Cutaneous T-cell lymphoma is a malignancy of skin-homing T cells. This unique population of lymphocytes requires alternative therapies to those used in nodal lymphomas. Although phototherapy and nitrogen mustard have been standard treatments for decades, newer therapies have been arriving with increased frequency. Moreover, some therapies, currently used to treat other diseases, have been used with good effect. These innovative therapies are discussed, with review of current data and examples of how these therapies may be used today.
The incidence of cutaneous T-cell lymphoma (CTCL) in the United States continues to rise. Although CTCL has come to encompass a broad group of cutaneous lymphomas, mycosis fungoides (MF) and Sezary syndrome (SS) remain the most common forms of CTCL. The staging system for CTCL applies only to MF/SS. Additionally, most of the literature on therapy and the treatments discussed in this article are focused on these variants. As the number of patients with MF/SS increases, so do the therapy problems that arise in their management.
Two dichotomies must help position available treatments ( Table 1 ). One is whether the treatment is localized or generalized. The second is whether the goal of therapy is to achieve remission or to achieve palliation. The initial therapies—topical nitrogen mustard (NM) and photochemotherapy with psoralen and ultraviolet A light (PUVA)—published for the treatment of MF/SS would be considered as generalized and intended for remission. NM was first applied topically to MF in 1959. Perhaps the most widely used generalized remittive therapy, phototherapy, has been in use for treatment of the disease since the 1970s. Undoubtedly the highest response rates for remission are with radiotherapy whether generalized (total skin electron beam ) or localized. Whereas the use of NM and PUVA has remained stable, there has been an expansion in the variety of therapies in the categories of localized ablative, generalized palliative, and generalized remittive (as outlined in Table 1 ). When lesions do not therapeutically respond to the first course of conventional therapy, then the innovative new therapies need to be considered. This article discusses some innovative therapies that are being used to treat MF/SS. Although randomized controlled trials are needed to verify the preliminary results, a patient’s predicament may benefit from the promising therapies reviewed.
| Palliative | Remittive | |
|---|---|---|
| Localized | Steroid cream | Radiograph Bexarotene gel Excimer laser Imiquimod Cryotherapy Surgical excision |
| Generalized | Prednisone Chlorambucil Methotrexate Photopheresis Oral bexarotene Oral vorinostat Denileukin diftitox Romidepsin Liposomal doxorubicin Alemtuzumab | Nitrogen mustard Total skin electron beam Phototherapy Allogeneic stem cell transplantation |
Localized ablative therapies
Photodynamic Therapy
Photosensitizers in photochemotherapy have been used to treat skin disease for centuries. The use of PUVA has been a key strategy in the treatment of MF for decades. With the application of photodynamic therapy (PDT), which uses aminolevulinic acid (ALA) and its derivatives in the presence of light, a new topical photosensitizing strategy has been used for the treatment of cutaneous malignancies. Topical PDT is activated in the visible light spectrum (580–720 nm). The specificity for PDT is still under investigation but it is known that only cells within the epidermis and epidermal appendages initially become photoreactive. Interestingly, tumors of epidermal origin are also photosensitive, even when located within the dermis, suggesting that the depth of ALA absorption or light penetration are not the only factors determining which cells may be targeted by this treatment. Activated T lymphocytes, as observed in skin lymphoma, can absorb ALA, whereas unactivated T lymphocytes can not. Therefore, the use of PDT in MF has a physiologic basis. Malignant T cells localized in plaques or tumors of CTCL can be bathed in ALA and thereafter photoactivated, causing cell death through the production of reactive oxygen species and apoptosis.
The use of PDT in CTCL dates back to 1994 when Svanberg and colleagues and Wolf and colleagues, each described patients with CTCL treated with PDT. Svanberg and colleagues treated two patients, once or twice for a year or less, showing a complete response. Wolf and colleagues also reported complete remission in two patients with 4 to 5 courses of PDT over 6 months. Later Orenstein and colleagues treated one patient with tumor-stage MF who showed a complete response confirmed by biopsy with 2 years of follow-up. In 2001, a larger case series was performed that showed the first histologic confirmation of clearance of MF with PDT. Ten patients with MF, 8 with plaque stage and 2 with tumor stage, were treated. They had all failed previous therapies, including etretinate, PUVA, and radiotherapy. In lesions that showed a clinical response to PDT, there was a decrease in CD3+ CD4+ cells in the epidermis and dermis, although a few foci remained. In a patient that did not respond, a longer incubation time with the ALA (18 hours instead of 5–6 hours), did not change the clinical or histologic appearance of the lesions after two treatments. The patient who had previously received radiotherapy had some baseline fibrosis and required 11 cycles of treatment to respond.
Various forms of ALA and various light sources have been used ( Table 2 ). There have been over 30 patients reported and the results have been favorable. Because there has been no consistency in methodology, response rates cannot be obtained; however, multiple studies have shown response in both plaques and tumors of MF. Although recurrence may occur, many remissions lasted at least 1 to 2 years during follow-up. Application time varied but most studies used a range of 3 to 6 hours. Light sources have also been variable although all were within the visible light spectrum. The various light sources, doses, and dose rate used in PDT have been reviewed elsewhere, and those used in CTCL are shown in Table 2 . For lesions of CTCL that are refractory to standard therapies, PDT is an emerging therapy with an extensive history awaiting further exploration.
| Photosensitizer | Application Time (hours) | Source | Dose (J cm −2 ) | Follow-up (months) | Patients | |
|---|---|---|---|---|---|---|
| Wolf et al, 1994 | 20% dALA | 4–6 | Modified slide projector | 40 | 8–14 | 2 |
| Svanberg et al, 1994 | 20% dALA | 5–18 | Doubled pulsed frequency Nd:YAG dye laser | 60 | 6–14 | 2 |
| Orenstein et al, 2000 | 20% dALA | 16 | Broad band red light (580–720 nm) | 170–380 | 24–27 | 2 |
| Edstrom et al, 2001 | 20% dALA | 5–6 | Waldmann PDT 1200 (600–730 nm) | 50–180 | 4–21 | 10 |
| Markham et al, 2001 | 20% dALA | 4 | Waldmann PDT 1200 | 20 | 12 | 1 |
| Paech et al, 2002 | 20% dALA | 4 | Waldmann PDT 1200 | 180 | 12 | 1 |
| Leman et al, 2002 | 20% dALA | 6–24 | Xenon short arc discharge lamp | 100 | 12 | 1 |
| Coors et al, 2004 | 20% dALA | 6 | Waldmann PDT 1200 | 96–144 | 14–18 | 4 |
| Umegaki et al, 2004 | 20% dALA | 6 | SUS66 illuminator | 120 | NR | 1 |
| Zane et al, 2006 | 20% mALA | 3 | Aktilite CL128 lamp | 37.5 | 12–34 | 5 |
| Recio et al, 2008 | 20% ALA | 4 | 585 nm pulsed dye laser | 8 | 24 | 2 |
| Hegyi et al, 2008 | 20% mALA | 3 | Aktilite lamp (635 nm red light) | 100 a | 16 | 1 |
PDT may be considered in cases of oligolesional persistent disease or for patients where a radiation-sparing therapy is needed due to prior sun, phototherapy, or radiotherapy.
Cryotherapy
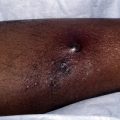
Cryosurgery is a readily available and overlooked modality in treating CTCL. The freeze-thaw cycle causes localized tissue injury that can destroy all living cells, leading to necrosis. Freezing should be fast and thawing should be slow. Most dermatologists use cryotherapy for the treatment of benign and premalignant lesions, including warts and actinic keratoses and, less commonly, malignancy. Nevertheless, there is little evidence in the medical literature to direct the physician regarding the exact procedural method. This is especially true for treating malignant T cells. There has been no study showing the optimal temperature for killing lymphocytes in human skin. Nevertheless, there is little doubt that cryotherapy can be used to destroy benign and malignant cells in the skin, including T cells.
In general most experts report that multiple freezes are better for malignant lesions than a single freeze-thaw cycle. It can be used to treat any part of the body, so is particularly useful for small lesions that have resisted previous therapies. As with other malignancies, a greater amount of necrosis is required to destroy a malignant lesion. A tissue temperature of −50° to −60°C will destroy malignant lesions. Because the depth of damage is determined by the freeze time, thaw time, and area treated, experience is needed to properly deliver the cryogen. However, malignant lymphocytes in the skin, even when within a tumor, can be readily killed with freezing. One guide is to choose a thaw time that is at least twice the freeze time. Although a lateral margin of normal skin should be treated, there is no data regarding its size. Topical and injected anesthetics can be used for treating larger lesions.
Patients with CTCL who are treated with cryosurgery have a typical tissue reaction that includes crusting and occasionally bulla formation. Weeping and sloughing of the skin are also common. Melanocytes are more sensitive to freezing than most other cell types in the skin and, therefore, patients should be warned about possible hypo- and depigmentation after treating the malignant lesion. As mentioned, although it has not been established in all cell types, destruction of malignant cells often requires colder temperatures. Therefore, the use of higher temperatures to protect melanocytes may ultimately lead to failure in adequately treating the malignancy.
Cryotherapy may be considered in cases of oligolesional persistent disease and for patients where a radiation sparing-therapy is needed due to prior sun, phototherapy, or radiotherapy.
Surgery
Surgery with steel can be an overlooked modality in treating CTCL. Although commonly used in the treatment of cutaneous B-cell lymphoma, the use of primary excision in T-cell lymphoma is infrequent. There have been reports of treating variants of MF including unilesional MF with surgical excision. Case reports have suggested that lesions do not recur. It is more natural to consider surgical excision in a patient presenting with one small lesion. However, surgical excision should also be considered as an option in patients who have failed first line therapies or have had a recurrence or nonresponse to treatment of a solitary lesion.
Surgery may be considered for patients with unilesional MF, pagetoid reticulosis, or small, solitary recurrent lesions.
Excimer Laser
The US Federal Drug Administration approval of the excimer laser for psoriasis and vitiligo has increased access to this therapeutic option. The xenon-chloride laser emits monochromatic light in the UV range at a wavelength of 308 nm. This wavelength is within the UV B spectrum (UVB) (320 nm–280 nm), which suggests that the excimer laser light would be effective in MF. There have been a number of case reports or series and open trials showing benefit. It has a safety profile similar to phototherapy because local phototoxicity is the major adverse event. However, the ability to localize treatment to lesional skin helps minimize this.
In 2004, Nistico and colleagues treated five patients with Stage IA patch-plaque MF, and 10 lesions in total, with excimer laser with one year of follow-up. All 10 lesions had a complete response without recurrence over 1 year ( Table 3 ). Five patients with patch-plaque disease were treated by Passeron and colleagues. All showed a clinical response, and four of five showed a complete response. At the end of the trial, all five had maintained the response, including the partial responder who had “minimal residual activity.” Mori and colleagues also presented a case series in four patients with Stage IA disease. Seven lesions were treated and showed complete response with follow-up between 3 and 28 months. One patient developed a new lesion in a different, untreated site during follow-up, while the other three remained disease free. In one case series of two patients, recurrence of MF after excimer laser was observed. The patient had patch-plaque disease and previously developed recurrence following a complete response with PUVA 15 months prior. Because the patient had previously experienced nausea with psoralen, excimer laser was used after disease recurrence. Several lesions showed a partial or complete response, but she continued to develop new plaques after discontinuation of therapy. Finally, two patients have been presented who had individual lesions treated with excimer laser while undergoing narrow-band (NB)UV light therapy for other lesions. One patient had a complete response to both NB-UVB and excimer laser-treated lesions. The other patient had a partial and incomplete response to her lesions with both NB-UVB and excimer laser.