Introduction
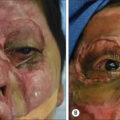
The external ear is a fixed, protruding appendage susceptible to trauma because of its prominent position on the lateral aspect of the head. Whereas recent studies report approximately 25% of facial burns also result in ear burns, isolated ear burns are extremely rare. , In burn patients with major injuries, the burned ear is typically relegated to a secondary concern, as priority is initially given to managing conditions and functions impacting the patient’s survival, with airway maintenance, fluid resuscitation, possible escharotomy, and early burn excision and coverage taking precedence. Once a patient’s more life-threatening issues have been adequately addressed, ear reconstruction can be pursued.
Ultimately, postburn ear deformities and any resulting asymmetry and sensory deficits can profoundly impair a patient’s long-term psychological well-being, social functioning, productivity, and engagement with their physical environment. Hairstyle choice and the ability to wear glasses are just two fundamental aspects of life that exemplify this. Meticulous planning for ear reconstruction, in coordination with the patient, is critical to mitigating these adverse outcomes and improving a patient’s quality of life.
Anatomy
A thorough understanding of ear morphology and anatomy and their variations is required for the surgeon tasked with restoring the dimensions and contours of a deformed ear. The auricle has a notoriously complex morphology created by a framework of elastic cartilage and its envelope of delicate skin. Reconstructing a burned ear as closely as possible to its native architecture presents a formidable challenge even to the most experienced surgeon.
The dimensions, shapes, and positions of the ear vary considerably from one individual to another, as well as the thickness, pigmentation, texture, and elasticity of its skin. Tanzer described the ear’s anatomy in four planes (the concha floor, conchal wall, scaphaantihelix, and helix) positioned at right angles. These four anatomical planes are helpful to remember when developing a surgical plan. However, the helical rim is arguably the most critical component of the ear’s silhouette; thus its reconstruction is vital to achieving an aesthetically acceptable outcome. In the normal population, the auriculocephalic angle, measured from mastoid skin to the posterior surface of the auricle, is 15 to 20 degrees. Although the ear achieves roughly 90% of its adult size by age 3, its dimensions are not fixed in adults but tend to increase in size as one ages, and the effects of gravity take their toll. This phenomenon is evident in the ear lobes of the elderly. The ear’s cartilage typically begins to harden by age 6, and by age 12, it has attained its adult size. The average ear length ranges from approximately 55 mm to 65 mm, with an average length of 62.4 mm in males and 58.4 mm in females. Ear width is about 55% of its length, with a reported average of 35.5 mm in males and 33.4 mm in females.
The auricle’s cartilage is supported by a delicate array of muscles and ligaments that help fixate it to the deep structures of the temporal bone. Its projection from the head defines the anterior and posterior aspects of the ear. At the same time, its variable thickness establishes the characteristic shape and contour of the ear except for the lobule, which hangs from the inferior segment of the cartilaginous skeleton. As the ear’s cartilage is avascular, a thin layer of perichondrium enveloping its anterior and posterior aspects and tightly adherent to the helix and along the ear’s folds provides nutritional sustenance. In cases where cartilage is removed as part of a graft, the perichondrium must be preserved and placed near a supporting vascular bed. ,
On the anterior surface of the cartilaginous skeleton is very thin skin separated by perichondrium and an incredibly attenuated subcutaneous layer of fat. The skin on the ear’s posterior aspect is thicker and more mobile than the anterior skin, attributed to two fat layers, with the deeper lamellar layer sliding smoothly over the cartilage. As with skin elsewhere in the body, auricular skin guards against thermal injury, trauma, and infection to the deeper tissue layers and enables thermoregulation, sensory perception, and immunologic protection.
Three branches of the external carotid artery (the posterior auricular, the superficial temporal, and the occipital arteries) supply the superficial and deep vascularization to the ear and adjacent tissues. The cartilage not only separates the anterior and posterior aspects of the ear but also serves as a division of its vasculature. The posterior auricular artery is the dominant blood supply to the external ear. After originating from the external carotid artery, it courses along the posterior auricular branch of the facial nerve before dividing into five branches. Of the five, the auricular branch vascularizes the medial surface of the ear, and the extrinsic auricular muscles and the occipital branch supply the postauricular skin and occipital belly of the occipitofrontalis muscle. The superficial temporal artery supplies the upper-half of the ear and the temporal and preauricular areas. The artery has three small branches (the superior, middle, and inferior anterior auricular arteries) that perfuse the anterior part of the lateral auricular surface, including the lobule and part of the external meatus. The superior and the inferior anterior auricular arteries form the helical rim arcade with branches of the posterior auricular artery. The deep auricular branch of the maxillary artery, which comes off the external carotid artery, perfuses the deep aspect of the external auditory canal and the tympanic membrane. ,
The veins of the ear correspond to its arteries, and venous flow leads into the external jugular vein via the posterior auricular vein. The superficial temporal and retromandibular veins provide outflow to the anterior and superoposterior segments of the ear.
The ear has six intrinsic and three extrinsic muscles. The intrinsic muscles, located within the ear and considered vestigial structures, help maintain the ear’s shape. The three extrinsic muscles (the anterior auricular, superior auricular, and posterior auricular muscles) anchor the ear to the zygomatic arch, the deep layers of the temporal fascia, and the mastoid, respectively, via a strong tendon the muscles form on the posterior aspect of the conchal cartilage. ,
The temporal branch of the facial nerve and the posterior auricular branch of the facial nerve provide motor innervation to the intrinsic and extrinsic muscles. The auriculotemporal branch of the mandibular nerve and the cervical plexus innervate the anterosuperior and anterolateral surfaces of the ear. In contrast, the concha is innervated by the auricular branch of the vagus nerve. The lesser occipital nerve innervates the posterosuperior aspect of the ear, and the great auricular nerve innervates the anteroinferior and posteroinferior aspects. The external auditory canal is innervated by the great auricular nerve on the anterior and superior walls and the auriculotemporalis nerve on the posterior wall. , ,
Pathophysiology
The ear’s unique anatomy, particularly its thin skin and avascular cartilage, makes it especially vulnerable to burn injury and subsequent deformation. The ear’s anterior surface, with its unprotected skin, is more vulnerable to burn injury than the shielded posterior aspect. The helical rim gains modest protection from a somewhat thicker skin. However, it is the most frequently involved ear component in burn injury because of its projection.
Postburn edema can lead to vascular obstruction and compromised perfusion, denying tissues vital nutrition and oxygenation. Swelling can potentially cause full-thickness skin and perichondrium loss, exposing the underlying cartilage and risking the onset of chondritis, which can occur in up to 25% of patients, and avascular necrosis. Such adverse outcomes motivate an aggressive approach to early identification and management of postburn ear injuries.
Classification of the burned ear
Various classification systems for acquired ear deformities have been proposed. They can potentially be helpful when assessing postburn ear deformities and developing a surgical plan—K’ung et al. classified postburn ear deformities into three tiers: mild, moderate, and severe. Salisbury and Bevin categorized them as either contracted, partial, or complete loss of the helical rim or total loss of the ear. Brent proposed classifying deformities according to loss of skin, cartilage, or full-thickness loss of ear tissue. Full-thickness losses can be further divided into upper-third, middle-third, and lower-third deformities, partial and total loss, and loss of the lobule. Luo et al. categorized posttraumatic ear deformities according to involved tissue components, size of defects, and status of surrounding soft tissues: type I is a severely cicatricial ear without cartilage defect; type II includes partial full-thickness defects; type III is near-total or total ear loss, periauricular skin intact; type IV is near-total or total ear loss, periauricular skin involved; and type V is near-total or total ear loss of healthy surrounding skin and soft tissue resulting in unavailability of temporoparietal fascia (TPF). Kumar et al. proposed a system in which the ear is divided into four equal zones, each separated by a 45-degree angle. Within each zone, defects are described by briefly listing lost anatomical features while characterizing surrounding skin.
Early management
Burns to the ear can potentially involve the cartilage framework, risking subsequent chondritis, necrosis, hypertrophic scars, and complications caused by infection. Injury can result in variable deformities and, at worst, total ear destruction, either directly from burn trauma or from chondritis.
One of the primary objectives in burn care of the ear, as with burns to any part of the body, is tissue preservation. This fundamental principle should guide both early conservative management and later reconstruction. In light of the ear’s location, thin skin and subcutaneous tissue, and absent intrinsic blood supply to the cartilage, management strategies are required that often diverge from the traditional approaches used for facial burns.
Before any reconstruction, one must make a meticulous assessment of the burned ear to identify tissues that are not irreparably damaged. The zones of injury typically form a heterogenous mix of central areas of coagulation and peripheral areas of stasis and hyperemia, features that help identify burn depth. Dry, nonsuppurative eschar should be left in place as it serves as a biologic dressing that protects the underlying cartilage from desiccation. Tissues with unclear viability and exposed cartilage should also be left undisturbed. Initial steps should include shaving the hair around the ear, which helps with dressing changes, local care with frequent washings, topical antimicrobial creams, and soft, supportive head dressings. Although early debridement and grafting is a central tenet to burn care, in the burned ear this tactic can cause undue deformity. A delay in surgical intervention confers various advantages, most importantly preserving tissue that may be needed to create a healthy and stable construct.
Edema
As previously mentioned, edema typically develops acutely in the burned ear and can subsequently impinge on the arterial vasculature, causing ischemia. Necrosis can set in, leading to exposed cartilage, possible chondritis, and significant deformity or even total ear loss. Protective padding, discussed in a forthcoming section, can help prevent inadvertent pressure of the ear against the skull and worsening edema. , Once edema has fully abated, partial-thickness burns can be expected to granulate and epithelialize. Skin grafts can be placed on preserved perichondrium, whereas exposed cartilage devoid of perichondrium will not heal and thus requires debridement and reconstruction. ,
Chondritis
Chondritis is a dreaded and disfiguring complication that presents 3 to 5 weeks after burn injury when the ear’s cartilage becomes infected. Its insidious onset is independent of the initial burn depth but is usually associated with exposed cartilage, although this is not essential. In their study, Engrav et al. reported that chondritis developed primarily from minor lesions in which pressure prevention and topical silver sulfadiazine had been discontinued before total healing or were never initiated. Once injured or exposed, cartilage becomes a nidus for infection. Although chondritis occurs most often in the helix and antihelix, it can involve the entire ear, including cartilage unaffected by the initial burn. Typically, it first presents as a dull pain that rapidly becomes strikingly tender, especially when pressure is applied. Edema, fluctuance, fever, and erythema are also evident. Although fluctuant areas may spontaneously drain and symptoms and fever improve, this can indicate that the infected cartilage has become necrotic. Pseudomonas aeruginosa is the causative bacteria in 80% to 95% of cases, with Staphylococcus aureus present in ∼50% of wound cultures. Other species include Candida albicans, Providencia rettgeri, Proteus mirabilis, Enterobacter cloacae, Corynebacterium diphtheriae, Klebsiella pneumoniae, and Escherichia coli. , , ,
Chondritis tends to recur and can manifest even after reepithelialization has taken place. However, once mafenide acetate cream (Sulfamylon, Viatris, Canonsburg, PA) became integral to burn care, the rate of chondritis has dropped to less than 5%. This topical synthetic sulfonamide is a critical prophylactic treatment as it is especially effective at penetrating eschar and controlling the local bacterial count. Systemic antibiotics are not indicated as they cannot penetrate the cartilage and attain therapeutic levels because of their avascularity. The best strategy for preventing chondritis is diligent avoidance of pressure to the ear and daily gentle washes with antibacterial soap. , , Early, frequent, and accurate ear assessment ensures prompt diagnosis of chondritis and timely treatment. Iontophoresis, described in a later section, is still employed by some burn centers to enhance the delivery of antibiotics to infected cartilage. However, it has largely fallen into disfavor.
Chondritis treatment typically entails debridement of infected tissue to prevent loss of all cartilage. One effective surgical technique, a bivalve-type incision, traditionally divides the helical rim along the subperichondrial plane. As serial debridements are usually necessary, significant deformity is often unavoidable. , , ,
Ear protection
Whereas the uninjured ear can maintain its shape, it largely cannot tolerate compressive forces. In its compromised state, the burned ear risks developing further injury, chondritis, necrosis, and deformity from excessive pressure. Intubated patients present an additional challenge, as it is not uncommon for the straps used to secure the nasal or endotracheal tube to impinge on the ear. As such, a crucial part of comprehensive burn care is eliminating pressure on the ear. Various cost-effective techniques have been described. Jordan et al. employed a headgear device with bilateral ear protection that prevented pressure necrosis in patients, all intubated, who were included in their study. Harries et al. created an ear protector constructed from foam and held in place with the tubular elastic netting Surgifix (FRA Production SpA, Dusino San Michele, Italy). These effective solutions permit normal burn wound care, visualization of the ears, and sleeping in the decubitus position.
Topical therapy
As most ear burns heal without excision and grafting, they are initially managed conservatively with local wound care, including daily washes and frequent application of topical antimicrobial agents. Although there is no consensus on the optimal topical agent for burn wounds to the ear, Sulfamylon is commonly used, whereas other burn wounds often benefit from bacitracin or Silvadene (Pfizer, New York City, NY). Sulfamylon can penetrate burn eschar and cartilage, providing broad bacteriostatic activity against numerous gram-negative and gram-positive organisms, including P. aeruginosa and several anaerobic bacterial strains. It is typically applied in a thick layer three to four times per day, helping to prevent cartilage desiccation and possible chondritis. , ,
Iontophoresis
Iontophoresis, first described by LeDuc in 1908, facilitates the delivery of chemicals across intact skin. For the burned ear, the technique uses polar-charged antibiotic agents, such as penicillin and gentamicin, pulsed across avascular tissue membranes using low-amperage direct current. This approach establishes a bactericidal level of antibiotics in cartilage, unlike conventional intravenous antibiotics. Although iontophoresis was once more commonly used as a treatment for chondritis, these days most burn centers do not employ it, primarily because of the efficacy of Sulfamylon. In a clinical and animal study by Desai et al., no differences in the incidence of chondritis were observed in patients who received gentamicin-based iontophoresis versus routine care only.
Early reconstruction
Successful ear reconstruction requires a systematic approach to developing a treatment. A careful examination of any postburn deformity should take note of its location, size, depth, subunits affected, and tissue quality and availability. This methodology takes heed of Gillies’s first principle of plastic surgery: “Observation is the basis of surgical diagnosis.” , Given the broad spectrum of wounds and deformities that can develop in the burned ear, it is not surprising that the management options are equally extensive. Regardless of the surgical technique ultimately employed, the following conditions, first articulated by Suraci in 1944 and still relevant today, should be considered when reconstructing the ear: (1) correct size, (2) identical appearance to the contralateral ear, (3) identical auriculocephalic angles, (4) identical ear levels, (5) durability of the construct to maintain its size and shape, (6) adequate selection of supporting and soft tissue for precise molding, and (7) appropriate color match to that of the contralateral ear. , Although the surgical approaches available today, refined over many decades, aim to achieve these conditions, many cases, especially those involving near-total or total ear deformity, rarely result in a genuinely native-appearing reconstructed ear.
Although the timing of surgery is variable, most initial surgeries for ear burns take place 1 to 3 weeks after injury. Partial-thickness burns to the ear often reepithelialize and heal without complication when expectant management, local wound care, and topical therapy are pursued while the edema resolves. If the underlying perichondrium is intact and viable and not infected, the wound can reepithelialize or, if needed, close with a skin graft. In cases where healing is not evident approximately 3 weeks after injury, surgical intervention is likely required. ,
Deep partial-thickness burns are at greater risk for more permanent injury and disfiguration, especially when edema and tissue loss are present. As previously stated, when perichondrium has been compromised, chondritis can develop, leading to significant tissue loss and deformity. In cases in which deep partial-thickness burns have not healed several weeks after injury, surgical intervention is required. After meticulous tangential excision down to the perichondrium, with care to minimize unnecessary cartilage exposure, the wound can be covered with skin grafts. To achieve optimal color and texture matching, the contralateral pre- or postauricular skin is ideal donor tissue. However, it is best for small burn wound coverage when availability is limited. When donor skin from around the contralateral ear is also injured, precluding harvest, thick split-thickness grafts can be taken from the scalp or medial upper arm or full-thickness grafts from the supraclavicular area or groin.
Full-thickness burns often result in exposure or injury to the underlying cartilage and always necessitate debridement and reconstruction to prevent further tissue loss and deformation. As always, topical antibiotics and preventing undue pressure to the ear are initiated during the acute phase of care. When the perichondrium has been damaged, vascular coverage is needed to save the cartilage, as skin grafts will not suffice. The size of the defect, the presence of infection, and the availability of viable surrounding tissue dictate the operative approach. For small full-thickness burns, any exposed nonviable cartilage should be lightly debrided or segmentally resected to prevent further tissue loss. Segmental resection of cartilage with reapproximation of the free cartilage edges typically does not markedly affect the overall aesthetics of the ear. The defect can then be covered using a local perichondrocutaneous flap.
Rosenthal described auricular zones of tissue preservation and sources of local cutaneous flaps. His approach includes eleven zones of the partially burned ear divided into three major areas. The first area consists of six zones that can be used as donor sites for advancement flaps (preauricular, cavum conchae, cymba conchae, upper antihelix and scapha, posterior helix and conchal skin, and mastoid skin). The second area consists of three zones that can be excised and closed without considerable disfiguration (lateral helical rim, termination of the helix and lobule, and inferior crus and antitragus). The third consists of two zones that warrant preservation whenever possible (ascending root of the helix to the middescending helix, superior crus and a significant portion of the antihelical convexity). ,
In cases in which tissues immediately surrounding a burn wound to the ear have also been injured, precluding their use for coverage of viable cartilage, regional or distant flaps can be leveraged. One workhorse regional flap is the temporoparietal fascia (TPF) flap, although severe concurrent burns to the scalp may preclude its use. The TPF flap is a thin, pliable layer of fascia extending from the subcutaneous musculoaponeurotic system inferiorly and the galea aponeurotica superiorly. It is well vascularized by the posterior branches of the superficial temporal artery and venae, allowing it to nourish both the cartilage to which it is applied and the overlying split-thickness skin graft that is subsequently placed. , , One must weigh the decision to use the TPF flap for primary coverage as doing so eliminates arguably the best source of vascularized tissue for secondary reconstruction.
Secondary reconstruction
Subtotal ear reconstruction
Postburn ear deformities result from scar contractures, partial loss of the helical rim and other auricular structures, and total ear loss. , Reconstruction addresses three components: contractures, framework defects, and cutaneous coverage. Extrinsic contractures, namely contractures of soft tissues surrounding the ear, can produce tension vectors that distort the ear’s morphology. These are distinct from intrinsic contractures found within the ear, and they also result in deformation. Both contractures can often be released using Z-plasties and V-Y advancement flaps, with skin grafting sometimes required. Typically, extrinsic contractures should be addressed before or concurrently with intrinsic contractures. Treatment options for hypertrophic scars and keloids are discussed in more detail in a later section.
Local flaps
Burn injuries to the ear resulting in obliterated tissue and localized deformity typically require more than simple debridement excision and skin grafting. In these cases, reconstruction benefits from techniques used in managing congenital and other acquired ear deformities. The final overall dimensions of the reconstructed ear should attempt to match as closely as possible the uninjured contralateral ear if present; however, the perfect symmetry of the ear’s intricate topology is not crucial. The observer typically only focuses on one ear at a time, and when both ears are in view, only their relative length, position, and protrusion are noticed.
Both preauricular and postauricular skin provide excellent sources for pedicled skin flaps. The postauricular area is often protected from burn injury, and its skin is closely matched in thickness and color to ear skin. The postauricular flap requires two stages, the first for inset and the second for release. Suppose coverage of larger areas, including the entire ear, is needed more than the available postauricular skin allows. In that case, a tissue expander can be placed under the retroauricular skin before flap mobilization, creating a three-stage procedure (expander insertion, flap inset, flap release). In some cases, involving focal defects (e.g., the helical rim), the flap may provide sufficient bulk to eliminate the need for a cartilage graft. Regarding flap creation, it should be developed down to postauricular fascia with its base, made as wide as possible, and positioned at the hairline. The flap’s width should match the defect’s dimension. Although creating a tubed postauricular flap is an option for helical or lobule reconstruction, this approach may be more likely to result in flap loss. The flap is typically divided at its base approximately 2 weeks after placement. The resulting donor area can be either covered with a skin graft or with skin undermined and then advanced to the postauricular sulcus. If increased ear projection is desired, a secondary procedure can be done to deepen the postauricular sulcus. , ,
For defects involving the lower third of the helical rim and the lateral lobule, an inferiorly based preauricular skin flap can be considered. If the flap is of adequate length, it can create both the lateral and medial aspects of the lobe. Alternatively, the medial lobe can be covered with a skin graft during the second-stage release of the flap. A cartilage graft, frequently required to mitigate flap contraction and distortion, can be harvested from the ipsilateral ear if injured or the contralateral ear if traumatized.
In cases involving small to medium cartilage defects of the helix or antihelix, the conchal transposition flap is an excellent and reliable option ( Fig. 60.1 ). This technique, first described by Davis in 1974 and later modified by Donelan, leverages conchal tissues spared from injury. To create the flap, uninjured conchal cartilage and its overlying skin are harvested and transposed superiorly on a narrow pedicle based at the crus helicis. The donor site is then covered with a split-thickness skin graft. This approach confers multiple advantages, including blending with surrounding tissues using the ear’s skin and cartilage, low morbidity and complication rates, and high patient satisfaction. ,
Conchal transposition flap. (A and B) An 11-year-old male patient from Haiti sustained an 11% total body surface area campfire flame burn injury to the head, neck, and face. Six years after injury, the patient presented with a subtotal cartilage deficit in the superior aspect of the right helical rim. (C and D) A conchal transposition flap was carried out in which the conchal bowl was incised along its full length, leaving a superior-anterior pedicle. The conchal cartilage was rotated into position and covered with a retroauricular skin flap. (E and F) A sagittal and oblique view of the reconstructive ear 3 months after a release and full-thickness skin grafting to the retroauricular space. The release and skin grafting were performed 1 year after the conchal transposition flap to elevate the helical rim and further improve ear protrusion.
Another option for localized defects of the helix and scapha up to 3 cm is the Antia-Buch chondrocutaneous advancement flap. In this single-stage procedure described in 1967, the helical rim is bisected into an anterior skin cartilage flap and a posterior skin flap after making an incision along the helical sulcus. The posterior auricular skin is elevated down to the posterior sulcus of the ear. An incision at the helical root allows an advancement flap of helical skin and cartilage from the root of the helix to fill the defect. For additional length, a V-Y advancement flap of the helical crus can be made. The primary disadvantage of the technique is that it results in a smaller ear and, if used for too large a defect, can lead to cupping of the ear. The modified Antia-Buch flap described by Noel et al. is designed to avoid the diminution of the auricle. This technique adds a 2- to 3-cm vertical incision in the temporal scalp to the original approach to facilitate defect closure. ,
When focal helical thinning is evident, a V-Y flap of the helical rim can be employed. Folding the flap onto itself establishes bulk along the rim. In cases involving extended areas of thinning along the helical rim, a tubed pedicle flap from the postauricular area or neck can be used.
Composite grafts
Burns that result in small- to medium-sized cartilage deformities can benefit from conchal cartilage composite grafts, which provide an excellent match with the remnant auricle ( Fig. 60.2 ). They are harvested from either the anterior or posterior aspects of the ipsilateral or contralateral ear, depending on tissue viability, and donor sites are then closed with split-thickness skin grafts.
Conchal cartilage graft. (A) A 1-year-old female patient from the Dominican Republic sustained a 50% total body surface area flame burn injury from a house fire. The patient presented 13-years postburn with a subtotal right ear cartilage deformity approximately 4 cm from the middle to the superior aspect of the helical rim. (B) For stage 1 of helix reconstruction, the patient underwent a retroauricular skin flap, and a composite conchal cartilage graft was harvested from the contralateral ear. (C) Stage 2 was completed 4 months later with a full-thickness skin graft placed in the retroauricular sulcus to elevate the composite graft. (D) A sagittal view of the reconstructive outcome 3 years after stage 2.
Tissue expansion
When tissues surrounding the ear are insufficient to permit needed skin coverage of the ear’s framework, tissue expansion is a valuable part of the surgeon’s armamentarium. In many cases, it obviates the need for more distally based flaps. Whereas tissue expansion is frequently used in microtia and other congenital ear deformities, its application to postburn ear deformities requires careful selection, given the variable quality of tissues and the potential for significant complications. Heavily scarred skin is not well suited for expansion and, when present, can lead to extrusion of the expander.
Tissue expansion is often used to enlarge retroauricular skin for enhanced coverage of exposed helical cartilage or cartilage grafts or to cover the postauricular sulcus when the ear is elevated. The incision for the expansion should be placed along the hairline, permitting suitable access to elevate the skin and subcutaneous fat. The same incision can be used to position the injection dome, either superiorly into the temporal area or inferiorly into the occiput. Different size and shape expanders are available with appropriate volumes typically less than 100 mL, with an 80-mL expander or two 50-mL expanders recommended in large or total ear burn cases to obtain sufficient skin. Care must be taken to avoid kinking or coiling of tubing to the expander’s remote valve. Inflation of the expander can begin as early as 7 days after its insertion, with serial injection of 5 to 8 mL of saline at each session, which usually occurs weekly but may be more frequent depending on the case. The targeted size can take up to 4 months, a slower process than in patients with congenital microtia and normal skin. When the expander is removed, one must decide whether to remove or leave the capsule in place. A thick capsule risks poor pliability and a softening of the contours of the underlying framework. However, its removal may compromise the flap’s blood supply and produce a thinned skin cover that produces a skeletonized appearance.
Total ear reconstruction
Taking on the reconstruction of an ear that is mostly or entirely lost can be a daunting task that mandates scrupulous planning. The accuracy of the scaffold and the availability of soft tissue for encasement largely determines a successful outcome. These cases often involve periauricular tissues that have been burned and lack elastic and well-vascularized tissue required for framework coverage. In such circumstances, the adjacent TPF flap is frequently uninjured and available. The contralateral free TPF flap or a free radial forearm fascia flap can also be used when the ipsilateral TPF is compromised.
An autologous or synthetic scaffold can recapitulate the complex underlying architecture of the ear. Both approaches have their attendant advantages and shortcomings, highlighted later. We have not yet achieved the optimal framework, which should be both thin and elastic but sufficiently resilient to resist the contractile forces during wound healing and pressure imparted on the ear during daily activities. Highly realistic prosthetic ears with osseointegration are another option favored by some patients.
Before proceeding with reconstruction, a careful assessment of the unaffected ear, if present, should be carried out. If both ears are absent or severely disfigured, one must locate the external auditory meatus and any auricular remnants to ascertain the optimal position for the reconstructed ears. The ear should be oriented approximately 15 to 20 degrees more vertically than the axis of the nose, with the superior helix aligned with the tail of the eyebrow. The auriculocephalic angle should be about 20 degrees, with the ear protruding 1 to 2 cm from the mastoid. , ,
Regardless of the technique employed, reconstruction should be considered only after the relevant soft tissues are in their chronic phase of healing.
Autologous scaffolds
Total ear reconstruction for cases of microtia using autologous cartilage was first described by Tanzer and popularized by Brent. , The techniques and strategies they employed have been successfully applied to burn cases ( Fig. 60.3 ). To fabricate a scaffold, cartilage is typically harvested from the confluence of ribs 6 to 8, usually from the contralateral side to leverage advantageous curves in the cartilage, and then carved into a detailed framework. In unilateral burns, the uninjured ear is the basis of a template created from clear x-ray film. In cases in which both ears are absent, the aforementioned anthropometric characteristics serve to guide framework creation. Given the limitations of two-dimensional templates, 3D scanning and printing technologies have recently been employed to create bespoke templates and teach framework carving. ,
Costal cartilage graft. (A) This male patient sustained a 30% total body surface area flame burn injury at the age of 1 year from a house fire in his home country, Libya. At 10-years postburn, the patient presented with a dual problem of severe scalp alopecia and complete loss of the right ear. The external auditory meatus was present, but no cartilage remnant was visible, and the overlying skin, which had been previously grafted, was of poor quality. (B) After 5 months of right temporal-parietal-occipital tissue expansion, the patient underwent resection of burn alopecia with hairline reconstruction and placement of a full construct costal cartilage graft. A significant portion of rib and synchondrosis was harvested and then utilized to sculpt a costal cartilage graft, guided by a transparent film template of the contralateral ear. (C–E) The construct was wrapped in an elevated temporoparietal fascia flap, sutured into position, and covered with a split-thickness skin graft. (F) A sagittal view of the reconstructive outcome 1 year after costal cartilage graft placement. During this interval, the patient underwent a Y-to-V advancement flap to address significant external auditory meatus contracture. A full-thickness skin graft was applied to the retroauricular sulcus for construct elevation.
Nagata described his two-stage approach using costal cartilage obtained from ribs 6 to 9 and, as with Brent, recreated the components of the ear from separate pieces of cartilage. After fabricating the scaffold, it is inserted into a skin pocket made in the auricular region that will nourish and protect the cartilage. In adults, where cartilage is often calcified, Brent reported excellent outcomes carving a scaffold from a solid block of costal cartilage. However, a separate piece can be sutured to the block if necessary to ensure adequate helical projection. Elevation of the neoauricle to create an auriculocephalic sulcus and improve projection can be done 3 to 6 months after graft placement using fascia (e.g., TPF) and an overlying skin graft to cover the postauricular defect. Projection can be enhanced with a crescent-shaped graft of cartilage banked during the first stage.
Given that costochondral cartilage has not yet achieved the necessary volume and stiffness to be used as a construct until a patient is around age 6, in pediatric patients we recommend waiting until then before considering an autologous cartilage graft. As a child’s ear has already attained 90% of its adult size by age 3, cartilage scaffolds in children can be fabricated in adult dimensions.
Costal cartilage grafts are resilient, have a low complication rate, and can produce good aesthetic results in skilled hands. Several key drawbacks deserve mention. Donor-site morbidity includes pain, infection, chest wall deformity, and pneumothorax (a rarely reported complication). Additionally, the graft, already less pliable than native ear cartilage, can calcify over time, leading to hardening and risk of fracture or extrusion. Warping, fracture, and resorption can also lead to visible changes in the reconstructed ear, necessitating additional corrective procedures.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree