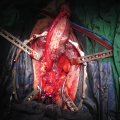
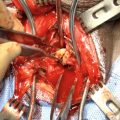
div class=”ChapterContextInformation”>
35. Tissue Engineering in Urethral Reconstruction
Keywords
Urethral scaffoldUrethroplastyAcellular bladder matrixMatristemUrethral strictureHypospadiasTissue engineeringSeeded scaffolds35.1 Introduction
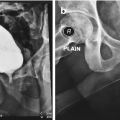
Urethra is the distensible tube that extends from the tip of the penis to the bladder neck. It functions as the pathway of urine in both female and males and semen in males only. It is divided into 2 portions anterior and posterior urethra. It is lined with pseudostratified epithelium except at fossa navicularis (stratified squamous epithelium) and posterior urethra (transitional epithelium) [1, 2]. It is subjected to many incidents during lifetime either congenital maldevelopment as hypospadias or epispadias or acquired diseases include trauma, inflammation, scarring or malignancy. All those events can lead to either obstruction of the urine passage or persistence of abnormal urethra opening not at the tip of the penis.
Many surgical techniques are available to repair urethral stricture or defects including the use of tissue transfer with high success rate. Tissues transferred for urethroplasty includes buccal mucosa, non-hairy skin, bladder or colonic mucosa [3].
However, the use of these tissues is hindered by their availability and morbidities associated with their harvest and transfer [4].
That is why there was a need for off-the shelf tissue to be used to bridge urethral defects and repair stricture in sufficient amounts and without morbidities. Tissue engineering for urethra has emerged as an ideal solution for that. Tissue engineering follows the principles of cell transplantation, materials science and engineering to would restore and maintain normal tissue and organ function [5].
Different strategies of urethral tissue engineering include the use of scaffolds alone, scaffolds with only growth factors/bioactive molecules or scaffolds with epithelial and muscle cells [6]. In all of those strategies, urethral scaffolds form the cornerstone for the regeneration of healthy urethral tissues.
35.2 Scaffolds
35.2.1 Functions of Urethral Scaffolds
Scaffolds can be stated as an artificial structures capable of supporting three-dimensional tissue formation, that allows cell attachment and migration, delivery and retaining of cells and biochemical factors enable diffusion of vital cell nutrients and expressed products [7].
In urethral tissue engineering, scaffolds provide mechanical support against the collapse, hence maintain the lumen of the future urethral tissue open and patent till full regeneration of the newly formed tissue. Also, they allow the delivery of both the epithelial and smooth muscle cells to the desired site for repair. By permitting the cell attachment, they help the growth and differentiation of the cellular component of rejuvenated tissues. Moreover, they can be used to deliver growth factors as VEGF (ref) to enhance tissue growth and/or inhibit fibrosis (like with an example and reference) [8–10].
35.2.2 Characters of Ideal Scaffold for Urethra
For any biomaterial used as scaffold in tissue engineering generally, it should biocompatible, biodegradable and non-toxic with unlimited availability. To be suited for urinary tract reconstruction (urethra, ureter and bladder), it needs to be non-permeable to water, resist encrustations and biofilm formation . To better fit in urethral regeneration, it is required to be pliable, complaint, stretchable and distensible.
The biomaterial should be biodegradable so that it can support the development of the new tissues and be absorbed at the end of this process. This avoids the inflammation and foreign body reaction that eventually may lead to scarring or the urethra. The absorption process of the biomaterial should occur at a rate that follows the rate of regeneration to prevent the mechanical failure of the newly formed segments [11].
The biomaterials provide temporary mechanical support sufficient to withstand in vivo forces exerted by the surrounding tissue and maintain a potential space for tissue development.
Biocompatibility refers to the ability of a material to perform with an appropriate host response in a specific situation [12].
Urethral scaffolds should allow cell attachment, growth and differentiation . It should not be toxic to the surrounding structures or stimulate inflammatory host response especially it will be implanted near the corpora responsible for erection.
Any biomaterial that will be used for urinary organs regeneration must be waterproof to prevent urine leakage into the developing tissues. Urine is cytotoxic to the growing cells and additionally, it can cause infection and extensive fibrosis. As the urine contains a lot of solutes, the scaffold should be resistant to encrustations and biofilm formation [13].
Specifically designed for urethral tissue engineering, the scaffold needs to be stretchable and pliable to avoid shearing of the material that could result from stress forces of erections especially in case of penile urethroplasty [14].
Additionally, it is mandated for the scaffold to be compliant and distensible so that it does not form rigid urethral tube . When urine passes through the rigid tube, there will be some sort of functional obstruction [15].
35.2.3 Types of Scaffolds
They can be classified broadly into nature and synthetic scaffolds.
Nature polymer as collagen and gelatin
Acelluar matrices as small intestinal submucosa, bladder submucosa, amniotic membrane, acelluar corpus spongiosum
Silk fibroin
Self-assembled scaffolds
Biodegradable as PLG and PLGA
Non-biodegradable as Poly(tetrafluroethylene) (PTFE)
Nature polymers as collagen or gelatin cannot be used as scaffold for urethral reconstruction as they are mechanically weak and relatively rapidly biodegradable. Likewise, non-biodegradabke synthetic material excites severe inflammatory reaction, infection and scar formation leading to rejection of the implanted scaffold [16].
Acellular Matrices/scaffolds: They are obtained by the decelluarization of tissues and organs with removal of cells through different mechanical and chemical processes [17, 18].
They have inherent advantages including similarity in biomechanical properties, presence of growth factors and structural proteins and availability from either xenogenic or allogenic sources. In a comparison between normal bladder tissue and acellular bladder matrix, strain, stress and elastic modulus were similar between them [19].
Growth factors have been found in SIS as TGF beta, FGF-2 and TGF and in bladder acellular matrix as VEFG and PDGF-BB [20, 21].
Organ specific acellular matrices are preferred over non-organ specifc matrices. In this context, bladder and corpus spongiosum acelluar matrices are preferred over SIS [22].
However, certain limitations exist with acellular scaffolds. The immune response is so intense that leads to severe inflammation and toxicity of the growing cells ending in fibrosis and urethral stricture. This may be attributed to residual cellular fragments after decelluarization [17].
Nevertheless, the new commercial bladder acelluar matrix has been found to have better immune response [23].
Addition of stem cells can help to modulate the immune response to acellular scaffolds. Although rare, there is a risk of infection with prion viruses especially with SIS [14].
Moreover, the size of the pores may not allow much infiltration of seeded cells into the scaffolds in case of using it as cell seeded graft. This is especially critical for smooth muscle cells which forms muscular layer that allows the regenerated tissues to be elastic, robust, prevent collapse, withstand the pressure during urine flow and prevent adhesions [24].
It seems that bladder acellular matrix would the best available option as a scaffold for urethral reconstruction being organ specific, availability in sufficient amounts (from porcine bladder) and relatively less immunologic reaction.
Synthetic polymers like polylactic acid (PLA) , PGA (polyglycolic acid), PLGA (poly lactic-co-glycolic acid). They have certain advantages including unlimited supply and ability to control porosity, ultrastructure and biomechanical properties , and degradation rate. They still lack intrinsic biologic activity mainly growth factors and proteins needed for cell attachment and migration [25].
Polyglycolic acid:poly(lactide-co-glycolide acid scaffolds been used in clinical studies as tubular cell seeded grafts to bridge urethral defect ranging 4–6 cm [26].
Self-assembly scaffolds: They are scaffolds made by the deposition of extracelluar matrix by stromal/fibroblast cells without exogenous materials. These scaffolds have many advantages including lack of immunologic reaction, having fibroblast cells with cell signaling and presence of many growth factors. However, there are some shortcomings concerning the need for biopsy to extract the stromal cells and relatively less favorable biomechanical features necessary for complex urethral reconstruction [6].
35.3 Approaches for Urethral Tissue Engineering
There are 2 main approaches for urethral regeneration; onlay/partial and tubularized/circumferential urethral replacement. Onlay urethral replacement requires at least half of urethral circumference is health so that it acts as a source of healthy cells to migrate and repopulate the scaffold [27].
Circumferential urethral replacement mandates the tabularized scaffold to be cell-seeded because the longest urethral defect that can be bridged without cells is only 5 mm; otherwise fibrosis will precede full cell growth [28].
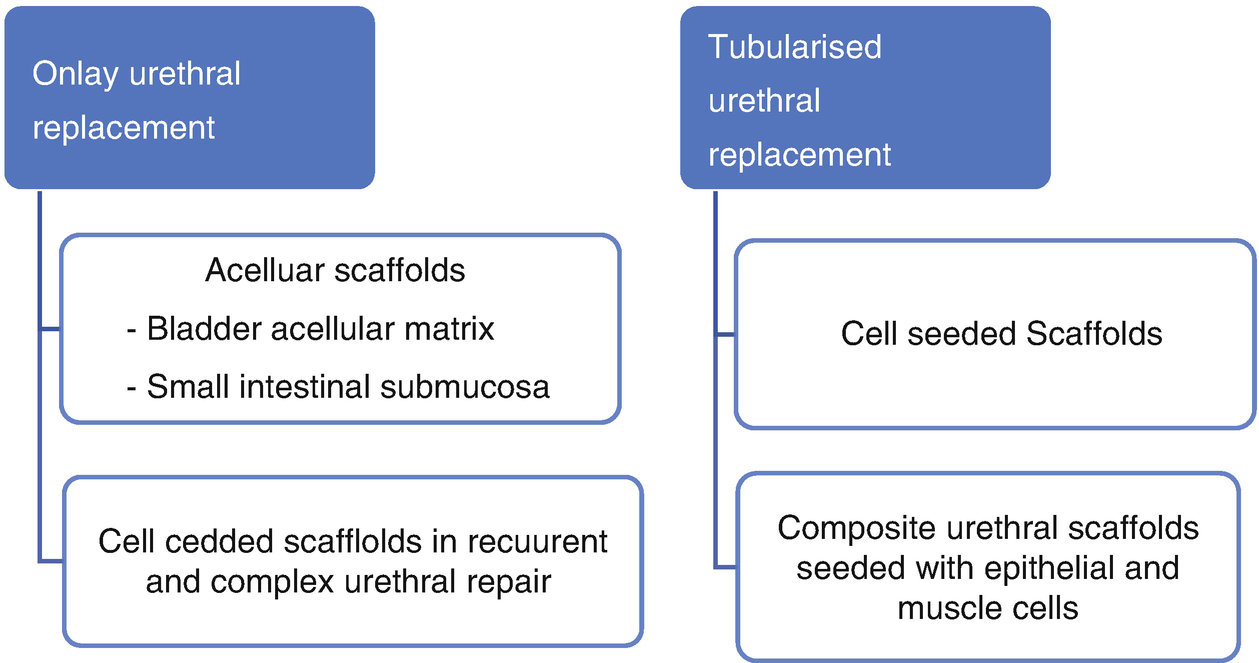
Strategies for urethral replacement with tissue engineering
35.3.1 Urethral Tissue Engineering Using Acellular Scaffolds
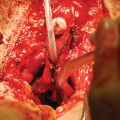
Unseeded SIS and bladder acellular matrix have been employed in many preclinical trials with variable degree of success. Human cadaveric bladder acellular matrix has been tested in few clinical trials. Four patients with a history of failed hypospadias surgery had neourethra created by anastomosing the cadaveric bladder matrix in an onlay fashion to the urethral plate. The created neourethra size ranged from 5 to 15 cm. Three of the four patients had a successful outcome both functionally and cosmetically. One patient with 15-cm neourethra created developed small fistula [29]. As for adults, in a randomized comparative study, El-Kassaby et al. concluded that bladder matrix can be reliably used as a graft biomaterial for urethral repair in stricture patients with a healthy urethral bed while buccal mucosa remains the best graft material for patients with 3 or more prior surgeries who usually have unhealthy urethral beds [27]. As cadaveric bladder matrix cannot dependable source of off the shelf material for urethroplasty, commercial porcine bladder matrix was developed. In one of most recent clinical studies using this matrix in 2-stage repair, 8 patients were involved with the length of repair segment ranged from 2.5 to 15 cm. In 7 patients, the acellular graft was well taken and urethra of 4 patients remained patent without the need for subsequent dilation [30].
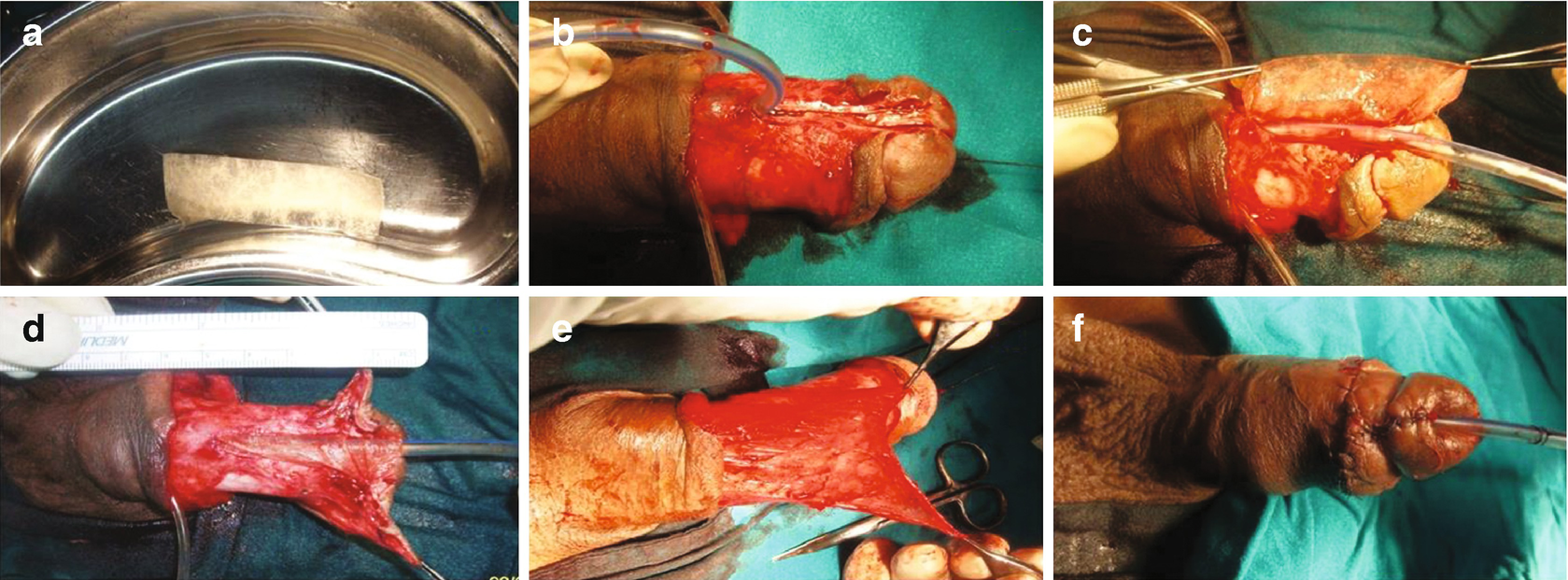
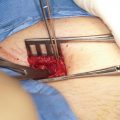
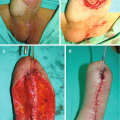
showing brief steps of hypospadias repair with SIS graft. (a) Penile skin degloving through a subcoronal incision preserving the urethral plate. (b) and (c) Suturing of small intestinal submucosa graft to the urethral plate in an onlay fashion. (d) Mobilization of dorsal penile dartos flap. (e) Splitting of the dartos flap into two equal halves to form second layers coverage for the graft. (f) Final postoperative appearance

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree