Enucleation and Techniques of Orbital Implant Placement
Enucleation refers to the removal of the eye and the anterior portion of the optic nerve from the orbit. It is a complex procedure that alters the volume and normal anatomic relations of the orbit and may be complicated by eyelid malpositions, superior sulcus deformities, and implant migration or extrusion. Ideally, the technique selected should minimize the potential for development of these complications while providing for volume replacement, prosthetic motility, and maintenance of adequate fornices for prosthesis retention.
Myriad orbital implants have been developed in an attempt to improve the results of enucleation surgery, many of which have been plagued by increased postoperative complications. Exposed integrated implants and tantalum mesh spheres were designed to improve motility but are now rarely used because of their high extrusion rates. The earlier semi-integrated implants such as the Allen, Iowa, and Universal implants transmit movement from the irregular anterior surface to the back surface of a custom-fitted prosthesis. Improved motility could be attained and, as the prosthesis was supported by the implant rather than the inferior fornix, lower eyelid malpositions and superior sulcus abnormalities occurred less frequently. Socket discomfort and extrusion, however, were found with both the Allen and Iowa implants, often attributed to conjunctival erosion over the surface prominences. The Universal implant was designed with lower, more rounded protrusions to reduce the incidence of these complications, and the extrusion rate appeared to be comparable with that associated with spherical implants.
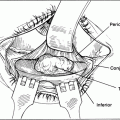
Due to problems of fitting, implantation, and exposure associated with buried, quasi-integrated implants, however, there was a subsequent shift back to simpler, buried spherical implants, such as silicone, glass, or polymethyl methacrylate. These spheres are positioned within the muscle cone either anterior or posterior to the posterior Tenon’s capsule. The largest implant that can be accommodated within Tenon’s space without undue tension at closure is placed. Implantation behind the posterior Tenon’s allows placement of a larger sphere with greater volume replacement and provides an additional tissue barrier to postoperative extrusion and migration. In addition, these spheres may be wrapped in sclera or fascia to reduce the risk of implant migration or extrusion and improve motility. Motility in unwrapped implants is felt to be dependent on forniceal movement, as the smooth contour of the spherical implant cannot engage the overlying prosthesis. Attachment of the extraocular muscles to the conjunctival fornices may thus enhance prosthesis motility. Imbrication of the extraocular muscles directly in front of the implant is less efficient in transmitting movement and is associated with postoperative sphere migration. When the implants are wrapped, the muscles may be attached to the “muscles windows” created in the sclera.
Porous orbital implants, such as coralline hydroxyapatite (HA) and porous polyethylene (PP), are currently among the most commonly used orbital implants. Coralline HA is a biocompatible integrated orbital implant, which first received FDA approval in 1989. Since then, numerous other porous implants have been developed. The Bio-Eye HA Orbital Implant (Bio-Eye; Integrated Orbital Implants, San Diego, California) is a natural HA derived from a specific genus of marine coral whose exoskeleton is completely porous: each pore connects with every other pore. In 2003, a newer version of the Bio-Eye was introduced, the Coated Bio-Eye HA Orbital Implant, which is coated in polymers that have a smooth surface and allow direct suturing of the muscles to the polymer. PP implants (MEDPOR; Porex Surgical Inc., Fairburn, Georgia) are less biocompatible than HA, but they are usually well tolerated and have a smoother surface allowing for easier implantation. Other porous implants include the synthetic HA implant developed by FCI (Issy-Les-Moulineaux, Cedex, France), which is not available in the United States, and the MOLTENO M-Sphere (Innovative Ophthalmic Products, Costa Mesa, California), which is a HA implant derived from the cancellous bone of calf fibulae. Bioceramic Orbital Implants (FCI; Issy-Les-Moulineaux, Cedex, France) are made of aluminum oxide (Al2O3), which is a ceramic implant biomaterial that is a porous inert substance allowing fibrovascular ingrowth similar to the Bio-Eye.
Fibrovascular ingrowth into porous spherical implants reduces the potential of implant migration and extrusion and allows the implant to be coupled directly to the prosthesis, thereby providing superior prosthetic motility. In 1992, members of the American Society of Ophthalmic Plastic and Reconstructive Surgeons were surveyed and 56% used HA for enucleations. In 2003, 70% of those surveyed used porous implants in >70% of enucleations and eviscerations. The implants are wrapped to provide an additional tissue barrier and reduce the risk of exposure, as uncovered porous polyethyelene implants appear to have a high exposure rate. The implants may be wrapped in preserved sclera, which provides a surface onto which the extraocular muscles are attached through the “muscle windows” created in the sclera. Other possible donor tissue may be used as wrapping material, such as bovine pericardium or acellular dermis. Thorough screening and appropriate precautions must be taken when implanting donor material to reduce the risk of transmission of infectious disease. In addition, autogenous tissue (fascia, posterior auricular muscle, or periosteum) or synthetic materials such as polyglactin may be used to wrap the implants and eliminate the theoretical risk of infection transmission with donor material. Autogenous tissue harvest adds surgical time, however, which must be weighed against the exceedingly low risk of infectious disease transmission, given appropriate screening and processing of donor tissue.
Newer technologies have been released that preclude the need for scleral wrapping by allowing for direct suturing of the muscles to the implant. The MEDPOR smooth surface tunnel implant (MEDPOR SST; Porex Surgical Inc., Fairburn, Georgia) is a PP implant with predrilled holes allowing for direct suturing of the muscle to the implant. The Alphasphere Orbital Implant (Alphasphere; Addition Technology, Inc., Des Plaines, Illinois) is a newer technology, which has 510 K approval but has not undergone FDA clinical trials. It is made of poly 2-hydroxyethyl methacrylate, which is modified physically to create a posterior gel and anterior spongy surface, with a Mersilene mesh placed at the transition between the two. The sutures are intended to pass through the spongy anterior gel and down through the Mersilene mesh to ensure that the muscles remain attached to the implant.
Once the implant is incorporated in the orbital tissues (-6 months after implantation), some oculoplastics specialists advocate the use of a peg that inserts into the implant and integrates with the ocular prosthesis to transmit the full range of implant motility. Peg insertion should only take place after the implant has become fully vascularized, which may be assessed with imaging studies or based on time from initial surgery. Peg placement couples to the prosthesis and may allow for increased prosthesis motility as they have been found to exhibit better large-amplitude saccades as well as improvement in horizontal ocular movement when compared to nonpegged implants (Shields et al., 1992; Guillinta et al., 2003). Despite these potential benefits, peg placement is associated with a high rate of complications (Jordan et al., 2001). Appropriate candidates for pegged implants must understand the increased follow-up visits and socket care as well as the increased complications of pyogenic granuloma, conjunctival overgrowth, extrusion, spontaneous loosening of the peg sleeve, pain, clicking, and chronic discharge (Jordan, 2001) associated with their use. In addition, peg placement requires coordination with the ocularist to couple the prosthesis for optimal outcome. Therefore, it is important to have appropriate patient selection for peg placement and to acknowledge the risks associated with pegs, which have led many to discontinue their use.
The original peg system was the Bio-Eye polycarbonate peg, which was attached to the posterior surface of the prosthesis and was associated with difficulty of insertion of the prosthesis (Kolberg, 1996). Subsequent modifications led to the eventual peg-and-sleeve system. In this method, the implant is placed primarily without simultaneous peg placement. Once the implant is completely vascularized (which is best assessed by gadolinium-enhanced magnetic resonance imaging or bone scan), a drill can be used to create a perpendicular shaft into the porous implant. This hole can then be enlarged until the sleeve can be screwed into the hole. A flat peg is then placed in the sleeve, which is later replaced with a round peg. A titanium modification of the peg-and-sleeve system was more recently developed and has been associated with lower complications when compared to its plastic counterpart (Shoamanesh et al., 2007). It is available for use with the bioceramic implant, FCI synthetic HA implants, and the Bio-Eye (Jordan and Klapper, 2006). Other titanium models include the MEDPOR motility coupling post, for use with PP implants.
Given that the complication rate associated with peg placement has been reported to be as high as 37%, most oculoplastic surgeons do not favor its use. In an attempt to improve implant coupling with the prosthesis without these associated risks, Porex Surgical developed a Universal-like implant made out of PP, the MEDPOR QUAD Motility Implant (MEDPOR; Porex Surgical Inc., Fairburn, Georgia) (Anderson et al., 2002). It is a rounded orbital implant with four mounds located anteriorly that project into Tenon’s and conjunctiva and allow coupling of the implant with the ocular prosthesis without penetrating through the conjunctiva. In addition, the implant creates two channels for suturing of the muscle stumps and has an increased posterior length, which is proposed to provide greater volume replacement. Each implant has certain advantages and disadvantages, and it is at the discretion of the surgeon to determine the most appropriate one for each case.
INDICATIONS
The choice between enucleation and evisceration is often controversial. However, in the surgical management of a suspected intraocular malignancy, enucleation is the procedure of choice. Evisceration risks incomplete tumor removal and precludes an intact specimen for pathologic examination. In the presence of an inadequate scleral shell, such as may occur with severe trauma, phthisis bulbi, or following scleral buckling procedures, enucleation is also preferable to optimize volume replacement. Enucleation removes uveal tissues more effectively than evisceration and is indicated when the slight risk of sympathetic ophthalmia following evisceration is unacceptable.
Enucleation may be considered in the treatment of any painful or unsightly blind eye for pain control or cosmetic rehabilitation. However, superior cosmesis and motility are usually achieved if the natural globe can be preserved and fitted with a scleral shell or cosmetic contact lens. A Gunderson flap may improve tolerance of a scleral shell if corneal sensation is problematic. It is often possible to restore comfort to a blind painful eye nonsurgically with topical medications, cyclocryotherapy, or retrobulbar alcohol injections. Retrobulbar alcohol injections, however, may be associated with increased orbital scarring, which may be encountered intraoperatively if the patient elects to have an enucleation in the future. In children, particularly, the globe should be preserved whenever possible to provide a stimulus for orbital growth and development.
PATIENT PREPARATION
The procedure is explained, including the temporary use of a conformer and the plan for prosthesis fitting at 5 to 6 weeks following surgery. The patient is prepared for the loss of full motility and the possibility of an asymmetric appearance. The potential complications of infection and extrusion should also be discussed.
ENUCLEATION
Enucleation may be performed under local retrobulbar or general anesthesia. If the eye to be removed is not readily apparent by external examination, the operative side should be reconfirmed by reviewing clinic notes, the operative consent, and by performing an indirect ophthalmoscopy in the operating room. The unoperated fellow eye is protected with a metal Fox shield under the drape throughout the procedure.
A speculum is placed between the eyelids and a 360° conjunctival peritomy is performed adjacent to the corneal limbus with Westcott scissors.
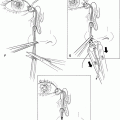
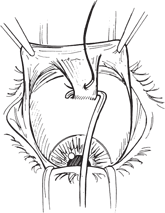
Tenon’s capsule is separated from the underlying sclera to the level of the rectus muscle insertions. A Stevens tenotomy scissors is passed into each quadrant and is gently spread to separate Tenon’s capsule from the sclera between the rectus muscles. A muscle hook is passed behind a rectus muscle insertion.
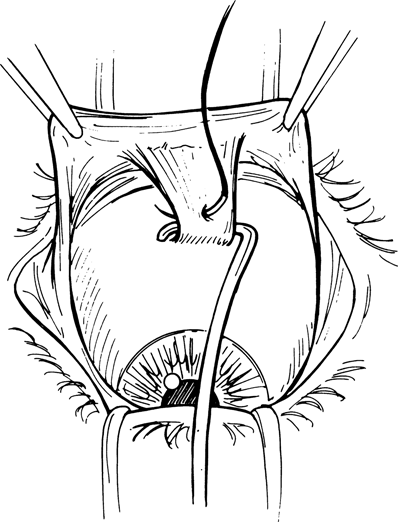
Figure 27-1. A locking suture of double-armed 5–0 Vicryl is placed in the muscle tendon near the insertion, and the muscle is then disinserted. The ends of the suture are clamped to the surrounding drape with a hemostat. This procedure is repeated with the remaining rectus muscles. A 1- to 2-mm stump of medial and lateral rectus tendon left adherent to the sclera is useful for traction later in the procedure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree