Fig. 34.1
Division and main branches of the common carotid artery. (1) Common carotid artery, (2) internal carotid artery, (3) external carotid artery (proximal part), (4) occipital artery, (5) superior thyroid artery, (6) lingual artery, (7) facial artery, (8) external carotid artery (distal part), (9) glossopharyngeal nerve, and (10) vagal nerve
Surgical Technique
Donor Rats
The detailed surgical technique for harvesting the composite full-face flap was described in the related literature (Figs. 34.2 and 34.3) [1]. Herein, only a brief description of the surgical technique is presented. First, a circular neck incision was made anteriorly and posteriorly. Then, elliptical incisions were made around the eyes and, finally, perioral incisions were made. The flap was started so that it was elevated above the plane of the facial muscles. In the neck region, the submandibular gland and posterior belly of the digastric muscle were removed, and the greater horn of the hyoid bone was divided. The carotid sheath was explored, and the internal carotid artery was ligated and divided at its origin from the common carotid artery. The external carotid artery was dissected up to its bifurcation into the facial and superficial temporal arteries following ligation of the occipital, superior thyroid, and lingual branches. The external jugular vein was dissected following ligation of the anterior facial vein branches [1].
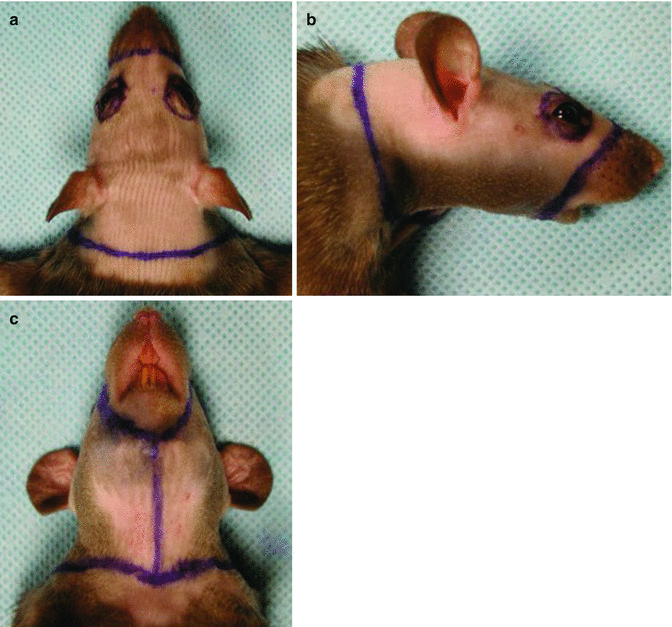
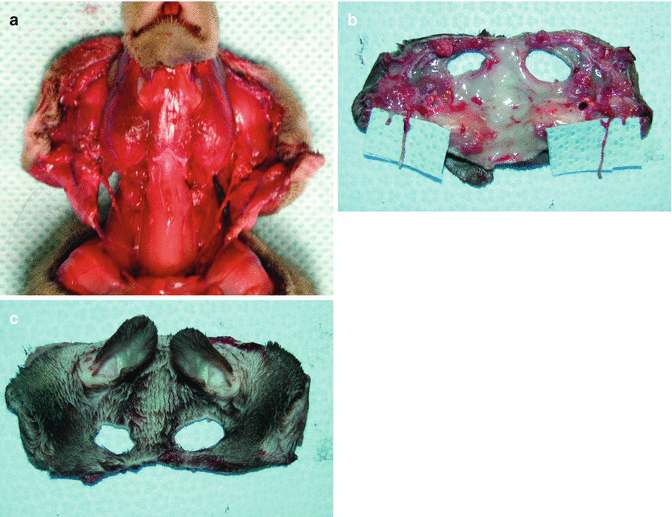

Fig. 34.2
The skin incisions in the LBN (RT11+n) donor rats. (a) Dorsal, (b) lateral, and (c) ventral appearances

Fig. 34.3
Harvesting the full face flap from the donor. (a) Full face flaps, including the external ears and scalp, based on the common carotid artery and external jugular vein, were harvested from the donors. (b) Outer, and (c) inner aspects of the harvested flap
The posterior auricular artery and superficial temporal artery were preserved, but the internal maxillary artery was transected. The external auricular canal was detached from its bony insertion and the ear was included in the composite fascial flap. Then, to maintain adequate lengths of the pedicles, the facial-scalp flap was harvested based on the bilateral common carotid arteries and external jugular veins (Fig. 34.3) [1].
Recipient Rats
In the recipient rats, similar incisions as described for the donors were made, and the entire face and scalp tissues, except the perinasal, perioral and periorbital tissues, were removed and a defect was created (Fig. 34.4). Different recipient vessels, including the common carotid artery, external jugular vein, external carotid artery, anterior facial vein, and facial artery, were prepared for the anastomosis [1].
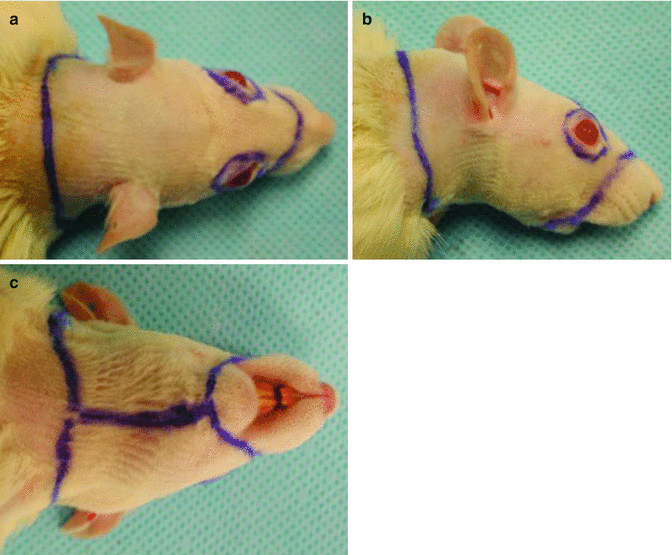

Fig. 34.4
Preparation of the Lewis (RT11) recipient rats. (a) Dorsal, (b) lateral, and (c) ventral appearances
Isotransplantation of the Non-vascularized, Composite, Full Face Transplantation Model
Following the completion of the anatomical study, the authors performed isotransplantation of the non-vascularized, full face, composite graft to demonstrate whether composite tissue could survive without anastomoses of the vascular pedicles. In this group, total flap necrosis was observed [1].
Allotransplantation of the Non-vascularized, Composite, Full Face Transplantation Model
Similar to the non-vascularized transplantation of the isografts, the authors also performed a non-vascularized transplantation of the full face, composite allografts to investigate whether the composite allografts could survive without anastomoses of the feeding vessels. In this group, total flap rejection and necrosis were observed [1].
Isotransplantation of the Vascularized, Composite, Full Face Transplantation Model
Before the allotransplantation studies of the full-face allograft, the authors performed 18 isograft transplantation between Lewis rats to test the feasibility of varying combinations of the vascular pedicle of the full face composite tissue. They found that when the distal part of the external carotid artery and anterior facial vein were used as recipient vessels, the highest animal survival rate was achieved. In this vascularized isograft group, total and partial flap necrosis were the most encountered complications. The survival times for the selected isograft groups ranged from 3 to 112 days [1].
Allotransplantation of the Vascularized, Composite, Full Face Transplantation Model
The authors performed allotransplantation of the vascularized, composite, full face transplantation model to confirm the feasibility of a total facial/scalp allograft transplantation. Thus, transplants were performed across major histocompatibility (MHC) barriers between semi-allogenic Lewis-Brown-Norway (LBN) (RT11+n) donors and Lewis (RT11) recipients (Fig. 34.5) [1].
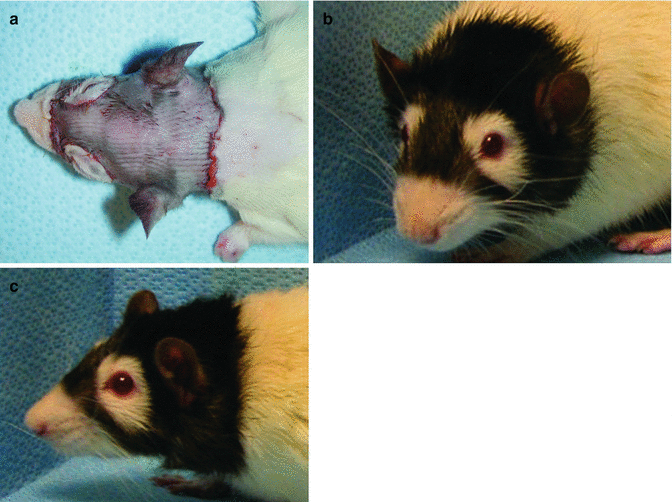

Fig. 34.5
Post-transplantation views of the recipients. Transplantation was performed between semi-allogeneic LBN (RT11+n) donors and Lewis (RT11) recipients. (a) Immediate post-transplantation view of the full face allograft recipient, (b, c) Late post-transplantation view of the full face/scalp allograft recipient on low-dose cyclosporine A monotherapy at day 200, showing no signs of rejection
As briefly mentioned in the surgical techniques section of this chapter, the entire facial skin, scalp, and both ears were harvested based on the common carotid arteries and external jugular veins from the semi-allogenic LBN (RT11+n) donors (Fig. 34.3).
In the Lewis (RT11) recipient rats, a defect was created, including facial skin, scalp and external ears, as occurred in the LBN (RT11+n) donor rats. However, so as to avoid functional deficits that could interfere with recipient animal feeding, breathing, and eye closure, the facial nerves and muscles, and the perioral and periorbital regions were preserved [1].
To vascularize the full face composite allograft, the bilateral common carotid or external carotid arteries were used. Arterial anastomoses were performed in an end-to-side fashion to the common carotid arteries or in an end-to-end fashion to the external carotid arteries of the recipients. Venous anastomoses were performed in an end-to-end fashion to the external jugular and anterior facial veins of the recipients [1].
Cyclosporine A (CsA) monotherapy was used in the allograft recipients during the postoperative period. The dose of the immunosuppressive regimen was 16 mg/kg/day for the first post-transplant week. In the following weeks, the dose was reduced to one-half of that of the previous week, and, finally, it was tapered to 2 mg/kg/day over 4 weeks, and maintained at this level during the follow-up period of over 200 days (Fig. 34.5b, c) [1, 2].
Modifications of Arterial Anastomoses of the Vascularized, Composite, Full Face Allotransplantation Model
Since the introduction of the full face composite tissue transplantation model, various models of face transplantation, such as hemiface transplantation, were created and developed in rats [3–16




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








