5 Endoscopic approaches to the breast
Synopsis
 Applications for surgical endoscopes have been developed in both cosmetic and reconstructive breast surgery.
Applications for surgical endoscopes have been developed in both cosmetic and reconstructive breast surgery.
 As in other surgical applications, surgical endoscopes allow for the use of distant, more cosmetically acceptable incisions in performing cosmetic and reconstructive breast surgery.
As in other surgical applications, surgical endoscopes allow for the use of distant, more cosmetically acceptable incisions in performing cosmetic and reconstructive breast surgery.
 Use of surgical endoscopes in cosmetic breast surgery allow for dissection of an implant pocket from a remote incision, typically located in the axilla.
Use of surgical endoscopes in cosmetic breast surgery allow for dissection of an implant pocket from a remote incision, typically located in the axilla.
 In reconstructive breast surgery, the endoscope allows for a skin sparing technique in the harvest of the latissimus dorsi muscle for partial breast reconstruction.
In reconstructive breast surgery, the endoscope allows for a skin sparing technique in the harvest of the latissimus dorsi muscle for partial breast reconstruction.
 Techniques and approaches utilizing surgical endoscopy to optimize visualization while minimizing scarring continue to evolve.
Techniques and approaches utilizing surgical endoscopy to optimize visualization while minimizing scarring continue to evolve.
Introduction
Endoscopic approaches to conventional surgical problems have significantly enhanced treatment options since their introduction in the latter half of the 20th century (Berger 1996; Paige 1997).6,15 Less traumatic tissue dissection in conjunction with smaller surgical incisions have enabled many patients to benefit from reduced postoperative pain, expedited recovery, and improved cosmesis (Cho 1997).8 The surgical endoscope has been used by the senior author to perform over 200 endoscopic augmentation mammaplasties and over 50 endoscopic lumpectomy reconstructions. Unlike intra-abdominal or thoracic applications, plastic surgery frequently involves extensive soft tissue and neurovascular dissection within enclosed potential spaces. Limited surgical apertures and confined optical cavities have therefore inhibited the development and widespread usage of minimally invasive plastic surgical techniques. Widespread availability of endoscopic equipment and refinements in technique have improved the relevance and utilization of endoscopic approaches in a wide variety of plastic surgical applications in recent years. As endoscopic approaches to the breast and other areas of plastic surgery have gained acceptance, it is important to have a fundamental understanding of the basic concepts of surgical endoscopy. These include the principle of the optical cavity, support systems, illumination equipment, imaging technology, incision planning, and some basic technical considerations.
History
Use of the surgical endoscope in cosmetic breast surgery
In the early 1990s, there was a surge in interest in endoscopic plastic surgery, primarily as an extension of the success of endoscopic cholecystectomy in general surgery. This expanded to application of the endoscope to breast surgery. The Emory group reported their experience with endoscopic breast augmentation through an axillary incision in 1993 using a specialized retractor and an air-filled optical cavity. Ho reported a technique that used glycine irrigation to create a liquid-filled optical cavity, although he now also uses a specialized retractor and an air-filled optical cavity.10 The increased control resulting from direct visualization of the dissection obviated many of the previous downfalls of the blind axillary approach. Howard demonstrated the benefits of the endoscope with the axillary approach by decreasing the incidence of implant malposition from 8.6% to 2%, when the endoscope was used.11
Use of the surgical endoscope in reconstructive breast surgery
Fine and colleagues were the first to report a clinical experience with endoscopic latissimus dorsi flaps in 1994.20 This harvesting technique utilized smaller incisions and was performed with the use of modified laparoscopic cholecystectomy instruments. Since this publication, the limiting factor for improving endoscopic harvest has been creation of an adequate optical cavity. Innovative ideas to optimize visualization within soft tissue planes have gradually emerged. Several authors have described external retraction with sutures, balloon dilation, CO2 insufflation, and the use of additional ports. We have further refined our technique, namely the endoscopic assisted reconstruction with latissimus dorsi (EARLi) flap. This procedure, first performed by the senior author in 1998, for reconstruction after breast conserving therapy, only requires an axillary incision and is therefore cosmetically appealing. Seminal studies in the late 1990s, documenting the equivalence of BC with mastectomy for the treatment of small breast cancers resulted in many women requesting BCT. Unfortunately, adequate carcinoma excisions, especially excisions for relatively large T2 or T3 breast cancers, may lead to poor cosmetic results. The severity of the aesthetic defect is a direct relation between the size of the tumor resection with adequate margins, and the size of the affected breast. A recent report has shown that BCT combining radiation and immediate myosubcutaneous latissimus dorsi flap reconstruction is an oncologically safe treatment for larger breast cancers. Several authors have described a latissimus mini-flap, a procedure for filling in the breast defect which utilizes an incision running from the apex of the axilla along the lateral border of the breast towards the outer aspect of the inframammary fold. This innovative approach allows both wide local excision in women who would otherwise have required a mastectomy as well as a more cosmetically appealing outcome. At our institution, we perform the EARLi flap, a procedure with even less scarring, to achieve a favorable cosmetic outcome after BCT. The principle goals of the EARLi flap are to replace excised tissue volume and prevent breast deformity following lumpectomy. In addition, breast size and contour are maintained, and scar tissue contracture is minimized. Because of the small incision and limited soft tissue dissection, postoperative pain is reduced and recovery time is diminished.
Endoscopic augmentation mammaplasty
Treatment/surgical technique
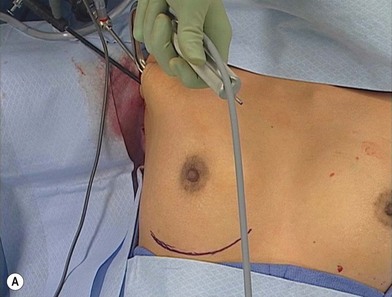
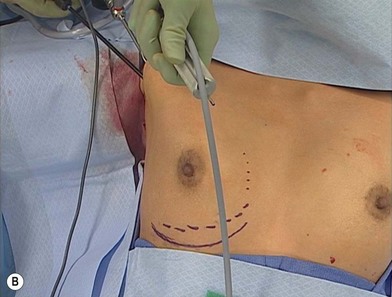
Preoperative considerations include accurate marking of the native and proposed placement of the inframammary crease, as well as anticipated areas of release of the pectoralis major muscle. The pectoralis muscle should be completely divided along its inferior origin from the rectus fascia (Fig. 5.1). This complete myotomy is transitioned gradually to a partial thickness release as the dissection approaches the medial origins along the sternal border until the level of the nipple is reached. Mark the first axillary crease with an incision behind the anterior axillary line. The incision should measure 3 cm if a saline implant is planned, 4.5–5 cm if silicone is to be used. If concealed in a natural skin crease within the hair bearing portion of the axilla, the incision will typically be extremely favorable if not disappear almost completely when fully mature (Fig. 5.2).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree