38 Cosmetic Use of Botulinum Toxins and Tissue Fillers
Interest in facial rejuvenation has increased considerably over the last decade. As patients are living longer, and as a “youthful” appearance remains a priority in our society, demands for noninvasive aesthetic procedures increase. The introduction of neurotoxins and soft tissue fillers has made facial rejuvenation more accessible to patients seeking less expensive, less disabling, and lower risk minimally invasive facial rejuvenation. While some aesthetic surgical procedures have seen diminishing use over the last decade, neurotoxin and filler procedures have gradually increased. Neurotoxins work best for dynamic wrinkles, but do not completely eliminate “static” wrinkles or gravitational changes. By contrast, fillers are an excellent choice to address static wrinkles and can also be used as volumizing agents to augment facial volume. Thus, neurotoxins and fillers are an excellent noninvasive combination for facial rejuvenation in selected patients. Neurotoxin and filler injection techniques are acquired with proper training and practice. This chapter introduces neurotoxins and fillers, their uses, techniques for delivery, and complications.
BOTULINUM TOXINS
As experience with neurotoxins has expanded, the popularity and range of treatment indications have grown. Botulinum toxin (BoTX) has many applications for improving facial appearance by altering muscle action. These include kinetic facial wrinkle lines, prominent orbicularis in the lower eyelids, and depression of the lateral oral commissure, dimpling of the chin, brow ptosis, and balancing of the facial aesthetic units. BoTX is a safe and effective treatment for hyperfunctional glabellar frown lines, crow’s feet, and forehead lines. BoTX has also been used to treat platysmal bands, brow position, mild eyelid ptosis, melolabial folds, migraine headache, and palmar and axillary hyperhidrosis. The applications of this medication will continue to expand as surgeons explore additional areas of use.
There are multiple types of facial wrinkling: gravitational redundancies, intrinsic aging with loss of elasticity, sleep creases, and dynamic facial lines. Dynamic lines induced by muscle contraction are the type of wrinkling most amenable to BoTX treatment. These dynamic facial wrinkles occur perpendicular to the force of muscle contraction. It is theorized that repeated creasing of the skin over time induces changes in the dermal structure, resulting in the eventual formation of a crease line at rest as well as deepening of the crease when the muscle is contracted. BoTX works by preventing the expression of impulses sent by the motor neurons to the neuromuscular junction. Injection of the solution results in a temporary weakening of the muscle contractions in the area of the injection.
Muscle weakening diminishes the dynamic wrinkles, but also has cosmetic applications for changing facial morphology. Brow elevation is accomplished by selectively denervating the depressor muscles of the brow, resulting in unopposed frontalis action. Decreasing the action of the depressor anguli oris may elevate the corners of the mouth. Relaxing the contraction of the lower eyelid orbicularis will soften the prominence that becomes apparent with smiling and contraction of the muscle. Meticulous knowledge of the actions and interactions of the facial musculature allows the physician to selectively modify facial features dependent on muscle contraction.
Pharmacology
Neurotoxins produced by the Gram-positive, anaerobic Clostridium botulinum are some of the most potent neurotoxins known to mankind, and are the causative agents of botulism. Seven distinct antigenic BoTXs (-A, -B, -C, -D, -E, -F, and -G) produced by different strains of C. botulinum have been described (Shone, 1986). The human nervous system is susceptible to five toxin serotypes (-A, -B, -E, -F, and -G) and unaffected by two (-C and -D) (Coffield et al., 1997). All serotypes act on the peripheral nervous system where they inhibit release of acetylcholine from the presynaptic terminal of the neuromuscular junction (Niemann, 1991). At higher doses, toxins may bind to nerve terminals at autonomic cholinergic ganglia with autonomic effects (Dressler and Benecke, 2002). BoTX may also have inhibitory effects on the release of other neurotransmitters such as substance p (Purkiss et al., 2000).
