Abstract
The major purpose of the immune system is to provide protection against harmful agents. This is achieved by a rapid “primitive” reaction, called the innate immune response , and a more highly developed specific reaction, called the adaptive immune response . The characteristics of an adaptive immune response are specificity and the accumulation of memory, thus enabling improvement with each successive encounter with a particular antigen. The key event during an adaptive immune response is antigen presentation, yielding either a cell-mediated or a humoral response. The cellular response involves primarily T cells, whereas the humoral response involves B cells that ultimately mature into antibody-secreting plasma cells. More recently it has become clear that there is a close crosstalk between the innate and adaptive immune systems. Immune responses are not always protective and can even be pathogenic if the response induces severe tissue destruction or is directed against an autoantigen. As a barrier organ to the external environment, the skin is endowed with the capacity and the necessary cellular components to mount an immune response.
Keywords
adaptive immunity, cutaneous immunology, antigen presentation, B cells, cytokines, dendritic cells, innate immunity, Langerhans cells, T cells, Th17
- ▪
The major purpose of the immune system is protection against harmful organisms. This is achieved by a rapid “primitive” reaction, called the innate immune response, and a more highly developed specific reaction, called the adaptive immune response
- ▪
The characteristics of an adaptive immune response are specificity and the accumulation of memory, thus enabling improvement with each successive encounter with a particular antigen
- ▪
The key event in an adaptive immune response is antigen presentation, yielding either a cell-mediated or a humoral response. The cellular response involves primarily T cells, whereas the humoral response involves B cells that ultimately mature into antibody-secreting plasma cells
- ▪
Immune responses are not always protective and can even be pathogenic if the response induces severe tissue destruction or is directed against an autoantigen
- ▪
As a barrier organ to the external environment, the skin is endowed with the capacity and the necessary cellular components to mount an immune response
Introduction
The skin is in a sense a defense organ, since it represents a major barrier against the outside environment. As such, it is constantly confronted with microbial, chemical, and physical insults. Within the past four decades, there has been a greater appreciation for the fact that the skin not only functions as a mechanical barrier to the outside world, but also uses the immune system for protection. Accordingly, the skin is endowed with the capacity to generate an immune response, which gave rise to the term “skin-associated lymphoid tissues” (SALT) .
The classical immune response, also referred to as the adaptive immune response, is characterized by specificity that is due to immunologic memory (specific immunity) . Innate immunity is a more primitive defense system that acts in a rapid but less specific manner. Both types of responses can be generated in the skin. Adaptive immune responses in the skin, however, are not always protective but can also be harmful in nature, e.g. allergic or autoimmune reactions. Numerous skin diseases are caused by T lymphocytes and are therefore immunologically mediated. Consequently, many dermatoses respond favorably to immunosuppressive therapy administered either systemically or topically.
Innate Immune Response
Innate immune responses are characterized by a lack of immunologic memory. These immune reactions are less complicated than adaptive responses and developed earlier in evolution . Nevertheless, failures in these “primitive” immune responses may be associated with severe, even fatal, health problems. Essential components of the innate response are neutrophils, eosinophils, natural killer cells, mast cells, cytokines, complement, and antimicrobial peptides. The innate response is more rapid and less controlled than the adaptive immune response.
Complement
The complement system plays an important role in innate immunity. It consists of at least 20 serum glycoproteins that are activated by an enzymatic amplifying cascade (see Ch. 60 ) . Three pathways can trigger this cascade. The classical pathway is stimulated by antigen–antibody complexes, the alternative pathway by polysaccharides derived from microbial cell walls, and the later identified lectin pathway by the interaction of microbial carbohydrates with mannose-binding proteins. All three pathways lead to activation of the central C3 component and, finally, the generation of a number of immunologically active substances. For example, C3b, the cleavage product of C3, binds to the surface of microbes. Since phagocytic cells express receptors for C3b, phagocytosis of the microorganisms is enhanced. In addition, complement components bind to antigen–antibody immune complexes, which help complement receptor-bearing antigen-presenting cells to target these immune complexes.
C5a is a powerful attractant for neutrophils. C3a, C4a and C5a, also called anaphylatoxins, induce the release of inflammatory mediators from mast cells. This increases vascular permeability, thereby enabling proteins (e.g. antibodies) to enter the tissue. Assembly of the complement components C5b, C6, C7, C8 and C9 forms the membrane-attack complex (MAC), which generates pores in cell membranes, causing death by osmotic lysis. Human cells are much less susceptible to killing by complement than are microbes, since human cells express the complement receptor type 1 (CR1, CD35), decay-accelerating factor (DAF, CD55), and membrane cofactor protein (MCP, CD46), which inhibit C3 convertase and thereby block progression of the complement cascade. CD59 is a protein that binds to C8 and inhibits insertion of C9 into the cell membrane.
Toll-Like Receptors
Innate immunity serves to recognize invading microorganisms and then induce a host defense response. Several families of pattern recognition receptors (PRRs) mediate responses to pathogen-associated molecular patterns (PAMPs) that are conserved among microorganisms. Toll-like receptors (TLRs; the mammalian homologs of the Toll receptors identified in Drosophila ) are one such family of PRRs. Ten TLRs have been identified to date ( Fig. 4.1 ), with the following specificities: TLR2 (in association with either TLR1 or TLR6), recognition of lipoproteins and peptidoglycans; TLR4, lipopolysaccharide; TLR5, flagellin (a component of bacterial flagella); and TLR9, bacterial CpG DNA sequences. TLRs may also be involved in the recognition of viral components.
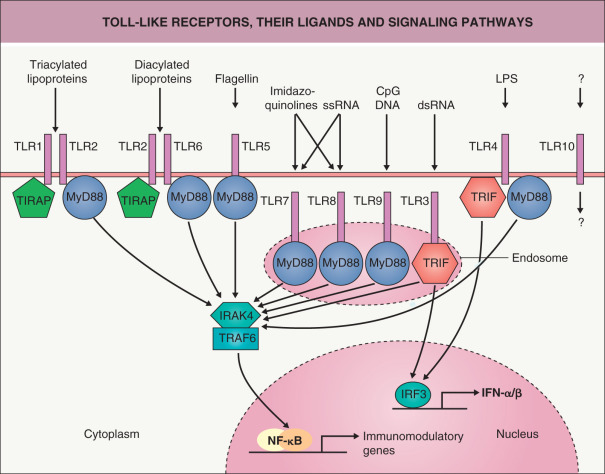
The signaling pathway of TLRs is highly homologous to that of the receptor for interleukin-1 (IL-1). Upon interaction with myeloid differentiation factor 88 (MyD88), IL-1 receptor-associated kinase (IRAK) is recruited, ultimately leading to activation of the transcription factor NF-κB (see Fig. 4.1 ). Activation of TLRs can also result in the release of interferons (IFNs) via activation of interferon regulatory factor 3 (IRF3).
Dendritic cells express several types of TLRs. Upon activation of these receptors by microbial components, the dendritic cells mature and migrate to the lymph nodes, where they present pathogen-derived antigens to naive T cells and induce an adaptive immune response. TLRs thereby bridge the gap between the innate and adaptive immune systems . These two systems constantly interact in the skin, and the innate immune system represents a potential target for modulating adaptive immune responses . Cutaneous dendritic cells that are stimulated by the innate immune system not only instruct T cells to respond but also tell them how and where. Different PAMPs and danger signals polarize dendritic cells, giving them the ability to produce certain cytokines and to induce T cells to differentiate into particular subtypes. Many of these danger signals are provided by keratinocytes, which also express PRRs. In addition, TLRs expressed in the skin direct control of pathogens by the epithelium.
Inflammasomes
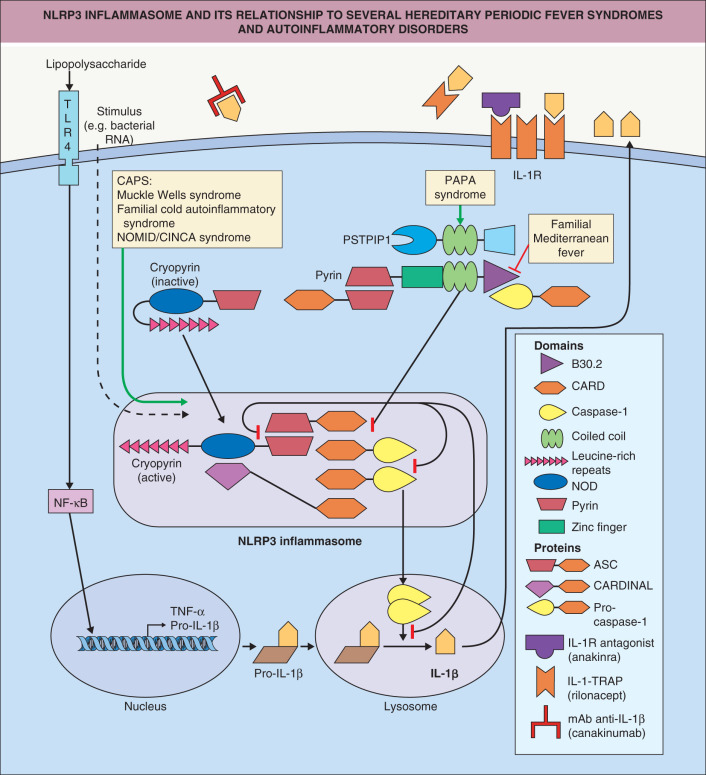
Closely related to these processes are inflammasomes, innate immune complexes which sense intracellular danger-associated molecular patterns (DAMPs) or PAMPs. Four types of inflammasomes have been identified: Aim2 ( a bsent i n m elanoma 2 ), the p yrin domain-containing NLRP1 ( n ucleotide-binding domain l eucine-rich r epeat-containing receptor 1 , also called NALP), NLRP3, and NLRC4 ( N od- l ike r eceptor C ARD domain-containing 4) . Aim2 is activated by double-stranded DNA resulting from either viral or bacterial intracellular pathogens, NLRP1 by muramyl dipeptide, and NLRC4 by flagellin. NLRP3 is triggered by numerous PAMPs and DAMPs and is thus the most important inflammasome ( Fig. 4.2 ). The ultimate result of activation of inflammasomes is the cleavage of pro-IL-1β into active IL-1β, a highly potent inflammatory mediator. NLRPs are large backbone proteins of the complex which, upon assembly of the inflammasome complex, bind to ASC ( a poptosis-associated s peck-like protein containing a c aspase recruitment domain [CARD]). The latter interacts with caspase-1, resulting in its activation. Active caspase-1 cleaves and activates pro-IL-1β, as well as the proinflammatory cytokine pro-IL-18. Dysregulation of inflammasomes is the cause of a group of inherited autoinflammatory diseases called cryopyrin-associated periodic syndromes (CAPS) which are associated with recurrent episodes of fever, urticarial skin lesions, arthritis, and systemic inflammation (see Ch. 45 ). These disorders respond very well to IL-1 blocking drugs (see Fig. 4.2 ).
Antimicrobial Peptides
To cope with an environment that is full of microorganisms, plants and invertebrates produce a variety of highly effective antimicrobial proteins. Human epithelia, including the epidermis, secrete such antimicrobial peptides as a mechanism of innate defense. The first antimicrobial peptide to be found in human skin (specifically from psoriatic scales) was human β-defensin-2 (hBD-2) . A number of other antimicrobial peptides have subsequently been isolated ( Table 4.1 ). In addition to antibacterial properties, some of these peptides possess antimycotic and likely antiviral activities. As demonstrated for psoriasin, which prevents Escherichia coli infection , these peptides may protect the skin from bacterial infections. Expression of antimicrobial peptides can be induced by bacteria, bacterial products, or proinflammatory cytokines via TLRs and other mechanisms. Enhanced and reduced production of these peptides in psoriasis and atopic dermatitis, respectively, may explain why superinfections are so rare in the former disease and common in the latter . However, others have found enhanced expression of antimicrobial peptides in atopic skin, which may reflect disruption of the epidermal barrier . Ultraviolet (UV) B radiation has been shown to induce expression of antimicrobial peptides, potentially explaining the lack of UVB-related bacterial infections despite its immunosuppressive effects (see Ch. 86 ) .
| SKIN-DERIVED ANTIMICROBIAL PEPTIDES | ||||||
|---|---|---|---|---|---|---|
| Name | Size (kD) | Cellular source | Antimicrobial activity | Inducibility (e.g. by bacteria, cytokines) | ||
| Bacteria | Fungi | |||||
| Gram+ | Gram− | |||||
| Antileukoprotease (ALP) | 11.7 | Keratinocytes Airway epithelia | ++ | ++ | ++ | − |
| Dermcidin (DCD)-1 | 4.7 | Sweat glands | +++ | +++ | ++ | − |
| Human β-defensin (HBD)-2 | 4.3 | Keratinocytes Airway epithelia Intestinal tract | (+) * | +++ | ++ | + |
| HBD-3 | 5.2 | Keratinocytes Airway epithelia | +++ | +++ | +++ | + |
| HBD-4 | 6.0 | Keratinocytes Airway epithelia (mRNA) | ++ | ++ | + | + |
| LL-37/cathelicidin antimicrobial peptide (CAMP)/human cationic antimicrobial peptide 18 (hCAP18) | 4.5 | Keratinocytes Airway epithelia Urogenital tract Granulocytes | ++ | ++ | ++ | + |
| Lysozyme | 14.7 | Keratinocytes Airway epithelia | ++ | ++ | − | − |
| Psoriasin | 11.4 | Keratinocytes Sebocytes | (+) * | ++ † | (+) * | + |
| RNase 7 | 14.5 | Keratinocytes Airway epithelia | +++ | +++ | +++ | + |
Beta-defensins can also attract immature dendritic cells and memory T cells via the chemokine receptor (CCR)-6, illustrating another link between innate epithelial defense and adaptive immunity . The antimicrobial peptide LL-37 (also known as cathelicidin antimicrobial peptide [CAMP]) mediates dendritic cell activation in psoriasis by binding self-DNA and forming structures that stimulate TLR9 and thereby induce IFN production .
Cytokines
Cytokines are a large, heterogeneous family of low-molecular-weight messenger substances that play a crucial role in intercellular communication. Cytokines can be secreted by almost any cell type, and they may act in an autocrine, paracrine, or endocrine manner. Cytokines exert their biologic activities by binding to specific cell surface receptors . Although the vast majority of cytokines occur in a soluble form, some can be membrane-bound, making the differentiation between cytokine and receptor difficult. Cytokines influence the proliferation, differentiation, and activation of cells. Each cytokine exhibits multiple activities, a fact that complicates strict categorization.
Cytokines that are produced by leukocytes and exert effects preferentially on other white blood cells are called interleukins (IL). Colony-stimulating factors (CSFs) are mediators that induce differentiation and proliferation of hematopoietic progenitor cells, while IFNs interfere with viral replication. Cytokines that have chemoattractant activity are termed chemokines, and they play a crucial role in leukocyte migration. The main subgroups of chemokines are differentiated according to the position of two cysteine (C) residues compared with the other amino acid residues (X), CXC (α-chemokines) and CC (β-chemokines) . Chemokines that recruit leukocytes are termed inflammatory chemokines, whereas those that regulate trafficking within lymphoid tissues are called lymphoid chemokines.
Early innate immune responses are dominated by cytokines with inflammatory (e.g. IL-1, IL-6, IL-18, tumor necrosis factor-α [TNF-α], inflammatory chemokines) and antiviral (e.g. IFN-α, IFN-β) capacities. Induction of adaptive immune responses is critically dependent on cytokines with immunomodulatory capacities (e.g. IL-2, IL-4, IL-12, IL-13, IL-17, IL-22, IL-23, IFN-γ). However, since most of these mediators exhibit multiple and sometimes overlapping activities, a strict separation into inflammatory and immunomodulatory cytokines is not possible. Due to structural similarities, some cytokines are grouped into families, e.g. the IL-6 family (IL-6, IL-11, oncostatin M, leukemia inhibitory factor [LIF]), the IL-10 family (IL-10, IL-19, IL-20, IL-22, IL-24, IL-26), and the IL-12 family (IL-12, IL-23, IL-27).
Macrophages and Neutrophils
Macrophages, phagocytic cells derived from blood-borne monocytes, carry receptors for carbohydrates that are usually not expressed on vertebrate cells (e.g. mannose). Through this mechanism, macrophages can discriminate between “foreign” and “self” molecules. Furthermore, macrophages possess receptors for antibodies and complement. Hence, microorganisms that are coated with antibodies and/or complement are more readily phagocytosed . After phagocytosis, the microorganisms are exposed to a variety of toxic intracellular molecules, including superoxide anions, hydroxyl radicals, hypochlorous acid, nitric oxide, lysozyme, and antimicrobial cationic proteins. Macrophages can also present processed antigens to T and B cells. However, their T-cell stimulatory capacity is less effective than that of dendritic cells.
Activated macrophages release granulocyte colony-stimulating factor (G-CSF) and granulocyte–macrophage colony-stimulating factor (GM-CSF). These two cytokines induce the division of myeloid precursors in the bone marrow, releasing millions of neutrophils into the circulation. Under normal conditions, neutrophils circulate in the bloodstream, some rolling along the vascular endothelium . To enter the site of an infection, neutrophils utilize a complex process that involves proinflammatory mediators, adhesion molecules, chemoattractants, and chemokines (see Ch. 26 ). The recruited neutrophils phagocytose organisms and kill them within phagolysosomes by using oxygen-dependent and oxygen-independent mechanisms. The former, called the respiratory burst, involves the production of hydrogen peroxide, hydroxyl radicals, and singlet oxygen. The oxygen-independent method utilizes highly toxic cationic proteins and enzymes, such as myeloperoxidase (MPO) and lysozyme. Organisms that are coated with antibodies or complement components bind to Fc and complement receptors, respectively, on neutrophils (as well as macrophages) and are more effectively phagocytosed and killed.
Ectosomes are vesicles released from the plasma membrane of neutrophils upon their stimulation (e.g. by bacteria) . They carry membrane surface receptors (e.g. CD15, L-selectin), phosphatidylserine, and granule proteins (e.g. CD66b, CD87, MPO, elastase, proteinase 3, defensins, lactoferrin, collagenase I). Depending on the composition, ectosomes can induce a variety of responses in their target cells which include endothelial cells, platelets, natural killer cells, and dendritic cells.
In addition, neutrophils can release neutrophilic extracellular traps (NETs) and thereby develop into anuclear cytoplasts . NETs are composed of extracellular strands of DNA bound to neutrophil-derived antimicrobial peptides and proteins. Because NETs can entrap bacteria, fungi and viruses, they may offer some antimicrobial protection. On the other hand, NETs can induce autoimmunity by stimulating immune responses against the NET-associated nuclear antigens (see Fig. 41.1 ). Whether formation of NETs contributes to the exacerbation of autoimmune diseases by infections remains to be determined.
Eosinophils
The major function of eosinophils is most likely to protect the host from infections by parasites, particularly nematodes. Infections with these organisms are associated with the production of antigen-specific IgE antibodies that coat the parasite. Via their low-affinity receptors (FcεRII, CD23), eosinophils bind to IgE antibodies and become activated. In contrast to macrophages and neutrophils, eosinophils are only weakly phagocytic. They harbor large granules that contain major basic protein, eosinophilic cationic protein, eosinophil peroxidase, and eosinophil-derived neurotoxin (see Ch. 25 ). Upon activation, eosinophils release these toxic products, which can kill parasites, together with prostaglandins, leukotrienes, and various cytokines . Eosinophils also play an important role in the pathogenesis of allergic reactions.
Basophils and Mast Cells
Basophils (found in the blood) and mast cells (located within tissues) exhibit similar functional and morphologic characteristics . At least two populations of mast cells exist, which can be differentiated by the enzymes they contain and by their tissue location. Mucosal mast cells contain only trypsin, while connective tissue mast cells contain both trypsin and chymotrypsin (see Ch. 118 ). In contrast to pulmonary, uterine and tonsillar mast cells, cutaneous mast cells express the receptor for C5a (CD88), which implies that triggering of mast cells through the anaphylatoxin C5a results in cutaneous, but not systemic, reactions . Basophils and mast cells express high-affinity receptors for IgE (FcεRI) that avidly bind IgE (see Ch. 18 ).
When a specific antigen binds to mast cell-bound IgE, the FcεRI becomes activated, which leads to degranulation and release of preformed mediators, including histamine and serotonin. Other mediators such as prostaglandins, leukotrienes (B 4 , C 4 , D 4 and E 4 ), and platelet-activating factor are also released, and they enhance vascular permeability, bronchoconstriction, and induction of an inflammatory response (see Ch. 18 ). Hence, basophils and mast cells play an important role in immediate-type allergic reactions such as urticaria and angioedema. There is also evidence that mast cells are involved in contact hypersensitivity reactions.
Natural Killer Cells
The major task of natural killer (NK) cells is to eliminate virally infected or malignant cells . NK cells can recognize their targets in two ways. Since they express Fc receptors that bind IgG (FcγRIII, CD16), NK cells can adhere to and kill target cells that are coated with IgG. This killing process is referred to as antibody-dependent cellular cytotoxicity (ADCC).
The second recognition system involves killer-activating and killer-inhibitory receptors. Killer-activating receptors recognize molecules that are expressed by nucleated cells. This provides a signal for the NK cell to kill the target cell by secretion of perforins, which make holes in the cell membrane through which granzymes are injected. Granzymes lyse target cells by activating the apoptotic caspase cascade. In addition, NK cells carry inhibitory receptors (KIR) on their surface that recognize major histocompatibility complex (MHC) class I self-molecules. KIRs shut off the killer signal and thus prevent autolysis of the host. Tumor cells and viruses often down-regulate MHC class I molecules to escape recognition by cytotoxic T cells. Paradoxically, this mechanism renders “MHC class I-low” cells susceptible to recognition by NK cells.
Adaptive Immune Response
The characteristic features of an adaptive immune response are its specificity and enhancement with each successive antigen encounter owing to the accumulation of “memory” . A crucial event during the generation of an adaptive immune response is antigen presentation.
Antigen-Presenting Cells
Various cells can present antigens, depending on how and where the antigen first encounters cells of the immune system. Interdigitating dendritic cells (DCs) located in the T-cell areas of the spleen and lymph nodes are the most effective antigen-presenting cells (APCs). Within the epidermis, Langerhans cells (LCs) are key APCs. Therefore, the following section will focus primarily on LCs.
Langerhans cells
In 1868, Paul Langerhans first described Langerhans cells as DCs located within the epidermis . Because of their dendritic shape, Langerhans thought that these cells might be of neural origin. More than 100 years later, it was shown that LCs are derived from bone marrow . However, in the 1990s it was observed that LCs are intimately associated with nerve fibers and that nerves, through the release of neuropeptides such as calcitonin gene-related peptide (CGRP), can modulate LC function .
Morphology of Langerhans cells
LCs cannot be identified in routinely fixed and stained histologic sections; their recognition requires electron microscopy or histochemical analysis. Ultrastructurally, LCs have rod-shaped organelles termed Birbeck granules (see Fig. 91.5 ). A Ca 2+ -dependent lectin with mannose-binding specificity called langerin is associated with and responsible for the formation of Birbeck granules . Birbeck granules are thought to result from the antigen-capture function of langerin, which routes antigen into these organelles and provides access to a non-classical antigen-processing pathway. However, langerin is not completely specific for LCs and can also be expressed by a certain type of dermal DC .
Activated LCs elongate their dendrites which can penetrate keratinocyte tight junctions and survey the area just beneath the stratum corneum for antigens . These penetrating dendrites take up antigens which co-localize with langerin/Birbeck granules. By forming new tight junctions, keratinocytes rapidly close the defects produced by the penetrating dendrites, thereby maintaining skin integrity during antigen uptake.
Histochemically, human LCs can be visualized by staining for adenosine triphosphatase (ATPase), a membrane-bound, formalin-resistant, sulfhydryl-dependent enzyme. In addition to langerin (CD207), antigenic moieties present on human LCs include the panhematopoietic marker CD45, MHC class II antigens (HLA-DR), CD1a, the S100 protein, and vimentin. CD1a is a useful marker for LCs, since within the epidermis (normal or inflamed) it is exclusively expressed on LCs, whereas HLA-DR antigens are expressed on keratinocytes in inflamed skin ( Table 4.2 ). Since CD1a does not exist in the murine system, staining for MHC class II antigens is often used for detection of murine LCs in non-perturbed skin ( Fig. 4.3 ). In addition, human LCs express the high-affinity IgE receptor (FcεRI) , which was initially thought to be exclusively expressed on mast cells and basophils.
| PHENOTYPIC MARKERS OF RESIDENT VERSUS MIGRATING HUMAN LANGERHANS CELLS VERSUS HUMAN LANGERIN − DERMAL DENDRITIC CELLS | |||
|---|---|---|---|
| Resident LC | Migrating LC | Langerin − dermal DC | |
| Birbeck granules | ++ | ++ | − |
| Langerin (CD207) | +++ | ++ | − |
| MHC class II | ++ | +++ | ++ |
| CD45 | + | + | + |
| CD1a | +++ | +++ | ++ |
| CD11c | + | + | + |
| CD11b | +/− | +/− | ++ |
| E-cadherin | ++ | + | − |
| Epithelial cell adhesion molecule (EpCAM) | + | + | − |
| CCR6 | + | − | − |
| CCR7 | − | + | + |
The density of murine LCs is dependent on a variety of factors, including strain, age, sex, and anatomic location. LCs are almost absent in the tail region, the pouch, and the cornea. In humans, the numbers of LCs are reduced on the palms and soles, genitalia, and buccal mucosa. In addition, the density of LCs decreases with age and is reduced in chronically UV-exposed skin .
Ontogeny of Langerhans cells
Bone marrow chimera experiments showed that LCs are derived and constantly replenished from the bone marrow . However, according to a more recent hypothesis , murine LCs are derived from radioresistant hematopoietic precursor cells that reside in the skin during embryonic development ( Fig. 4.4 ). The formation of LCs depends on transforming growth factor-β1 (TGF-β1) and macrophage colony-stimulating factor receptor (M-CSFR) ligands (M-CSF, IL-34), which are provided in an autocrine fashion. LCs that are depleted during the steady state or after minor injuries are repopulated locally, independent of circulating precursor cells. In lethally irradiated mice reconstituted with congenic bone marrow cells, half of the LCs are eliminated in the first week after transplantation, but LCs repopulate locally within 3 weeks. After UVB irradiation, which does not affect the dermis or the hair follicle, LCs appear to repopulate from the hair follicle . By producing different chemokines, distinct subpopulations of hair follicle keratinocytes can promote (via release of CCL2 and CCL20) or inhibit (via release of CCL8) repopulation of LCs .
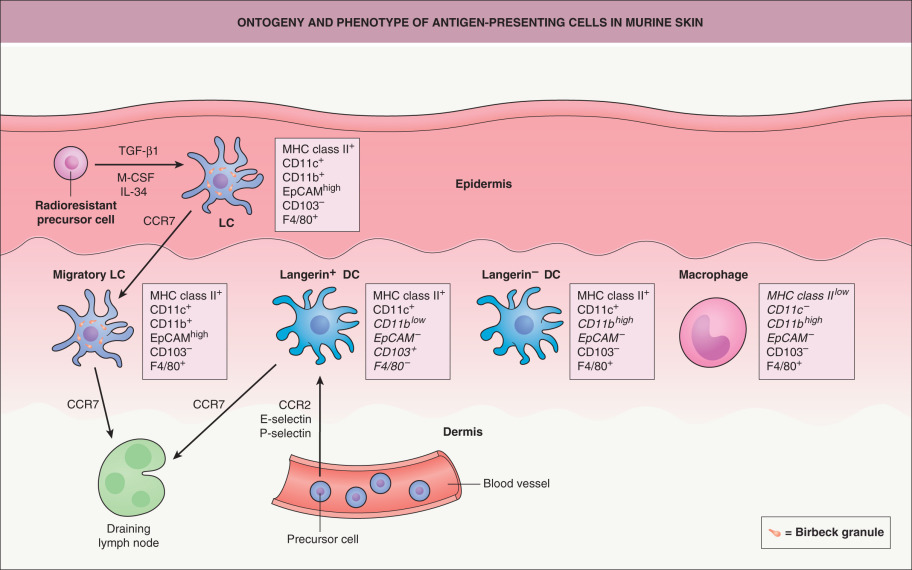
As noted above, langerin is not an exclusive marker for epidermal LCs but can also be expressed by dermal DCs (see Fig. 4.4 ) . Langerin + dermal DCs differentiate from radiosensitive circulating precursor cells, independent of TGF-β1 and M-CSFR ligands but dependent on CC-chemokine receptor 2 (CCR2), E-selectin, and P-selectin. LCs that emigrate from the epidermis to the lymph nodes in a CCR7-dependent manner can also be detected in the dermis but can be distinguished from langerin + dermal DCs by the differential expression of CD11b, epithelial-cell adhesion molecule (EpCAM), and CD103 (see Fig. 4.4 ). Two additional types of APCs can be found in the dermis, langerin − dermal DCs and dermal macrophages, and each has a particular surface marker expression pattern.
In humans, the situation is less clear than in mice, since only studies with tissue explants are available. Subtle differences in the surface marker expression of resident LCs, migrating LCs, and langerin − dermal DCs have been described (see Table 4.2 ).
Other dendritic cells
DCs are defined as professional APCs that display an extraordinary capacity to stimulate naive T cells and initiate a primary immune response . This capacity was initially described for the interdigitating DCs in the spleen. It has become apparent that the DC system is very complex, and many questions still remain unanswered.
This complexity is based on the fact that DCs can arise from several types of progenitor cells, and a variety of functional phenotypes of DCs can also be generated from the same precursor cell . In addition, differences exist between experimental systems used to study human and murine DCs. In the murine system, DCs are primarily obtained from the bone marrow or spleen, while human DCs are almost exclusively generated from peripheral blood. Furthermore, many markers/antibodies exist in the human but not the murine system, and vice versa. Whether each of the different types of DCs has a distinct immune function is still a matter of debate .
Concerning DC function, there is evidence in the murine system that spleen-derived CD8α + DCs induce T helper 1 (Th1) responses, while CD8α − DCs favor T helper 2 (Th2) responses . In contrast, in the human system, lymphoid/plasmacytoid DCs were found to induce Th2 responses, and myeloid DCs generated Th1 reactions . Consequently, myeloid DCs were referred to as DC1 and lymphoid/plasmacytoid DCs as DC2. Activation of TLR7 via the immunomodulator imiquimod, an imidazoquinoline (see Fig. 4.1 ), drives myeloid DCs to express perforin and granzyme B and plasmacytoid DCs to express TNF-related apoptosis-inducing ligand (TRAIL). This enables killing of tumor cells, indicating that both myeloid and plasmacytoid DCs are directly involved in imiquimod-induced destruction of skin cancer .
However, the type of immune response that is generated also critically depends on the state of maturation of the stimulating DC at the time of antigen presentation. For the induction of Th1 responses, the presence of IL-12 is crucial. DCs tend to produce IL-12 directly after an activating step in their maturation, driving T cells into a Th1 phenotype if they meet the DCs at this stage . At later time points, the production of IL-12 decreases, thereby favoring the development of a Th2 response. This process is also heavily influenced by the innate immune system (e.g. stimulation by TLRs; see above).
Taken together, the parameters that are responsible for the type of Th cell differentiation that is induced by DCs remain to be fully defined. On the other hand, there is agreement that immature DCs induce tolerance due to incomplete T-cell activation, resulting in T cells of the regulatory type that suppress immune responses .
Antigen presentation
Antigen presentation cells: activation and migration
To initiate sensitization, antigens must be presented to T cells by APCs. For many years, LCs were thought to be the most important APC in the skin since contact sensitization could not be induced in sites that were naturally devoid of LCs (e.g. murine tail skin) or in which LCs had been depleted (e.g. by UV radiation) . However, transgenic mice in which LCs are completely depleted via the diphtheria toxin receptor technique (i.e. short-term, inducible ablation) demonstrated variably diminished but not completely abrogated sensitization responses ; in one such model, the sensitization response was completely normal . In a different knockout mouse model characterized by constitutive and durable absence of epidermal LCs , an enhanced sensitization response was actually observed, suggesting that LCs may have regulatory functions. This “LC paradigm” proposes that LCs may be tolerogenic when they present antigens under steady-state non-inflammatory conditions, but sensitizing upon stimulation by inflammatory mediators. Which of these activities is the main function of LCs remains to be determined, and there is accumulating evidence that dermal DCs are equally if not even more important than LCs in presenting antigens.
Cutaneous APCs actively take up antigens in the skin, but antigen presentation to lymphocytes takes place in the regional lymph nodes. In the presence of inflammation, the APCs become activated and leave the skin, migrating to the draining lymph nodes. During emigration, they change their phenotypic and functional behavior and develop into mature DCs. For example, molecules involved in antigen uptake and processing (Birbeck granules, Fc receptors) are down-regulated on activated LCs . In addition, expression of E-cadherin, which mediates the attachment of LCs to neighboring keratinocytes, is reduced, thereby enabling the emigration of LCs.
CD44, a hyaluronic acid receptor involved in the tissue homing of leukocytes, is upregulated on activated LCs. The splice variant CD44v6 supports binding of LCs to T-cell-rich areas of lymph nodes . Furthermore, the integrins α 6 β 1 and α 6 β 4 , which exhibit affinity to the basement membrane zone, are induced on the surface of emigrating LCs . The release of proteolytic enzymes like matrix metalloproteinase-9 (type IV collagenase) may enable their penetration through the basement membrane. Their dendricity becomes more pronounced, and surface molecules necessary for antigen presentation and T-cell priming are upregulated (e.g. MHC class I, MHC class II, CD40, CD54, CD58, CD80, CD86). At this stage, LCs emigrating from the epidermis are almost indistinguishable from DCs obtained from lymphoid organs . The same phenotypic and functional changes may occur in other APCs of the skin that have important roles in antigen presentation.
Antigen presentation to T cells
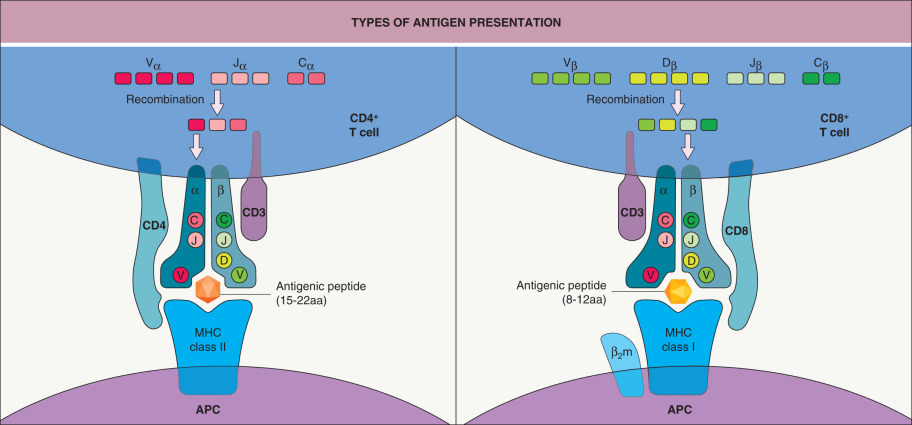
In contrast to B cells, T cells cannot recognize soluble protein antigens per se; instead, the T-cell receptor (TCR) recognizes antigen-derived peptides bound to MHC locus-encoded molecules expressed on APCs. CD4 + T cells recognize antigens in association with MHC class II molecules, while CD8 + T cells, the majority of which become cytotoxic, recognize antigens in association with MHC class I molecules ( Fig. 4.5 ).
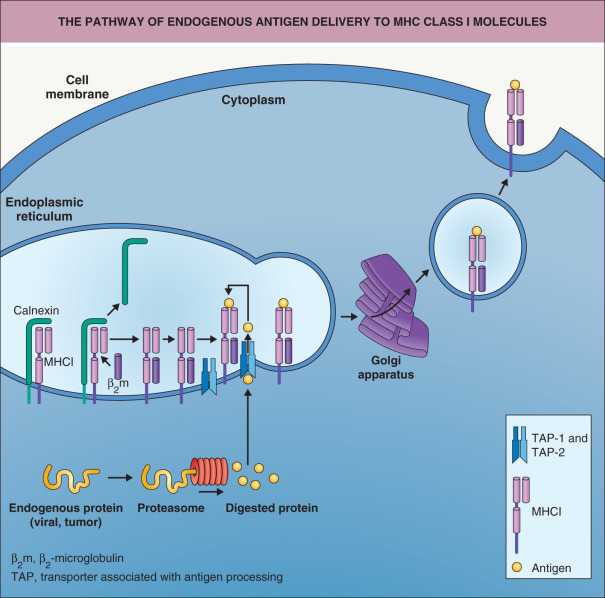
Two pathways exist by which antigens can be “loaded” onto MHC molecules. If the antigen has been produced endogenously within the cell (e.g. viral or tumor proteins), it is complexed with MHC class I molecules through intracellular processing pathways ( Fig. 4.6 ) . The proteasome degrades cytosolic antigens produced by the cell. The resulting peptides (which consist of 8 to 12 amino acid residues) are imported into the endoplasmic reticulum (ER) in a TAP (transporter associated with antigen processing)-dependent manner and loaded onto MHC class I molecules . After binding to the MHC class I–β 2 microglobulin complex, these peptides are transported to the cell surface via the Golgi apparatus. Alternative pathways by which exogenous proteins are phagocytosed and the phagosome fuses to the ER may also exist. The proteins would subsequently be retransported out of the ER into the cytoplasm (by an as yet unclear mechanism) and then degraded by the proteasome. The degraded peptides could then enter the pathway that is normally employed for endogenous proteins via the TAP protein. As the vast majority of nucleated cells express MHC class I molecules, many cell types can serve as APCs for MHC class I-restricted antigen presentation in a secondary immune response.