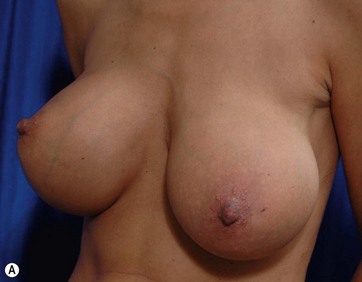
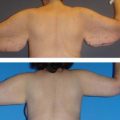
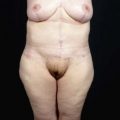
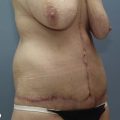
Chapter 7 Fat grafting to the breast
• Fat harvesting should be done with maximum care and minimal negative pressure to prevent damage to the fat cells.
• Patient selection and education to manage expectations will maximize patient satisfaction.
• The fat should be injected in multiple layers to prevent pooling and cyst formation.
• The use of 3D imaging will help to quantify the pre- and post-injection breast volume. This will help to measure the fat survival percentage.
• Fat harvested from different donor sites should be mixed before injection to have a similar fat cell population in both breasts.
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
Introduction
The current popularity of fat grafting, in many ways, is because of the work done by Dr. Sydney Coleman. He popularized the technique for harvesting the fat using a handheld syringe and centrifuging for compaction.1
The need for large volume fat harvesting and injection could not be met by the current techniques due to the length of time needed to harvest fat and centrifuge the fat. The average time for harvesting, processing and injecting fat by the Coleman technique is 100 cm3 of fat in 1 hour.2
History of Fat Grafting
The history of fat grafting goes back to 1893 when Neuber used it for facial defects.3 In 1895, Czerney used fat grafting for chest wall deformity post mastectomy.4 The early experimentation with fat grafting was not popular because, at a time when little was known about tumescent liposuction, most fat harvesting was performed by excision. The use of tumescent liposuction has given us the ability to harvest large amounts of fat with minimal donor site morbidity.
In 1987, Mel Bircoll published his work on fat grafting to the breast.5,6 The shortcomings of his work were poor long-term follow-up and a lack of mammographic studies as indicated by letters to the editor from 1987.7 American Society of Plastic Surgeons (ASPS) published a position paper in 1987 “Deploring the use of fat injection in breast augmentation” due to the fear of interference with breast cancer detection.8 The main concern at that time was that calcification from necrotic fat would cause interference with breast tissue evaluation by mammogram. At the same time published papers stated that surgical procedures such as breast reduction,9,10 augmentation11,12 and any breast procedure can also cause calcification. Further studies indicated that calcification from fat necrosis is easily distinguished from breast cancer calcification.13–20
There was a long gap before any work was published on fat grafting to the breast. Dr. Coleman published his work on fat grafting to the breasts in 2007. His results were remarkable and showed long-term survival of the injected fat.2
In 2007, ASPS and The American Society for Aesthetic plastic surgery (ASAPS) jointly and strongly supported the ongoing research efforts that would improve the safety and efficacy of the procedure21 but also cautioned against fat grafting for breast augmentation.
Principle of Low Pressure Fat Harvesting
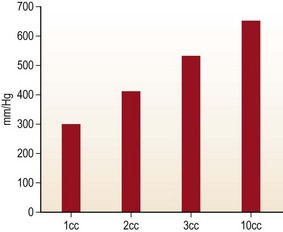
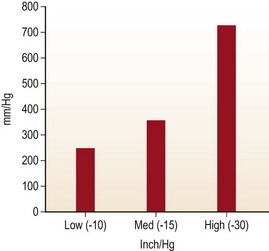
Handheld syringe aspiration is accepted as a gentler technique for fat harvesting. There are no published data to support this claim. We at LSU division of Plastic and Reconstructive Surgery undertook the study to find out the negative pressure that is generated by handheld syringes and liposuction machines. The graph in Fig. 7.1 shows the in-situ maximum pressure within the 1, 3, 5, 10, 30 and 60 cc syringes. Negative pressures were measured in vitro and in situ with 1, 2, 3, and 10 cc pull on a 10 cc Luer-Lock syringe as well as a conventional liposuction machine at 254 mmHg, 381 mmHg, and 762 mmHg (Figs 7.1, 7.2, and 7.3).
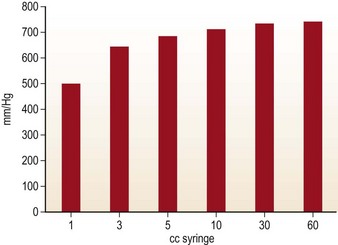
FIG. 7.1 The graph shows the in-situ maximum pressure within the 1, 3, 5, 10, 30, and 60 cm3 syringes.
The graph in Fig. 7.2 shows the negative pressure produced in a 10 cc syringe to be as high as 660 mmHg with full pull and 280 mmHg with only 1 cc pull on the 10 cc syringe.
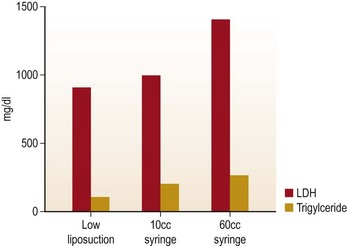
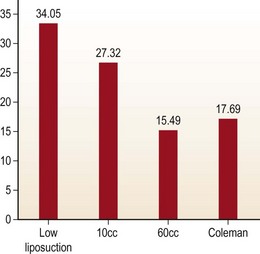
In order to evaluate the extent of damage to fat cell caused by all harvesting techniques, lipoaspirant samples were analyzed using lactate dehydrogenase (LDH) and triglyceride levels (Fig. 7.4). Glucose-3-phosphodiesterase (G3PD) assay was used to measure cell viability (Fig. 7.5). LDH and triglyceride are intracellular components and their presence in the aspiration fluid indicates cell damage and cell wall disruption. The results suggests that the samples harvested with the low negative pressure liposuction machine were lower in LDH and triglycerides. The G3PD assay suggests cell viabilities were high in samples from the low-pressure liposuction machine in comparison to the handheld syringe, which indicates more viable cells by the low pressure technique.
Harvested fat cells were stained with fluorescent dyes for imaging using scanning electron and confocal laser electron microscopes (Fig. 7.6). The slides show the outer cell layers bursting from the negative pressure.
Autologous Fat Grafting to the Breast
Technique
Fat Cell Preparation
The process of centrifugation increases the risk of contamination and cell damage. One advantage of centrifugation is to compact the fat cells. Another advantage of centrifugation is the ability to separate damaged cells from intact cells. Centrifugation at a low speed for a short period (100 g/30 s) will give the same compaction as a higher speed and longer period (1400 g/3 min). Refer to Fig. 7.7. The other observation is the lack of oil on the top of the syringe with low pressure fat harvesting. This observation is consistent with our study showing that the fat cells harvested by low pressure are damaged less, and less oil leaks out.
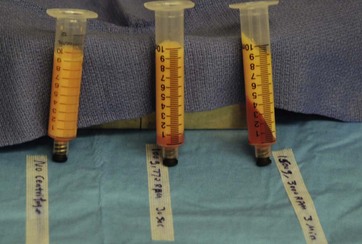
FIG. 7.7 The picture shows the lack of free oil on the top after centrifugation at 100 g and 1500 g.
< div class='tao-gold-member'>