There are three distinct steps involved in botulinum’s neurotoxicity effects. The first step is the irreversible binding of BoTX to presynaptic cholinergic receptors via the H chain’s 50-kDa carboxy terminal (Black and Dolly, 1986a,b; Schiavo et al., 1992). The second step involves internalization of the neurotoxin through a receptor-mediated endocytosis (Brin, 1997). This process is independent of calcium and is partially dependent on nerve stimulation. After internalization, the disulfide bond is cleaved by an unknown mechanism. The third step is neuromuscular blockade. Within the synapse, isoforms of proteins form a complex platform for docking and fusion of acetylcholine vesicles to the cell membrane before they can be released (Sollner et al., 1993; Huttner, 1993). These protein isoforms are vesicle-associated membrane protein (VAMP, also known as synaptobrevin), synaptosomal-associated protein (SNAP-25), and syntaxin. Alteration of these membrane proteins prevents the binding of the vesicles that release the acetylcholine. The BoTX serotypes act in different locations on these docking proteins. In theory, the more profoundly the membrane proteins are structurally altered, the sooner they are replaced, and the faster the effects of the toxin abate. This may explain some of the differences in duration of effect of the different serotypes. While still inactivating the docking process, the longer acting serotypes may not alter the conformation of the molecule significantly enough to signal immediate replacement. With BoTX-A, clinical expression of the effects typically takes 24 to 48 hours, and maximal muscle weakening is not seen for a week. The onset of action for BoTX-B is more rapid, and the onset of different formulations of BoTX-A may vary.
Preparations
There are currently four commercially available BoTX preparations in the United States: BoTX-A: OnabotulinumtoxinA—Botox (Allergan, Irvine, California); AbobotulinumtoxinA—Dysport (Medicis, Scottsdale, Arizona); IncobotulinumtoxinA—Xeomin (Merz Pharmaceuticals, Frankford, Germany); and BoTX-B: RimabotulinumtoxinB—Myobloc (Elan Pharmaceuticals, San Diego, California). Comparable dosing for the four commercially available preparations is still under investigation, but may be in the range of 50 to 150 mouse units (MU) or (U) of Myobloc to 1 U of Botox and 3 U of Dysport to 1 U of Botox (Poewe, 1992). It should be noted that the Botox/Dysport conversion ratio does not account for antigenic potential (Aoki, 1999). Preliminary studies with Xeomin suggest that the dose range and activity may be similar to that of Botox.
Botox and Dysport both have U.S. Food and Drug Administration (FDA) approval for cosmetic treatment of glabellar folds. Botox, Myobloc, and Xeomin are indicated for the treatment of adults with cervical dystonia, with Botox and Xeomin having the additional indication of blepharospasm (U.S. Food and Drug Administration, 2010). Botox alone carries the additional FDA-approved indications of axillary hyperhidrosis, migraine headache, and strabismus (U.S. Food and Drug Administration, 2009). During the next decade, it is likely that other formulations of BoTX-A will become available. Mentor is actively developing an additional formulation of BoTX-A.
Vials of Botox contain 100 U of Clostridium BoTX-A, 0.5 mg of human albumin, and 0.9 mg of sodium chloride in a sterile, vacuum-dried form without a preservative. The vials are stored in the freezer or are refrigerated before reconstitution for clinical use. Vials may be stored up to 36 months prior to reconstitution. The recommended diluent is nonpreserved or preserved normal saline. Preserved saline may diminish injection discomfort. It should be noted that BoTX may become denatured by agitation; therefore there is no need to “shake” BoTX vials when reconstituting or diluting them. After reconstitution, the product is stored in the refrigerator at 2°C to 8°C, and the manufacturer’s labeling recommends use within four hours of reconstitution. Recent studies have shown that storage and use at up to one month following reconstitution does not seem to decrease efficacy or safety (Hexsel et al., 2003). The concentration of the material is dependent on the volume of added saline and may be described by how many units are present in 0.1 ml.
Botulinum Preparation
Either preserved or unpreserved sterile, injectable saline (0.9% sodium chloride) is used for reconstitution. Final dilution is a matter of preference; however, higher concentration solutions allow for more accurate delivery to a specific area by decreased diffusion due to volume effect (Table 38-1).
Table 38-1 Botulinum Preparations
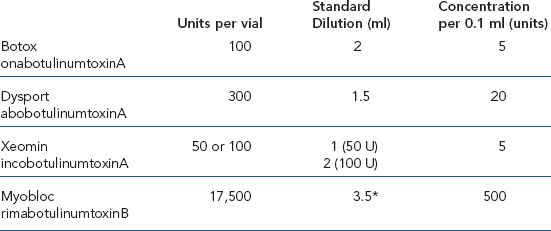
*Myobloc is already in a 0.9% saline solution at a concentration of 5,000 U per 1.0 ml, but may be further diluted if desired.
OnabotulinumtoxinA (Botox) Reconstitution. Either preserved or unpreserved sterile, injectable saline (0.9% sodium chloride) is used for dilution (Table 38-2).
The reported volumes of solutions used for cosmetic indications of Botox vary from 0.025 ml to greater than 1.0 ml per site. The effects of BoTX-A are dependent on the location, concentration, and the volume of solution that is injected. For cosmetic uses of Botox, the reported concentrations of the solution vary from 1 U/0.1 ml to 10 U/0.1 ml.
Dysport differs from Botox in terms of mouse units, chemical properties, biological activities, and molecular weight (Van den Bergh et al., 1995). It is supplied in 300-U vials, produced by column-based purification rather than by the precipitation technique used for Botox, and may be stored at room temperature. The recommended diluent is also unpreserved normal saline (Lowe, 1998) (Table 38-1). When used at a dosing ratio of 3:1 U, Dysport is slightly less expensive than Botox. Because there are 300 U of Dysport and 100 U of Botox per vial, if the surgeon wishes to use a 3:1 conversion ratio, one option is to add the same volume of diluent into the Dysport vial that one normally places in the Botox vial and then inject the same volume that one normally would if using Botox. This will result in a 3:1 dose ratio. However, dilution possibilities are limited by the size of the Dysport vial. The dosing protocols provided by Medicis, Dysport’s vendor, suggest using 2.5 ml/vial of saline diluent. This amount or less works well. Fitting 4 ml into the vial is difficult.
AbobotulinumtoxinA (Dysport) Reconstitution. Either preserved or unpreserved sterile, injectable saline (0.9% sodium chloride) is used for dilution (Table 38-3).
Myobloc is the only commercially available preparation of BoTX-B and is supplied as a sterile injectable solution in 3.5-ml glass vials. The vials are available in 2,500 U, 5,000 U, and 10,000 U volumes. The BoTX-B is in solution with 0.05% human serum albumin, 0.01 M sodium succinate, and 0.1 M sodium chloride at pH 5.6. The bottles are all ready-to-use solutions and do not require reconstitution. Myobloc is stable for 21 months in refrigerated storage. Myobloc is FDA-approved for use in cervical dystonia. BoTX-B has a faster onset than BoTX-A, but the duration of action is noticeably shorter at 7 to 8 weeks, making its use for cosmetic applications limited (Matarasso, 2003; Yamauchi and Lowe, 2004).
Contraindications for BoTX Treatment
The following are contraindications for BoTX treatment:
- Prior allergic reaction
- Injection into areas of infection or inflammation
- Pregnancy Category C
- Breast-feeding (unknown if excreted in human milk)
- Diseases of the neuromuscular junction (e.g. myasthenia gravis or Lambert-Eaton neuropathy) or peripheral motor neuropathic diseases (e.g., amyotrophic lateral sclerosis)
- Aminoglycosides or agents that interfere with neuromuscular transmission
- Contains human blood products (albumin)
Treatment Techniques
Pretreatment Considerations
Per FDA regulations, a copy of the Medication Guide must be distributed directly to each patient every time he/she receives a botulinum injection. A detailed patient chart/procedure note outlining injection pattern, units injected per site, type of botulinum injected, lot number of botulinum vial(s) used, and response/reactions to previous treatments should be kept for each treatment. Keep in mind, rhytids that are not dynamic in nature do not respond as well to treatment (e.g., caused by alteration in collagen, fat, or skin texture such as in age-related changes, gravitational descent, or photo damage). In general, treatment effects will not be visible for 3 to 5 days. Duration of treatment is variable, with 3 to 4 months being average. Be sure to set appropriate expectations for the outcome of treatment (i.e., deep, long-standing rhytids will not completely resolve with one treatment).
BoTX is administered by subcuticular or intramuscular injection. The supplies required for injection are basic and readily available (Table 38-4). Anesthesia is not routinely used and majority of the patients do no require preinjection anesthetic. However, if the patient’s pain tolerance is low, ice or a topical eutectic mixture of lidocaine 2.5% and prilocaine 2.5% (EMLA, APP Pharmaceuticals, Schaumburg, Illinois) or the equivalent may be applied over the prospective injection sites 30 minutes prior to treatment. Vibratory stimulus may also be of use in minimizing the perceived injection discomfort.
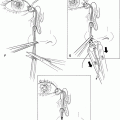
The patient should be seated comfortably in an upright position. To select the sites of injection to treat wrinkles, the patient is asked to squeeze and relax the muscles in the affected areas. The surgeon identifies the location of maximal skin displacement during the contraction of the muscle that is to be treated. A marking pen then marks the desired injection sites. The muscles should be relaxed before injection to minimize discomfort. Using the thumb and index finger of the noninjecting hand to “pinch” the skin at the site of injection may decrease discomfort and also aid to localize the injection point, particularly in the glabella. The needle is inserted at an angle into the muscle layer or the subdermal tissue just above the muscle. Unlike soft tissue filler techniques, the BoTX should be aimed at the muscle rather than into the crease to achieve most effect. The patient will experience the discomfort of the needle stick followed by a localized “stinging” as the solution is injected. There may also be some pressure sensation from the volume of the fluid injected. Some physicians believe a smaller volume of injection is less painful (Babamiri and Nassab, 2010).
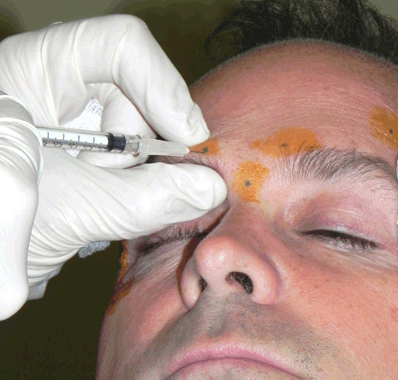
Figure 38-1. While grasping the skin at the site injection, the needle is inserted into the tissues above or into the muscle being treated.
The onset of action is variable from one patient to another and different from one injection to another in the same patient, but most notice alteration in muscle contraction within 24 to 48 hours. The maximal response in muscle weakness may not occur for 10 days with BoTX-A (Binder et al., 1998). The muscle weakening effect of BoTX-A is temporary and typically resolves four months after the injection. When the patient begins to notice the onset of BoTX effect, two phases of response are occurring. The early alteration in the dynamic wrinkle lines is due to a relaxation of resting muscle tone, decreased force of contraction, and perhaps shift in the tissue fluids. The second and more chronic process is remodeling of the dermis that should occur when the mechanical pressure of the contraction is relaxed. In addition to decreasing the wrinkle lines that are present, prolonged use of BoTX should prevent further deepening of the creases. The most dramatic responses to treatment are seen in patients in the age ranges of 30 to 50 years. In this patient population, the injections may obliterate the kinetic lines. With deeper wrinkles, treatment with BoTX flattens the edges of the indentation, but additional filler techniques are usually necessary to make the area smooth. BoTX has been used to treat glabellar folds, lateral periocular rhytids, lower eyelid orbicularis ridges, brow ptosis, eyelid ptosis, horizontal forehead wrinkles, perioral lines, mouth corner depression, and platysmal bands in the neck. It can be used as a singular therapy or in conjunction with other cosmetic surgical interventions. BoTX can be used to augment the results of laser resurfacing, chemical peels, collagen injections, endoscopic forehead surgery, and lower eyelid blepharoplasty. The treatment techniques discussed in this chapter are starting points and should be modified by the treating surgeon based on experience and patient response. Because Botox and Dysport are the only FDA-approved cosmetic BoTXs, the subsequent dosing descriptions will all refer to Botox with relatively equivocal Dysport dosages in brackets[]. Although currently not approved for cosmetic applications, the dosage of Xeomin would be similar to Botox.
Horizontal forehead furrows
Horizontal forehead rhytids result from repeated contraction of the frontalis muscle. Injecting the frontalis may change brow position, thus symmetric treatment for the two sides is recommended. This is particularly true for the patient’s first injection, until the physician can assess the individualized response to the injection locations. The patient is asked to wrinkle up the brow as if surprised, and a pattern above and below the crease lines is injected across the central forehead.
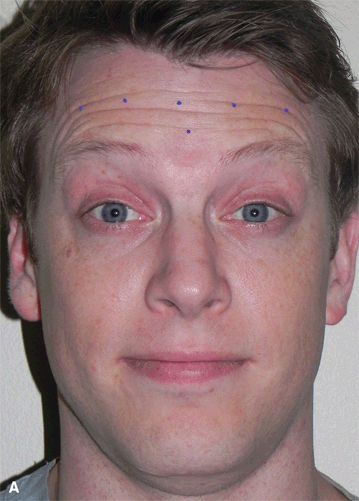
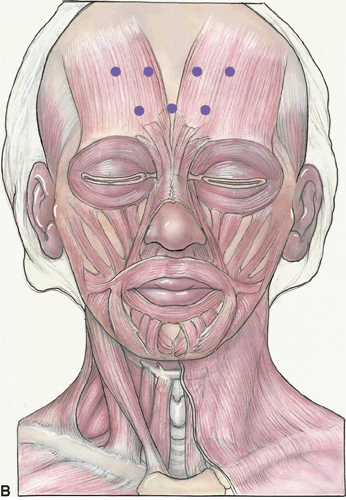
Figure 38-2A&B. Injection sites for forehead furrows are marked for the specific furrow pattern of the patient. Weakening the frontalis, the muscle that creates the horizontal wrinkles, will decrease brow elevation. The anatomical diagram (Figure 38-2B) illustrates the possible injection sites and the targeted underlying musculature. The concentration of the forehead injections is 5 U BoTX-A per 0.1 ml. The volume injected is 0.025 ml per site. Typically, 5 to 20 U [10–50 U Dysport] of Botox is used in the central forehead. In general, one injects with conservative caution in the area just above and at the lateral superior brow to minimize postinjection brow ptosis. Occasionally, this may result in overelevation of the lateral brow and require an adjustment of adding a small amount of Botox just above the eyebrow. It is easier to correct the frontalis overshoot than to improve postinjection brow ptosis. When using Dysport to treat the horizontal forehead lines, this is one area where Dysport may act somewhat differently than Botox, and lower doses are recommended. Here one may use a 2.5 to 1 dosing relationship between Dysport and Botox that is recommended in the package insert, rather than the 3 to 1 conversion recommended for elsewhere in the face. Anecdotally, there may be more diffusion of the Dysport in the frontalis than one sees with Botox. In the central forehead, one may wish to use a lower ratio of Dysport to Botox, perhaps 2.5 to 1 instead of 3 to 1.
Glabellar folds develop from the repeated contraction of the corrugator, orbicularis, and procerus muscles. The vertical folds are created by the corrugators, while the horizontal folds across the bridge of the nose are formed by the procerus muscle.
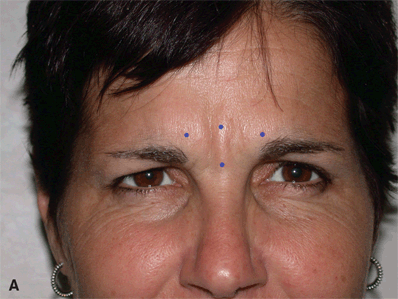
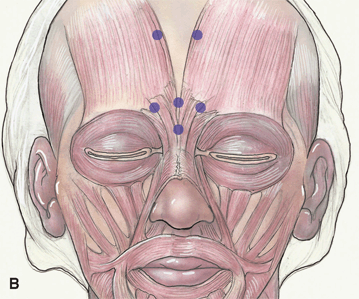
Figure 38-3A&B. To select injection sites for the glabellar folds, the patient is asked to squeeze the brows toward the midline and the location of the anterior bulge of the muscle is noted and marked. Mark the areas of bunching to correspond to the contraction of underlying corrugators and procerus musculature B. Anatomical diagram illustrating possible injection sites and the underlying musculature.
This is typically approximately 5 mm lateral to the vertical wrinkle lines. The patient may have two or more vertical wrinkle lines. The two sides of the face may be asymmetric, and the injection sites may be modified to accommodate for the asymmetry. The procerus is usually injected centrally between the two brows just above the bridge of the nose. In the glabella, the concentration of Botox solution is 5 to 10 U/0.1 ml, and the volume injected is 0.05 to 0.10 ml per site. About 20 to 30 U [50–90 U Dysport] is a typical injection dose for the glabellar region.
Lateral periocular wrinkles (Crow’s feet)
Treating the lateral orbicularis muscles diminishes crow’s feet lines. The concentration of the solution is 5 U/0.1 ml, and the volume per injection site is 0.025 to 0.05 ml per site. The injection sites follow the lateral orbital rim from midline up into the lateral brow.
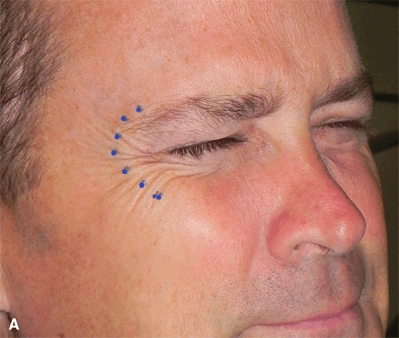
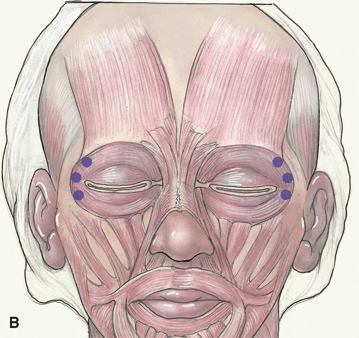
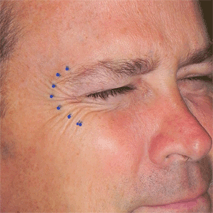
Figure 38-4A&B.Injection sites to treat lateral periocular wrinkles (Crow’s feet) are marked along the lateral orbital rim; sometimes treat up into the eyebrow cilia to get to the orbicularis. Anatomical diagram illustrates the strategic placement of injection sites to address the underlying circumferential band of orbicularis oculi muscle (Fig. 38-4B). Typically, the injections are given in three or four locations per side and have a total dose of 7 to 15 U [20–50 U Dysport] per side. When treating the lateral wrinkles, injections should be delivered temporal to the lateral orbital rim to minimize the chance of unintended eye muscle weakness and diplopia. The zygomaticus major/minor complex on the maxilla is also avoided by staying superficial in this area to prevent lateral mouth droop.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree