Hypertrophic Scars and Keloids
Jill S. Waibel
Ashley Rudnick
BACKGROUND
After injury a dynamic process of tissue regeneration occurs, which consists of 3 wound healing phases, including inflammatory phase, proliferative phase, and remodeling phase.1 All phases contribute to the ultimate healing of the scar; however, the final remodeling stage, which begins 3 weeks after the initial injury, is responsible for differences in scar outcomes. In the normal process of wound healing, the inflammation, granulation formation, and extracellular matrix formation balance is maintained by controlling the fibroblast activity.2 Continuous collagen production and degradation occurs after an injury. Abnormal scars such as keloid scars develop as a result of an imbalance of collagen production versus degradation in the months following an injury. If control of the fibroblast activity is lost, the result is either a hypertrophic or keloid scar. The first reported keloid was in 1700 CE Egypt in the Smith Papyrus. Dermatologist Baron Jean-Louis Alibert coined the term “cheloide” (Greek chele) in the 19th century to describe the “crab claw”-like appearance of keloids.3,4 Only humans are thought affected by keloids, and both dominant and recessive modes of inheritance have been described. Keloid scarring is a fibroproliferative disease that affects humans after injury and an abnormal wound healing process.
PRESENTATION
Keloids are thick growths of tissue that extend beyond the boundaries of the initial injury following scar wound healing. Hypertrophic scars present as red, itchy, raised areas overlying the boundaries of the initial wound.
DIAGNOSIS
Clinical Diagnosis
On clinical examination, keloids are firm, often tender, nodules with glossy surface epithelium and no
telangiectasia. The epithelium of a keloid is thin, and often there are focal areas of ulceration. Keloids are pink to purple in color and often have hyperpigmentation.
telangiectasia. The epithelium of a keloid is thin, and often there are focal areas of ulceration. Keloids are pink to purple in color and often have hyperpigmentation.
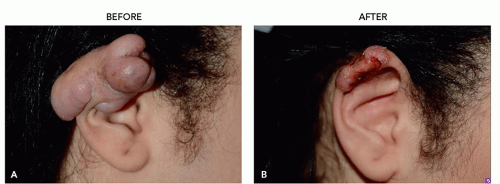
Keloids usually start as erythematous (pink) and hyperpigmented (brown) in color, and as they mature, they often become pale. The most common anatomical locations of keloids are in decreasing order earlobes, shoulders, and presternal skin. One unique aspect of these locations is these areas have no hair follicles or other glands. Keloids may cause symptoms of pruritus, pain, and hyperesthesia.5
Keloids and hypertrophic scars are often disfiguring. However, some keloids or hypertrophic scars can cause contractures, which may result in loss of function if overlying a joint, or significant disfigurement if located on the face.
Most keloids continue to grow for weeks to months and others grow for years. Growth is usually slow, but keloids occasionally enlarge rapidly, tripling in size within months. Once they stop growing, pruritus generally decreases and they may remain stable or involute slightly.
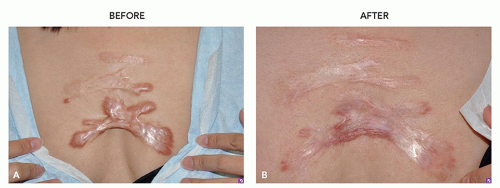
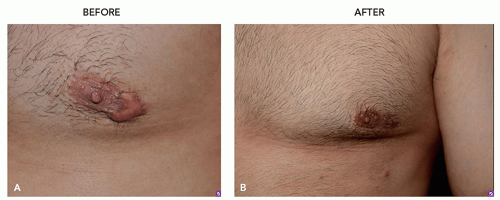
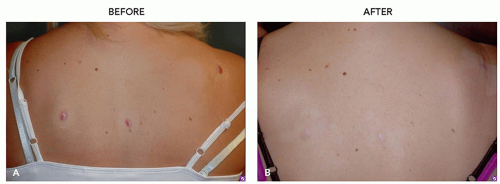
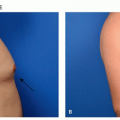
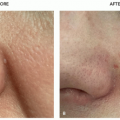
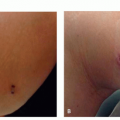
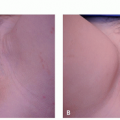
There are many shapes and forms of keloids. Keloids on the ears, neck, and abdomen tend to be pedunculated. Keloids on the extremities and chest are generally raised with a flat surface and a wide base. Most keloids are round, oval, or oblong with regular margins; some are clawlike with irregular borders (Figures 8.2.1, 8.2.2, 8.2.3, 8.2.4).
Most patients present with 1 or 2 keloids; however, a few patients, especially patients with spontaneous keloids, have multiple lesions, as do patients who develop keloids as a consequence of acne or chicken pox.
Age
Keloids may occur in all age groups, although they are rarely found in newborns or elderly persons. The highest incidence occurs in individuals 10 to 30 years of age.6 In the past 20 years there has been an increase in the number of presternal keloids, which resulted from coronary artery bypass operations and other similar procedures now undertaken by people in older age groups.
Race
The prevalence of keloids is 10% to 16% in the African American population.7 Keloids form more frequently in Chinese and Polynesian populations and exceed that observed in Indian and Malaysian persons. Caucasians are less commonly affected.
Sex
The prevalence has been reported to be slightly higher in young females than in young males. Keloids and hypertrophic scars affect both sexes equally in other age groups.
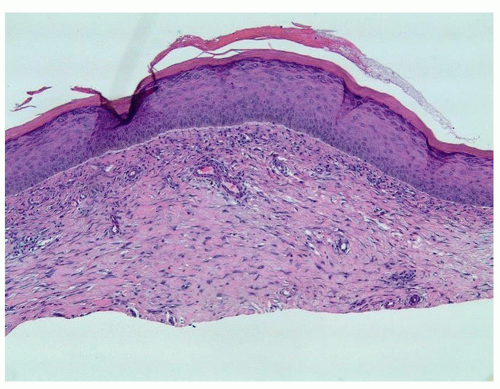
Histopathology
Histologically the composition of keloids is haphazard, thick collagen bundles with extra myofibroblasts and many overactive fibroblasts8 (Figure 8.2.5).
Subtypes
Keloids and hypertrophic scars can be both painful and pruritic. There are currently 4 clinical types of keloids: postincisional keloids (high tension areas, e.g., sternum,
shoulders), earlobe keloids (ear piercing) and other piercings, spontaneous keloids (acne lesions), and acne keloidalis nuchae.
shoulders), earlobe keloids (ear piercing) and other piercings, spontaneous keloids (acne lesions), and acne keloidalis nuchae.
PATHOGENESIS
The pathophysiology of keloid scar formation is not fully understood. Trauma to the skin, both physical (eg, earlobe piercing, surgery) and pathological (eg, acne, chicken pox), is the primary cause identified for the development of keloids. The presence of foreign material, infection, hematoma, or increased skin tension can also lead to keloid or hypertrophic scar formation in susceptible individuals. In other types of scars (trauma, burn, acne), scarring occurs when dermal injury occurs at a minimal depth of 0.56 ± 0.03.9 Keloids may form after any disruption of the skin integrity, including superficial injuries from scratches and insect bites. Keloids commonly occur after piercings, vaccinations, surgery, and/or skin diseases such as acne. In many cases, keloid formation is spontaneous after allergic reactions or skin disease.
Recently the National Institutes of Health called keloids a disease, and the pathology is defined by scarring. NIH—Epigenetics—identified 6 genes consistently present in all patients who develop keloids. It has been studied that genes originated in Africa, and 30% of all Caucasians have African genes. Keloid-associated genetic syndromes include Rubinstein-Taybi syndrome, Goeminne syndrome, and Ehlers-Danlos syndrome. Keloids and hypertrophic scars are associated genetically with HLA-B14, HLA-B21, HLA-Bw16, HLA-Bw35, HLA-DR5, HLA-DQw3, and blood group A.10
Keloid disease has a failure to suppress the wound healing process in a way that results in an excess growth of scar tissue. In 1962 Mancini and 1970 Peacock classified excessive scarring into hypertrophic or keloid scars. Based on these classifications, both scars grow above normal skin.11,12 Keloids are benign dermal fibroproliferative growths that extend beyond the original wound edges and invade adjacent normal tissue. Once a keloid is formed regression is very rare, and in fact, keloids may grow larger many years after the initial injury. The ability to grow years later is unique to keloid scars versus other types of scars. Chipev et al hypothesized this may be due to myofibroblast phenotype and apoptosis in keloid.13 Although keloids are technically classified as benign dermal growths, they often have the behavior of malignant cells with the ability to invade and have the hyperproliferative features of malignant tumor cells.7
The exact mechanism of keloid pathogenesis is still unknown.14 Abnormal cellular proliferations of fibroblasts produce the keloid. Keloid fibroblasts proliferate faster than those of hypertrophic scars and produce massive amounts of collagen and matrix metalloproteinases.15 Ehrlich et al have proposed that proinflammatory genes are upregulated via an inflammatory response in the microenvironment, which activates a series of signals to create keloids.15 Saint-Jean et al proposed in a recent study that acne scarring is revealed, even though acne is a disease of the pilosebaceous gland; however, the scarring occurred in locations far from the pilosebaceous gland. The authors hypothesized that inflammation is the single greatest reason for acne scar development and that the severity of scaring was due to the patient’s innate immunity inflammatory response.16
Fibroblasts in keloids become unregulated, and in keloid scar formation there is widespread proliferation of fibroblasts and apoptosis inhibition. Owing to the massive output of the overactive fibroblasts, there is an imbalance between collagen production and degradation of the extracellular matrix and an increase in specific cytokines.2
Several clinical studies have examined the possible role of wound tension, sebum secretion, and inflammation as possible factors of keloid scar formation.17 An interesting newer hypothesis has emerged suggesting that nutritional factors may play a role in keloid production. It has been observed that the triglyceride component in keloid scars is around 60%, which is significantly higher than in normal skin.17 Normal skin and keloids share the same ratios of cholesterol and fatty acids. This theory suggests that the alteration in lipid metabolism related to essential fatty acids stimulates the inflammatory reaction in keloids.
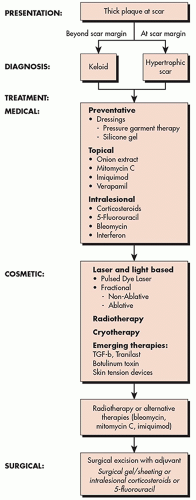
TREATMENT
The treatment of keloids and hypertrophic scars involves topical therapy aimed at maintaining light compression, medical treatment, most commonly intralesional corticosteroid or fluorouracil, fractional ablative lasers, and surgical excision.
Table 8.2.1 and Algorithm 8.2.1 describe the different treatment options and emerging options for keloids and hypertrophic scars on any surface area on the body. More extensive and aggressive treatments on the algorithm are required for keloids as opposed to hypertrophic scars.
TABLE 8.2.1 Keloid Modalities | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical
Preventive
All patients with keloids should carefully consider any elective procedure owing to the increased risk of keloid formation. If surgeries or procedures are medically necessary,
then topical, physical, and laser therapies may be tried as preventative modalities. Treatment of keloid scars is difficult and challenging because of this scar’s ability to recur. Wide ranges of therapies have been used to treat keloids.
then topical, physical, and laser therapies may be tried as preventative modalities. Treatment of keloid scars is difficult and challenging because of this scar’s ability to recur. Wide ranges of therapies have been used to treat keloids.
Dressings
Pressure Garment Therapy. Pressure garment therapy (PGT) has been used over the past 50 years for elevated scars.18 PGT is used in many medical centers as first-line treatments. The recommended pressure is 24 to 30 mm Hg for a period of 6 to 12 months after injury. However, Anzarut et al performed a meta-analysis involving 6 randomized, controlled trials with 300 patients and revealed there was not a significant difference between scars that received PGT and those that did not.19 Despite this meta-analysis, there has been some basic science research, which has emerged showing that wayward fibroblasts may be “turned off” by applying pressure.19 It is believed that compression works through activation of mechanoreceptors, which induce apoptosis.20
Occlusive Dressings: Silicone Gel Sheeting. Occlusive dressings are an important agent in the prevention and treatment of keloids. The effects of these dressings are believed to be related to both hydration as well as pressure from the occlusion. Emerging research has shown possible sodium (Na2+) channels may contribute to scar formation. Xu et al reported that the inflammatory response that results from disruption of epithelial barrier function after injury results in excessive scarring. They hypothesized that hydration status changes sodium homeostasis and induces sodium flux in keratinocytes, which result in activation of pathways responsible for scars.21 Different occlusive dressings include Cordran tape, non-silicone containing sheets, silicone gel sheets, and dressings.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree