- •
Adipocytolysis refers to the subcutaneous injection of chemicals, such as the detergent sodium deoxycholate, which ablates or lyses rather than shrinks fat cells
- •
Injection lipolysis, otherwise known as pharmaceutical lipoplasty, refers to the subcutaneous injection of compounds, such as the beta agonist, salmeterol xinafoate, which mobilizes triglyceride and reduces fat cell size without lysing or ablating the fat cell
- •
While there are currently no FDA-cleared injectable agents for fat reduction, two therapies with differing mechanisms of action, salmeterol xinafoate and sodium deoxycholate, have demonstrated promising outcomes in clinical trials
Introduction
Body contouring refers to optimizing the definition, smoothness, and shape of the human physique. With the substantial rising demand for body contouring over the past decade, noninvasive methods for fat reduction have become increasingly available. Historically, the approach to body contouring has largely involved invasive procedures such as liposuction and abdominoplasty. Liposuction is one of the most popular cosmetic surgical procedure in the United States, but it is invasive (surgical) with attendant downtime and rare but potential risks, including complications from anesthesia, infection, embolism, and even death.
In recent years, increasing numbers of nonobese patients have been looking for body contouring procedures with minimal downtime and with minimal risk, even if they are not as effective as liposuction and may require multiple treatments. Currently available nonsurgical body contouring devices include cryolipolysis, low level laser therapy, low energy nonthermal ultrasound, and high intensity focused ultrasound. While these devices have demonstrated safety, their efficacy across a wide range of patients is variable. Furthermore, the level of scientific rigor needed for FDA clearance of devices – small, open label, primarily safety-oriented clinical studies – does not approach the level of rigor required for a medication; that is, large, randomized, double blind, placebo-controlled studies with stringent safety and efficacy endpoints. Conducting pharmaceutical drug registration studies takes a decade or more and costs well up to $100 000 000. While this has thus far limited the number of injectable, minimally invasive therapies in the therapeutic pipeline, it ensures that those therapies that make it to market are safe and efficacious.
As of this writing (2014), two subcutaneously injected medications, salmeterol xinafoate (SX) and sodium deoxycholate, are under investigation in FDA-regulated clinical trials and show great promise.
Injection lipolysis (or pharmaceutical lipoplasty) refers to the subcutaneous injections of compounds that mobilize triglyceride and reduce fat cell size without lysing or ablating the fat cell, such as the beta agonist salmeterol xinafoate.
Background and mechanism of action
Fat cell ablation
Injectable fat-reducing techniques were first described by Brazilian dermatologist Patricia Rittes MD, when she reported reduction of infraorbital fat using direct, transcutaneous injection with Lipostabil ® (Sanofi-Aventis, Paris, France). Lipostabil is a solution consisting of soy-derived phosphatidylcholine (5%) with its solvent sodium deoxycholate (2.5%) in sterile water. In this initial report, 30 patients were injected with a total of 0.4 mL Lipostabil (50 mg/mL phosphatidylcholine (PC), 25 mg/mL deoxycholate (DC)) under each eye, and the treatments were 15 days apart. Most patients (22/30) in the study only had one or two treatments. Based on subjective data, all patients experienced reduction of infraorbital fat herniation, and the results persisted after observing them for 2 years. Rittes’ report inspired numerous clinicians to request and use compounded PC (which indeed also contained DC) as a minimally invasive fat loss procedure for trunk and extremity fat. This practice lead to relatively widespread use of treatments using pharmacy compounded PC/DC, called Lipodissolve, which was unregulated and as a result was wrought with controversy. Tighter regulation and additional scientific investigation aimed at purifying and simplifying the injectable formulation led to a diminution of PC-containing compounded medications and focus on its solvent, DC.
Originally, it was thought that PC was the active fat-reducing ingredient in Lipostabil. Furthermore, PC was hypothesized to induce a cascade of intracellular signals that lead to apoptosis, or it directly lyses fat cell membranes, emulsifies triglycerides, upregulates lipoprotein lipase, and facilitates transit of triglycerides across cell membranes. None of these theories could be proven experimentally or clinically. However, the discovery that DC, the solvent of PC, rather than PC itself, was the agent responsible for fat reduction changed the landscape of clinical investigation. Ironically, PC was found to be a bystander rather than an active ingredient in the original Lipostabil studies and Lipodissolve treatments.
Numerous studies have supported the original observation that DC was the primary agent responsible for fat cell destruction. Gupta et al. demonstrated that DC alone and PC/DC are cytotoxic to cultured adipocytes, endothelial cells, fibroblasts, and skeletal muscle cells. DC alone produced cell death and cell lysis in vitro (keratinocytes) and ex vivo (pig adipose tissue) equal to that produced by the PC/DC combination. These results were confirmed by the work of Rotunda et al. that PC was the fat-reducing agent since it was again demonstrated that DC produced nonspecific cell lysis independent of PC. Klein et al. further demonstrated that decrease in fat volume after Lipostabil injections is likely attributable to the detergent effect of DC.
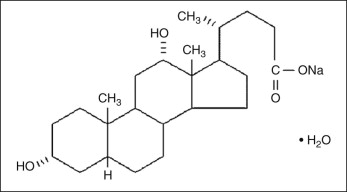
Physiologically, deoxycholic acid is a secondary bile acid produced by intestinal bacteria after the release of primary bile acids in the liver. Chemically, DC is a detergent ( Fig. 11.1 ). Detergents or surfactants are amphipathic molecules that contain both polar and nonpolar groups that are soluble in water. DC disrupts the integrity of biological membranes by introducing their polar hydroxyl groups into the cell membrane’s phospholipid bilayer hydrophobic core. Once in contact with cell membranes, its amphipathic tendencies cause it to integrate into cell membranes. The process involves an ‘attack’ of the detergent on the membrane, solubilization of membrane-associated proteins, saturation of the membrane with detergent, and then finally, with increasing detergent concentration, membrane integrity breakdown, and solubilysis. With high enough concentration, detergents fully saturate then destabilize cell membranes, which leads to solubilization and cell destruction ( Fig. 11.2 ). Any detergent-based formulation elicits adipocyte lysis, and so it will not effectively stimulate adipocyte lipolysis, which is a process that requires a fully functioning, or viable, fat cell. In order to more accurately describe injectable methods that employ detergents to diminish fat, the term ‘adipocytolysis’ can be used.
Adipocytolysis refers to the subcutaneous injection of chemicals that ablate or lyse rather than shrink fat cells, such as the detergent sodium deoxycholate.
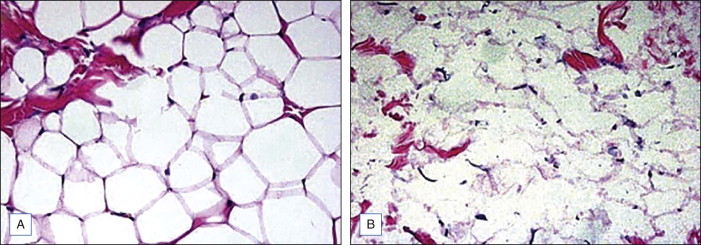
Subcutaneously injected DC leads to tissue necrosis as a result of its cytotoxic, detergent effects on the cellular membranes. In vivo and ex vivo animal or human tissue exposed to DC demonstrate fat cell lysis, fat, muscle, and collagen necrosis; erythrocyte extravasation; a mixed infiltrate consisting of polymorphonuclear leukocytes, lymphocytes, macrophages; multinucleated giant cells, and fibrosis.
As noted, in vitro, DC destroys nonadipose cells. Since it has the capacity to destroy nonadipose cells, we need to explain how it can be selective for adipose tissue ( Fig. 11.3 ). The ability of DC to lyse cells is inversely related to the amount of protein surrounding it and within tissue with which it comes into contact. The presence of albumin appears to inhibit the cell lysing activity of DC. Albumin is found in high concentrations in vital tissue but low concentrations in fat, which can explain why injections of low dose DC into fat are relatively safe clinically. This ‘safety valve’ effect of albumin on DC has fortunately made it an ideal candidate for subcutaneous fat injection.
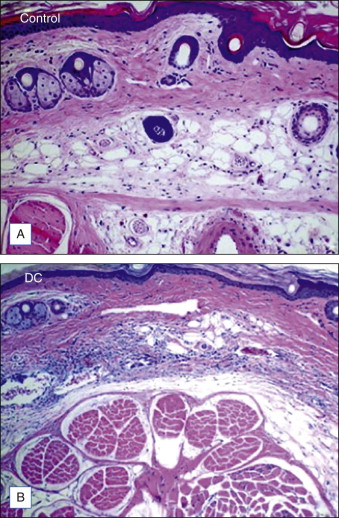
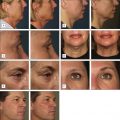
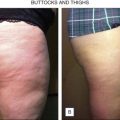
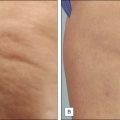
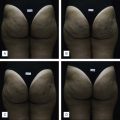
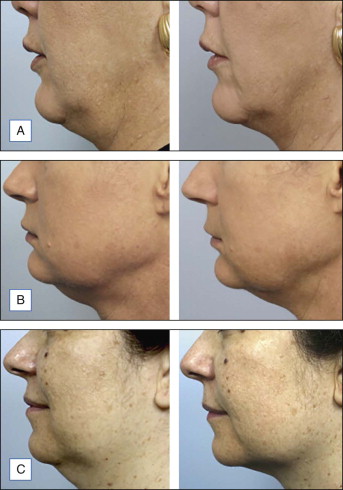
At the time of this publication, there is a synthetic, purified (nonanimal derived) sodium deoxycholate, referred to experimentally as ATX-101 (KYTHERA Biopharmaceuticals, Calabasas, CA). ATX-101 is a low-dose formulation of a purified synthetic version of deoxycholic acid that causes focal adipocytolysis, which describes the destruction of adipocytes while leaving surrounding tissue largely unaffected. The resulting expected local tissue response involves macrophages eliminating cell debris and lipids from the treatment area, and the recruitment of fibroblasts, which are believed to be responsible for neocollagenesis. ATX-101 has completed Phase III clinical trials in the US and the European Union. The first indication of the treatment is for the reduction of submental fat ( Fig. 11.4 ). Since there is no current FDA-approved injectable drug to reduce submental fat, this treatment may become a viable option for a growing, unmet need.
Fat cell lipolysis
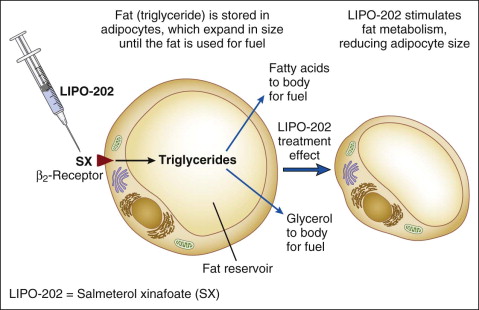
Whereas detergents work by causing focal adipocytolysis, other compounds, such as salmeterol xinafoate (SX), work by mobilizing intracellular triglyceride and reducing fat without lysing or ablating fat cells. The terms ‘injection lipolysis’ or ‘pharmaceutical lipoplasty’ refer to subcutaneous injections of compounds that mobilize and reduce fat without lysing or ablating cells. The term lipolysis is used to describe hydrolysis or breakdown of lipids into constituent fatty acid and glycerol building blocks. Lipolysis can occur within adipocytes or within the vascular space of muscle and fat tissue, and is governed by hormone sensitive lipase and lipoprotein lipase. Medications (e.g. salmeterol, isoproterenol, etc.) and neurotransmitters (e.g. epinephrine, norepinephrine) that bind to specific adrenergic receptors located on adipocyte membranes can cause lipolysis.
LIPO-102 and LIPO-202
Under investigation is an injectable form of the asthma medication ADVAIR ® (salmeterol xinafoate with fluticasone propionate), known as LIPO-102 (Lithera Inc., San Diego, CA), for localized subcutaneous fat reduction via lipolysis. SX is a highly selective long-acting β 2 -adrenergic receptor agonist, and fluticasone propionate (FP) is a synthetic trifluorinated glucocorticoid. Activation of β 2 -adrenergic receptors located on human fat cells by salmeterol triggers the breakdown of triglycerides in these cells to free fatty acids and glycerol by lipolysis ( Fig. 11.5 ). Glucocorticoids, such as fluticasone, have various effects on β-adrenergic receptor function in vivo: they enhance the coupling of β-adrenergic receptors to G proteins and the resulting activation of adenylate cyclase, and they decrease β-adrenergic receptor downregulation (tachyphylaxis) due to chronic receptor stimulation.