen in a separate group of subjects with PV treated with rituximab. This finding suggests that rituximab may preferentially decrease the numbers of autoreactive (short-lived) plasma cells, while not affecting or increasing the antibody release from pathogen-specific (long-lived) plasma cells.
Over time, the B-cell population re-emerges, though these cells have the phenotype of naïve B cells (not primed B lymphocytes). The repopulation with naïve cells may explain the long-lasting effect of rituximab in many cases.
The successful use of rituximab in over 130 individual cases of treatment resistant pemphigus vulgaris and pemphigus foliaceus have been reported. The majority of cases used a protocol previously described for patients with lymphoma of 375 mg/m 2 intravenously for four weekly dosages. Ahmed and colleagues described nine subjects with PV who received two cycles of 375mg/m 2 of rituximab intravenously for three weekly dosages followed by intravenous immunoglobulin (IVIG) 2g/kg intravenously in the fourth week; then subjects received four monthly infusions of intravenous rituximab and IVIG. Because of rituximab’s slower onset of action (generally 2 to 3 weeks), it has also been combined with immunoadsorption, which has a quick onset of action. Finally, several series describe chronic infusions every 4 to 12 weeks after an induction of four weekly infusions. B-cell depletion following treatment was seen in all cases in which it was measured. In most cases, improvement was noted within the first 2 to 6 weeks. However, there are several reports of a delayed response, where improvement was not noted for 6 to 12 months.
A recent literature review of rituximab in patients with pemphigus noted in PV a 40% complete remission (clinical remission with no further therapy necessary); 37% clinical remission (clinical remission, but further immunosuppression required); and a 21% partial remission. In pemphigus foliaceus, 40% experienced a complete remission, 45% clinical remission, and 10% partial remission. The relapse rates for PV and pemphigus foliaceus were 13% and 18%, respectively.
Rituximab was well tolerated in most of the PV and PF cases reported, consistent with the observations made in lymphoma and other autoimmune disorders. However, nine serious infections were reported; including three bacterial pneumonias, a relapse of septic arthritis of the hip, four instances of sepsis, two Pneumocystis carinii pneumonias, cytomegalovirus gastritis, and pyelonephritis. These events may indicate that close surveillance of patients with pemphigus undergoing rituximab treatment is warranted until the incidence and characterization of infectious complications in this patient population is better understood.
Multiple case reports suggest rituximab’s efficacy is less impressive in paraneoplastic pemphigus (PNP). Four case reports describe significant improvement in oral and cutaneous lesions after rituximab ; however, one report describes only partial improvement and five additional reports describe less successful results, especially with regard to mucosal lesions. The mechanism of action in PNP is likely similar to that in PV. In addition, most of the treated patients experienced at least partial remission of the underlying neoplasm and this may have contributed to the observed improvement.
Epidermolysis Bullosa Acquisita
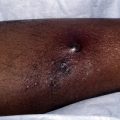
There are seven reports of patients with refractory epidermolysis bullosa acquisita (EBA), treated with rituximab. Four patients achieved complete remission, two had a partial response, and one patient died of Pseudomonas pneumonia 1 week after the patient’s first rituximab infusion. Clinical response was seen after 8 weeks to 5 months. In one patient, IgG immunoblot reactivity to type VII collagen was undetectable 12 weeks after rituximab was initiated. Complete remission was achieved 11 weeks after the first infusion and was maintained for over 1 year of follow-up allowing for tapering and discontinuation of the patient’s prednisolone and colchicine. Another patient, who had clinical remission 5 months after rituximab, subsequently developed small mechanical blisters and was treated with continued mycophenolate mofetil (MMF) and a decreased dose of prednisolone. IgG immunoblot testing to type VII collagen was 1:10 1 year after treatment (from a height of 1:3200 before beginning rituximab). Two patients were treated with a combination of immunoadsorption and rituximab; one had complete remission, the other only had a partial response. The mechanism of action of rituximab in EBA is likely similar to that in PV.
Bullous Pemphigoid
Eight cases of recalcitrant bullous pemphigoid (BP) treated with rituximab have been reported. The efficacy appears to be quite high, though use of multiple disparate dosing regimens along with only several case reports makes interpretation of efficacy difficult. Of the eight subjects, six had documented clinical remission, one had a partial response, and one died 6 weeks after starting rituximab therapy from nosocomial bacterial pneumonia. Two of those responding also had chronic lymphocytic leukemia (under complete remission). These subjects were treated with the more standard four weekly infusions of 375 mg/m 2 , but they subsequently received a rituximab dose of 375 mg/m 2 every 2 months without recurrence of bullae (3 years of follow-up reported). One of the two subjects reported by Schmidt and colleagues (the second died 6 weeks after starting rituximab) was a 2-year-old child who achieved clinical remission after rituximab therapy (24 months of follow-up reported). The subject received four weekly doses of 375mg/m 2 intravenously and had a decrease in BP 180 autoantibody levels from 3538 units/mL to 77 units/mL. A second pediatric subject had complete clinical remission after two courses of rituximab (the dose during the second course was decreased to 187.5 mg/m 2 ), though there was only 20 weeks of follow-up. Reguiai and colleagues reported a subject who had complete clinical and immunologic response after two courses of rituximab (2.5 years of follow-up reported). The one subject with bullous pemphigoid reported with a partial response only received two doses of rituximab (375 mg/m 2 ) 4 weeks apart. Notably, in the face of continued clinical disease the subject had undetectable B-cell levels within 3 weeks (persisting for at least 28 weeks) and eventual negative anti-basement membrane zone antibody titers. The mechanism of action is likely similar to that in PV.
Mucous Membrane Pemphigoid
There are three cases of successful use of rituximab in refractory mucous membrane pemphigoid. One subject received four weekly infusions of rituximab (375 mg/m 2 ) and was able to stop oral steroids within 6 months, without recurrence of disease at 12 months after rituximab therapy. Another subject received two infusions of 862.5 mg and remission was maintained at 12 months while the subject continued on prednisolone 10 mg daily and MMF 1 g twice a day (onset of improvement not discussed).
Primary Cutaneous B-cell Lymphoma
Based on its successful use in nodal B-cell lymphoma, rituximab has been employed in the treatment of primary cutaneous B-cell lymphoma (PCBCL). Approximately 60 individual cases of PCBCL treated with systemic rituximab have been reported in the literature. The cases represent a variety of histologic subtypes, including primary cutaneous follicle center lymphoma (PCFCL); primary cutaneous marginal zone lymphoma (PCMZL); primary cutaneous diffuse large B-cell lymphoma-leg type (PCLBCL-LT); diffuse large B cell lymphoma; and mantle cell lymphoma.
Intravenous and intralesional administrations have been used. Intralesional injection allows for considerably smaller doses of rituximab compared with intravenous administration, with single doses of less than 10% of the recommend dose for systemic therapy. However, case reports to date suggest an increase in recurrence rate with intralesional therapy. Additionally, one subject treated with intralesional rituximab achieved clearance of one untreated lesion, suggesting a systemic therapeutic effect of locally injected rituximab. This is further supported by the fact that local administration of rituximab also causes depletion of B lymphocytes in the peripheral blood.
Primary Cutaneous Marginal Zone Lymphoma
Five reports describe the use of intravenous rituximab in 13 subjects with PCMZL (375 mg/m 2 weekly for 4 to 8 weeks, 12/13 had multifocal disease). Overall, complete remission was seen in seven (54%), partial remission in four (31%), stable disease in one (8%), and progressive disease in one (8%). Intralesional rituximab (5–30 mg once or three times per week) was reported in 10 subjects (all subjects with less than four lesions clinically), with eight (80%) achieving complete remission and the other two subjects (20%) achieving partial remission. The relapse rates were four out of seven (57%) and five out of eight (62%) for intravenous and intralesional subjects, respectively.
Primary Cutaneous Follicle Center Lymphoma
Use of intravenous rituximab has been reported in 49 subjects with PCFCL (375 mg/m 2 weekly for 1 to 9 weeks). Overall, complete remission was seen in 40 (82%), partial remission in eight (16%), and progressive disease in one (2%). Intralesional rituximab (10–30 mg two to three times per week; though treatment duration varied from 2 weeks to 6 months) was reported in 12 subjects, with 10 (83%) achieving complete remission and two (17%) achieving partial remission. The relapse rates were 12 out of 40 (30%) and 2 out of 12 (17%) for intravenous and intralesional subjects, respectively.
Primary Cutaneous Diffuse Large B-Cell Lymphoma-Leg Type
Eight reports describe the single-agent use of intravenous rituximab in 13 subjects with PCLBCL-LT (375 mg/m 2 weekly for 4 to 8 weeks). Overall, complete remission was seen in five (38%). None of the five subjects with complete remission relapsed, though the average duration of follow-up was short at 7 months. Because of the more aggressive clinical course of PCLBCL-LT, current recommendations are for systemic chemotherapy with or without the addition of rituximab (though single agent rituximab may be a consideration in patients who cannot tolerate other therapies). A recent review of 60 cases of PCLBCL-LT noted a complete remission in 11 out of 12 (92%) subjects treated with various combinations of anthracycline-containing chemotherapies and rituximab, whereas those treated with other therapies had a complete remission of 62%. The authors suggest a short-term advantage to anthracycline-containing chemotherapies and rituximab (follow-up was short, with a mean of 19 months).
Dermatomyositis
There are two pilot studies describing the use of intravenous rituximab in 15 subjects with dermatomyositis (DM). In one study, seven subjects received four weekly infusions at a dosage of 100 mg/m 2 (three subjects) or 375 mg/m 2 (three subjects). In the second study, eight subjects received two infusions of 1 g given two weeks apart. Subjects were allowed to continue on a stable regimen of concomitant immunosuppressive medications during the trials. In the first study, all subjects had significant improvement in cutaneous and muscle disease (from 36%–113% improvement in strength scores over baseline testing). In the second study, three out of eight (38%) achieved partial remission (>50% improvement in muscle strength), although there was no change in cutaneous disease in any of the subjects. The authors postulate that the less dramatic results than the previously described open-label study may have been caused by a study population with milder disease or the different dosing regimens used.
The proposed mechanism of rituximab in DM is not well elucidated, and there are still many uncertainties regarding the role of B cells in this disease. The current model for the pathogenesis of DM holds that autoantibodies bind to an antigen on the endothelial cell wall of endomysial capillaries, leading to the activation of complement C3 and the deposition of C5b-9 membrane attack complex. This action is followed by endothelial cell swelling, necrosis, perivascular inflammation, movement of activated T cells into perimysial and endomysial spaces, and finally muscle ischemia. Immunopathologic studies also provide support for the role of microvascular injury in skin disease. Rituximab may act by decreasing B-cell production of one of the defined autoantibodies or of an as yet unidentified autoantibody to an endothelial cell autoantigen. Alternatively, it may act through dysregulation of the B cell/T cell interaction and disrupt the expansion of autoreactive T-cell populations as previously suggested in SLE.
Cutaneous Lupus Erythematosus
There have been two cases of refractory subacute cutaneous lupus erythematosus treated with rituximab. One subject responded within 8 weeks of rituximab therapy (four weekly dosages of 375 mg/m 2 ). There was disease recurrence at 11 months, at which time rituximab was reintroduced as a maintenance regimen of 375 mg/m 2 every 8 weeks for 2 years without recurrence. The second subject received two 1 g dosages 1 week apart. The subject had a partial response and remained under good control with an eventual discontinuation of oral corticosteroids. The follow-up time was short at 6 months.
Graft Versus Host Disease (Acute and Chronic)
The use of rituximab in the treatment of chronic GVHD has been reported in several case series and small clinical trials. The largest series is a phase I/II trial featuring 21 subjects with steroid-refractory disease. Cutaneous findings included sclerodermoid and lichenoid changes. Subjects were treated with one, two, or three cycles of four weekly infusions of rituximab at a dosage of 375 mg/m 2 . Subjects were allowed to continue on stable dosages of other immunosuppressive medications throughout the trial. The overall clinical response rate was 70%, including two complete responses. Cutaneous and musculoskeletal manifestations of GVHD were more amenable to treatment with rituximab than mucous membrane and hepatic manifestations. The overall response rate in all reported cases was between 43% and 80%; most responses were partial (rare cases of complete response). Currently, there is a phase I/II trial looking at rituximab as a prophylactic treatment for chronic GVHD. Experimental evidence has indicated that T cells and NK cells play a central role in the pathogenesis of chronic GVHD. Evidence for the involvement of B cells has also been accumulating. In one mouse model of chronic GVHD, an expansion of host B cells directed by CD4+ T cells is a critical step in the development of disease. Subjects with chronic GVHD develop autoantibodies similar to those seen in patients with autoimmune disease. Antibodies to Y chromosome-encoded minor histocompatibility antigens are generated after sex-mismatched transplantation and the presence of these antibodies has been correlated to the occurrence of GVHD. In the phase I/II trial discussed previously, there were four male recipients of female grafts. All four of these subjects had autoantibodies before treatment that became undetectable after rituximab, which correlated with their clinical response.
In mice, rates of acute GVHD were decreased with B-cell depletion. Kamble and colleagues reported three cases of rituximab therapy inducing complete remission in acute GVHD. The subjects received between two and four weekly dosages (dosage not reported) and response times were between 15 and 37 days. One of the subjects did go on to develop residual, limited chronic GVHD and died of sepsis 160 days after transplantation (102 days after initiating rituximab). Additionally, rituximab given as part of a conditioning regimen shortly before or after transplantation resulted in lower acute GVHD rates, whereas posttransplant rituximab did not reduce acute GVHD rates in one series of subjects receiving allogenic stem cell transplantations. The authors of a recent review suggest that the timing of rituximab administration is likely a relevant factor. The role of B cells in acute GVHD is unknown, but their implication is questioned by the efficacy of rituximab in acute GVHD and the increasing research implicating B cells in the pathogenesis of chronic GVHD.
Vasculitis
Rituximab for antineutrophil cytoplasmic antibody-associated vasculitis (AAV) (Wegener’s vasculitis and microscopic polyangiitis) has been reported in multiple case reports, case series, prospective open label trials, and recently in a retrospective multicenter study. The results of the multicenter study mirrored the cumulative results of the case reports and series. Most subjects (n = 65) received two infusions of 1 g given 2 weeks apart (n = 32) or four infusions of 375mg/m 2 given at 1-week intervals (n = 26). Overall, 49 out of 65 (75%) had complete remission, 15 out of 65 (23%) had partial remission, and 1 out of 65 (2%) had no response to rituximab. Aries and colleagues reported on eight subjects, of which six did not respond to rituximab. These subjects had prominent granulomatous disease and B-cell depletion was not associated with decreasing antineutrophil cytoplasmic autoantibody (ANCA) titres. Omdal and colleagues reported two subjects with prominent granulomatous disease who did not achieve remission. Conversely, most subjects with vasculitis-prominent disease responded well. This finding suggests rituximab may be effective for AAV vasculitis, whereas the utility for granulomas in WG remains unclear.
A primary effect of rituximab is to deplete the B-cell population. This depletion would decrease the production of ANCA, an important player in the pathogenesis of AAV. ANCA titres, though, do not correlate well with disease activity and patients may relapse without detectable ANCA levels. Therefore, the exact role of B cells (and rituximab) in AAV is indeterminate.
In addition to ANCA associated vasculitis, case reports show rituximab may also be efficacious in cryoglobulinemic vasculitis, Churg-Strauss syndrome, and Henoch-Schönlein purpura.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree