The history of dermabrasion dates back to 1550 BC when Egyptian physicians used alabaster and pumice to smooth scars and skin blemishes.1 They recognized that producing partial-thickness abrasion improved the appearance of skin. Modern era dermabrasion was pioneered in 1905 by Kromayer.2 In 1953, Kurtin refined the dermabrasion technique with modified, powered dental equipment using topical refrigerants and a wire brush.3
There are three main modalities for skin resurfacing: chemical peel, laser resurfacing, and mechanical dermabrasion. The contemporary trend favors the use of the laser-resurfacing technique. Many clinicians feel that laser resurfacing allows for more controlled thermal injury and more consistent outcomes with less operator-dependent variability. Whereas the newest laser technology has significantly reduced risk associated with thermal injury to surrounding tissues, in experienced hands, dermabrasion provides great manual control to the depth of destruction without spreading the thermal injury to deeper layers of the skin.
29.2 Mechanism
There are five layers of epidermis: stratum corneum at the surface followed by lucidum, granulosum, spinosum, and basalis. The thickness of epidermis varies depending on age of the patient and location of the skin. Average thickness of facial skin epidermis is approximately 100 µm thick, although at the eyelids it may be only 50 µm thick.4 Four major cell types are found in the epidermis, including keratinocytes (80%), melanocytes, Langerhans cells, and Merkel cells. In the basalis layer, melanocytes generate skin pigmentation through the actions of the tyrosinase enzyme.5 The rete ridges are projections of the epidermis into the dermis that help increase the surface area of contact between the two interfaces. In both processes of aging and scar formation, histological changes observed include loss of elastic fiber, atrophy of collagen bundles, and flattening of rete ridges.6,7
The dermis consists of papillary and reticular layers. The papillary layer is located below the basement membrane and contains mostly loose collagen and fibrocytes. The reticular layer contains thicker, compact collagen, and the adnexal structures of the skin such as sweat glands, hair follicles, and sebaceous glands. On the hairless part of the face, such as the nose, the sebaceous unit is the predominant adnexal structure. Within the adnexal structures are the epithelial stem cells capable of differentiation and re-epithelialization.8
The goal of dermabrasion is to mechanically remove the epidermis while preserving the adnexal structures (▶ Fig. 29.1). This is accomplished with the use of a motorized handpiece and a rotating tip composed of either a wire brush or diamond particle-coated fraise (▶ Fig. 29.2). The depth of injury is controlled by the surgeon carefully abrading the skin to the desired layer (▶ Fig. 29.2). Controlled injury to the epidermis and papillary dermis heals without scarring; however, if the injury penetrates deep into the reticular dermis where the adnexal structures and the epithelial stem cell reservoir are located, then complete re-epithelialization is compromised and scar tissue will develop. Because the skin receives its vascular supply via the arborized papillary plexus and the deep vascular plexus at the dermal-subcutaneous tissue junction, pinpoint bleeding serves as an important landmark to indicate the depth of the papillary dermis.9
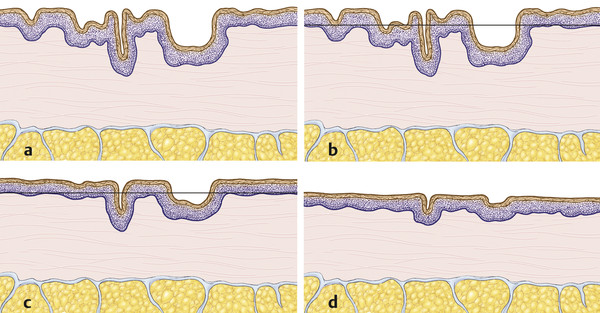
Fig. 29.1 (a) Skin prior to dermabrasion showing multiple imperfections. (b) Dermabrasion is performed to the depth of the papillary dermis. (c) After this has re-epithelialized, a second dermabrasion can be performed (at least 3 months between procedures) in order to achieve further improvement. (d) After this again re-epithelialized, the initial skin imperfections are markedly improved.
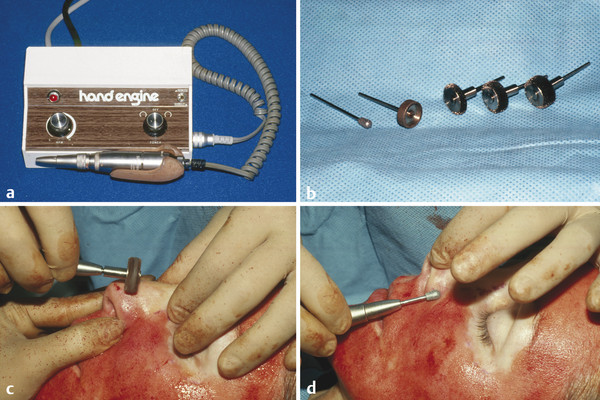
Fig. 29.2 (a) Hand engine and handpiece for dermabrasion. (b) Diamond fraise (left) and wire brushes (right). (c) Wire brush is used to treat broad skin surface. (d) Diamond fraise is used to treat nasal alar groove.
The healing process is initiated by coagulation of the wound edge and crust formation during the first 24 hours. Locally, activation of coagulation cascade triggers complex signaling pathways including the release of chemotactic and growth factors (i.e., transforming growth factor-beta [TGF-β], fibroblast growth factor [FGF], epidermal growth factor [EGF], transforming growth factor-alpha [TGF-α], and platelet-derived growth factor [PDGF]) involved in endothelial cell and fibroblast proliferation. The proliferative phase begins within the first 24 hours and continues for up to 3 weeks. During this phase, re-epithelialization happens at the wound margin by migrating epithelial cells from the adnexal structures in the dermis. Epithelial cells fill the defect of the wound margin and proliferate until apposition of advancing epithelial cells inhibits further migration. At the papillary dermal and subcutaneous layers, fibroblast proliferation is initiated and peaks by day 4 after the treatment. With ongoing fibroplasia in the deeper layer, the epidermis begins to thin and retreats to the superficial plane. During this time, fibroblasts produce collagen, elastin, fibronectin, and glycosaminoglycans.10,11 The maturation phase begins after approximately 3 weeks and continues for up to 18 months after treatment. During this remodeling stage, the collagen is replaced from type III to type I and reorganized from its initial random orientation to a more parallel position relative to the skin surface.9 The matured collagen fibers contract and coalesce with increased diameter and decreased length. Collagen remodeling also triggers the regression of neovascularization, resulting in improvement of the raised, pink scar from early wound to a flatter, white wound after maturation.
Immunohistochemical studies on facial skin following dermabrasion confirm upregulation of procollagen in papillary dermal fibroblasts.12 Harmon et al have shown that dermabrasion of evolving scars modulates the expression level of extracellular glycoprotein and the expression pattern from a localized to a more dispersed form.13 This may promote epithelial cell migration from along the basement membrane and fibroblast movement across the scar boundary to optimize wound healing.
29.3 Indications
Dermabrasion is considered a medium-to-deep skin-resurfacing technique. Whereas numerous indications are described in the literature, the most commonly treated conditions are acne scars, deep facial rhytids, traumatic or iatrogenic scars, actinic damage, and rhinophyma.
29.3.1 Acne Scars
Acne scars that are partial skin thickness respond nicely to dermabrasion treatment. Results may often be improved by a second or third procedure spaced at least 3 months apart. For acne scars that are “ice pick” in nature and extend full thickness, dermabrasion alone is not as effective in improving the appearance of the scar. In these cases, punch grafting is an appropriate therapeutic option. One should perform a punch excision of the deep acne scar, followed by a full-thickness punch graft transferred from a donor site with similar skin color and texture match (▶ Fig. 29.3a, b).14,15 After 4 to 6 weeks, dermabrasion of the grafted site and surrounding area should be performed (▶ Fig. 29.3c–e). In cases with severe acne scars, one may encounter multiple adjacent acne scars that share subcutaneous tracts. If these areas are deeply dermabraded, it may unroof the tracts resulting in a greater surface deformity.
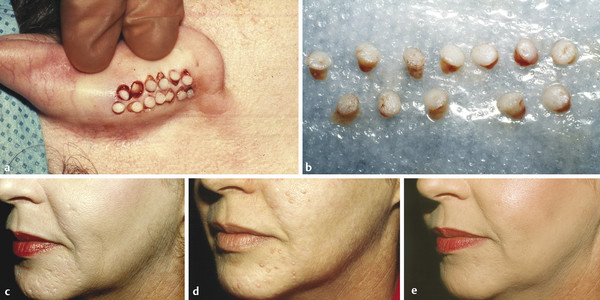
Fig. 29.3 (a) Punch grafts are taken from the postauricular donor site. (b) Full-thickness skin punch grafts. (c) Ice-pick-type acne scars. (d) 3 months after punch grafts were placed. (e) 3 months after dermabrasion performed at the grafted site.
29.3.2 Deep Facial Rhytids
Many aesthetic surgeons consider dermabrasion to be the treatment of choice for deep, vertical rhytids at the perioral and lip regions. Perkins and Castellano describe combining a full-face deep chemical peel with perioral dermabrasion for optimal deep facial rhytids’ rejuvenation.16 Treatment may need to be repeated for recalcitrant deep rhytids with at least a 12-month interval between sessions.
29.3.3 Scar Revision
For elevated scars with persistent induration and textural change, dermabrasion is a very effective treatment (▶ Fig. 29.4 and ▶ Fig. 29.5).17 Dermabrasion is not ideal for wide, depressed scars. Because most scars tend to improve over time, conventional wisdom is to observe for at least 6 months prior to planning the revision treatment. However, if the scar is not showing any evidence of improvement, then dermabrasion may be performed within 6 to 8 weeks after the initial wound repair. Yarborough advocated the early use of dermabrasion within 4 to 8 weeks from the initial injury, when high fibroblastic activity is observed. Earlier than 8 weeks, the wound may lack the appropriate tensile strength to achieve optimal results.8 Brenner and Perro advocated dermabrasion at 6 to 12 weeks after the initial injury, when collagen modeling may be at its peak.18
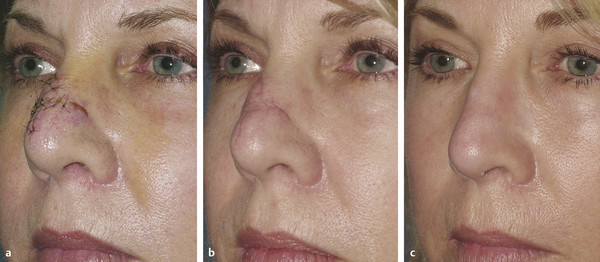
Fig. 29.4 A 48-year-old woman after a dog bite to the nose. (a) 6 days after primary closure at an emergency room. (b) Predermabrasion. (c) 4 years postdermabrasion.
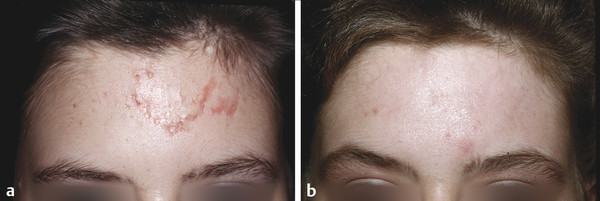
Fig. 29.5 A 27-year-old woman with scar from motor vehicle accident with embedded glass fragments. (a) 2 months from initial injury. (b) 3 months postdermabrasion.
29.3.4 Actinic Damage
The first description of using dermabrasion to treat precancerous skin changes dates back to the 1950s. Numerous studies since then have shown the efficacy of dermabrasion in addressing precancerous lesions.12,19,20,21 In a series of 23 patients with actinic damages who underwent dermabrasion, 79% were cancer-free at 3 years, 64% at 4 years, and 54% at 5 years. These outcomes were superior to results observed from cryosurgery, 5-fluorouracil, or chemical peel as prophylactic treatment against actinic keratosis.22
29.3.5 Rhinophyma
Dermabrasion can be particularly useful in the treatment of rhinophyma (▶ Fig. 29.6 and ▶ Fig. 29.7). Rhinophyma is a manifestation of end-stage rosacea and is characterized by sebaceous hypertrophy and inflammation resulting in multiple abscess and skin thickening.23 This is often a difficult skin condition to manage medically. Surgical treatment is frequently necessary to improve the associated deformity and morbidity. In severe cases, it may be useful to use a scalpel to shave the superficial layers of skin and then complete the process with dermabrasion. When treating a severe case of rhinophyma, controlling the depth of dermabrasion is particularly challenging because one can encounter significant bleeding and sebum exposure. Such an event will obstruct the characteristic features representing the depth of treatment. If dermabrasion is taken too deep, the treated area will heal with a shiny and unnatural surface lacking any sebaceous pores. When utilized properly, dermabrasion can achieve remarkable results that may be difficult to achieve with any other modality in patients with rhinophyma.
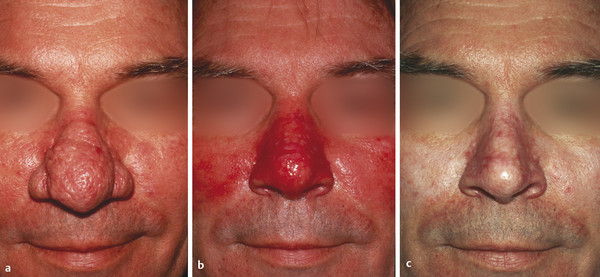
Fig. 29.6 A 54-year-old man with rhinophyma treated with dermabrasion. (a) Predermabrasion. (b) 2 weeks postdermabrasion. (c) 8 months postdermabrasion.
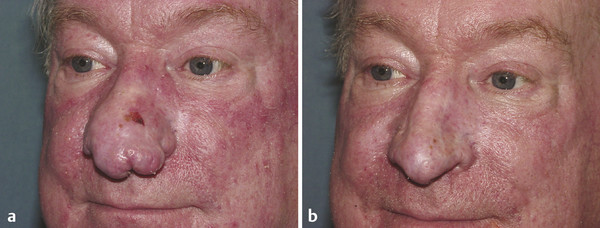
Fig. 29.7 A 57-year-old man with rhinophyma treated with dermabrasion. (a) Predermabrasion. (b) 9 months postdermabrasion.
29.4 Contraindications
Dermabrasion is contraindicated for any patients who have decreased adnexal structures of the dermis and therefore may not be able to mount optimal re-epithelialization. Comorbidities associated with poor re-epithelialization include history of radiation treatment to the facial areas, long-term systemic steroid therapy, or history of deep burns.
Patients with active herpetic lesions should delay dermabrasion until any current infection resolves. Active infection may disseminate into newly abraded wounds, and subsequent infection may lead to full-thickness tissue loss.8 Patients presenting with acne scars may have been or currently may be on systemic isotretinoin (13-cis-retinoic acid). This medication leads to atrophy of the sebaceous glands and may compromise wound healing and re-epithelialization. There are conflicting studies that challenge whether systemic isotretinoin causes atypical scarring after dermabrasion.24,25 Nevertheless, to minimize any risk of scarring, one should wait at least 6 months and preferably a full year before proceeding with dermabrasion on patients taking systemic isotretinoin.
Medications that contain estrogen or progesterone may lead to pigmentary changes following dermabrasion. Therefore, if possible, it is recommended that patients discontinue such medications for 3 to 6 months posttreatment.
Finally, detailed histories should be obtained to rule out patients with blood-borne pathogens such as human immunodeficiency virus (HIV) or hepatitis B/C virus (HBV/HCV). Dermabrasion treatment can create aerosolization of the blood pathogens. Although blood-borne pathogens may transmit through mucous membrane exposure, there are no known instances of a blood-borne pathogen being transmitted by an aerosol in a clinical setting.26 Nevertheless, it is good clinical practice for the surgeons and assistants to have appropriate protective shielding against potential aerosol particles (< 10 µm in diameter) and splatter during the procedure.
29.5 Preoperative Preparation
Patients must demonstrate clear understanding of the realistic outcome, posttreatment care, and posttreatment appearance prior to consenting for the treatment. They need to be aware that some pigment changes are expected during the recovery process. Also, they must commit to rigorous posttreatment care in order to achieve the optimal outcome, including protection/avoidance from the sun for at least 3 months posttreatment.
Topical application of tretinoin 0.5% prior to dermabrasion has been shown to improve healing. Patients placed on tretinoin at least several weeks before dermabrasion healed 2 to 3 days faster than those without pretreatment, and had reduced risk of postinflammatory hyperpigmentation.27
For patients with a dark skin complexion (Fitzpatrick skin type IV or higher) or history of developing hyperpigmentation after skin resurfacing, hydroquinone 4% topical cream should be initiated several weeks prior to dermabrasion, and resumed soon after posttreatment re-epithelialization. Hydroquinone works by inhibiting the actions of the enzyme tyrosinase, thereby blocking the melanin production involved in skin pigmentation.5,28
Oral antiviral prophylaxis against herpes simplex virus (HSV) should be considered for all patients and particularly those with a history of reactivation.
29.6 Technique
The dermabrasion handpiece operates at a range of 1,500 to 50,000 revolutions per minute. The handpiece can accommodate various sizes and types of debrider heads (▶ Fig. 29.2a, b). The two most commonly used debrider heads are the diamond fraise and wire brush. The wire brush abrades skin by creating multiple microscopic lacerations and tends to achieve the required depth more quickly than the diamond fraise (▶ Fig. 29.2c). Nevertheless, this distinction also makes the wire brush more challenging to use for an inexperienced surgeon. A diamond-fraise tip may abrade at a slower rate, but is relatively easier to control and viewed as the safer option. For the areas that are small and hard to reach such as the grooves of the nasal alae or near the eyelids, the diamond fraise should be utilized (▶ Fig. 29.2d). Traditionally, cryogenic agents were applied topically to freeze the skin and create a rigid surface for even treatment. Unfortunately, these agents are no longer available. After the banning of fluorocarbon refrigerants, tumescent anesthesia has been used to induce turgor over the skin with similar effect. The assistant should provide tension and stabilization of the skin being treated, while the surgeon uses the nondominant hand to protect the eyes, nose, and mouth. Gauze should not be used to protect these areas because gauze may actually damage the eyes, nose, or lips when caught with the wire brush. If a deep scar or pitted acne is being treated, one may use gentian violet to stain the epidermis in order to denote these areas after the first pass of dermabrasion.
Typically, the area to be dermabraded is marked in a grid pattern. Most surgeons plan on treating the entire aesthetic facial unit instead of a single spot because the treated and untreated areas of the facial unit tend to blend better and will be easier to camouflage. One should abrade deeper in the area of concern such as the acne scar or deep crease, while feathering lightly in the surrounding area to blend in the texture and color.
Uniform pressure is applied over the treated area, and the movement of the dermabrader is maintained slowly and steadily in a direction perpendicular to the plane of rotation. The depth of treatment is determined through several landmarks. Removal of the epidermal layer is characterized by changes in skin pigmentation (or the ink marking made by the surgeon). The small capillary pinpoint bleeding indicates that the depth of treatment has reached the papillary dermis level. If the superficial reticular dermis is exposed, one will notice the parallel-oriented strand of yellow layer. To optimize the visualization of skin depth and avoid blood pooling, the procedure should be planned to begin in the gravity-dependent areas.
While treating near critical structures of the face such as the lips or the eyes, one should orient the spinning wheel toward these structures. This will prevent the brush from grabbing and retracting the lips or eyelids and causing them to tear or avulse. Bony areas such as the mandible and malar prominence warrant special caution. These areas are naturally more prone to scar formation―likely due to their thin skin and constant tension from facial movements―and should be treated more conservatively.29
29.7 Postoperative Care
Application of occlusive dressing or topical ointment such as petroleum jelly has been shown to accelerate the healing process following dermabrasion. The covered wound retains liquid and tends to form less crusting than the wound exposed to the air. The crusting becomes a mechanical barrier for epidermal migration and slows down the re-epithelialization process. The rate of re-epithelialization may increase up to 40%, with complete epithelialization occurring within 24 hours when the wound is appropriately covered.8 Starting posttreatment day 1, patients are instructed to shower up to six times daily with gentle, lukewarm water to clear crusting at the treated area. Following the rinse, topical ointment should be applied immediately to keep the wound retaining its moisture and prevent crusting formation. Once re-epithelialization has been completed, usually between the 1- and 2-week follow-up visits, topical care can be transitioned from a thick ointment to a gentle skin moisturizer. Ideally, the skin moisturizer should not contain any sunscreen agents because these may irritate the nascent skin. At this point, patients may also begin to wear makeup to camouflage the posttreatment erythema. Water-based makeups are recommended because they are easier to remove. The posttreatment erythema usually subsides after 8 weeks; however, for some patients this may take several months. During the first 3 months patients should avoid any sun exposure. Particularly for recalcitrant ice-pick acne scars and rhinophyma, dermabrasion may be repeated. After the treated area has re-epithelialized, a second dermabrasion may be performed at least 3 months later to remove residual or deep skin defect and achieve further improvement.
29.8 Complications
Approximately 50% of patients will develop milia during 1 month after dermabrasion.8 Milia are superficial inclusion cysts limited within the epidermis during re-epithelialization (▶ Fig. 29.8a). They are typically self-limiting, but large milia can be opened with an 18-gauge needle for quick resolution. Posttreatment 0.05% topical retinoid has also been shown to be effective in reducing incidence of milia.27
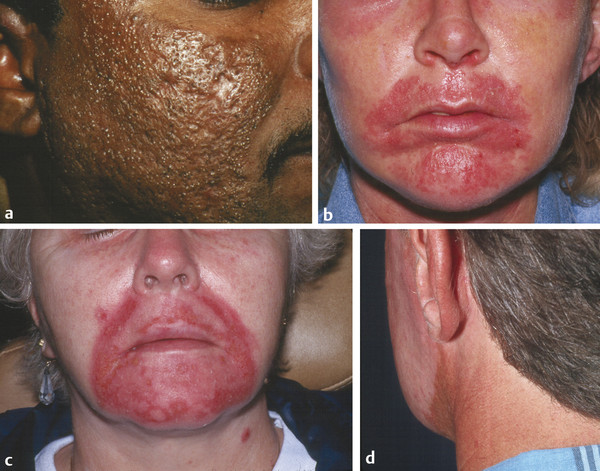
Fig. 29.8 (a) Milia and temporary hyperpigmentation are noted at the treated sites. (b) Tenderness, erythema, and white reticular streaks are signs of candidal infection at the treated site. (c) Postdermabrasion herpetic lesions clinically present as out-of-proportion tenderness with vesicular eruption. (d) Permanent hypopigmentation. Notice the line of demarcation along the angle of mandible.









