4 Current concepts in revisionary breast surgery
Synopsis
 Breast augmentation is the most common aesthetic procedure performed in the United States.
Breast augmentation is the most common aesthetic procedure performed in the United States.
 Revisionary breast surgeries are complex, challenging, and unpredictable.
Revisionary breast surgeries are complex, challenging, and unpredictable.
 Four main drivers for revisionary surgery: capsular contracture, implant malposition, ptosis, and implant visibility or palpability.
Four main drivers for revisionary surgery: capsular contracture, implant malposition, ptosis, and implant visibility or palpability.
 Authors introduce revisionary surgery techniques, utilizing a site change operation with the use of acellular dermal matrix (ADM).
Authors introduce revisionary surgery techniques, utilizing a site change operation with the use of acellular dermal matrix (ADM).
Introduction
It is estimated that over 300 000 primary breast augmentations were performed in the United States in 2009, and therefore there are now over 3 million women with augmented breasts in this country.1–3 Based on current data, between 15% and 30% of these women, will have a reoperation within 5 years of their initial procedure.1–3 Unfortunately, this rate climbs to 35% in patients with a prior history of revisionary breast augmentation.4 As procedures become more complex in nature and number, new techniques and solutions are required of surgeons who perform these challenging operations to improve long-term patient outcomes.
Capsular contracture has historically been the most common complication of aesthetic and reconstructive breast surgery and remains the primary reason for most revisionary surgeries.2,3,5,6 While increasing data suggests capsular contracture can be minimized in primary augmentation by technical detail including precise, atraumatic, bloodless dissection; appropriate antibiotic breast pocket irrigation; and minimizing any points of contamination during the procedure,4,7 treatment of an established capsule remains even more challenging than the application of these techniques alone.
The enforcement of the US FDA restrictions on silicone gel implants in the early 1990s led American surgeons to use saline implants.1 While prior to the 1992 “moratorium”, the majority of silicone gel implants were placed in the subglandular position, saline implants (due to their palpability) began to be placed under the muscle in an effort to conceal the untoward contour irregularities of these implants.8 As a number of these implants were of larger volumes, many patients experienced thinning of breast parenchyma and the overlying soft tissue, whether the implants were in subglandular or subpectoral positions. The thinned tissues, in turn, can lead to long-term complications, which had led to some key drivers of revisionary breast surgery.
The four primary reasons (or “Drivers”) for aesthetic revision surgery in the Pre-Market Approval (PMA) studies were as follows:9 (1) capsular contracture, (2) implant malposition, (3) ptosis, and (4) implant visibility or palpability. Frequently, these indications were not singularly distinct, but were combined with two or more being present in a given patient.
Hints and tips
• There are four key drivers to revisionary breast surgery: (1) capsular contracture; (2) implant malposition; (3) ptosis, and (4) implant visibility or palpability. Frequently, these indications are not singularly distinct, but were combined with two or more being present in a given patient.
• Capsular contracture is the most common driver for revisionary breast surgery.
• Site change and acellular dermal matrix are key for successful outcomes in revisionary breast surgery.
History
Historically, our options for revision and improvement have included: replacing saline implants with gel implants, capsulorrhaphy, use of capsular flaps, or performing a site change operation. The site change principle, described in the mid-1980s, combined total or partial capsulectomy with conversion to a different pocket (generally a subglandular to subpectoral site change) for the replacement implant.10 While site change procedures have been successful, none of these procedures alone have resulted in complete resolution of the described complaints
In our experience, the majority of breast revisions require a new pocket for the new implant. Since many patients have had implants in multiple pockets, and as the majority of implants in revisionary patients today are already in the subpectoral position, we developed (in 1991) the “neopectoral pocket” concept to create a new pocket in the “subpectoral-precapsular space”.11 Initially developed for inferior malposition, we also applied our concept to medial malposition, capsular contracture, ptosis, and conversion of round to anatomical shaped implants (demanding a snug, hand-in-glove “fit”). The detailed operative technique of creation of the neopectoral pocket has been described in a previous publication.8 For patients presenting with implants in the subglandular position, a sub-pectoral site change is utilized.
Notwithstanding the importance of the site change concept, the most important addition to our clinical armamentarium has been the utilization of acellular dermal matrix as a regenerative construct to help solve these challenging clinical presentations.12 The successful use of acellular dermal matrices (ADMs) has been reported in a range of clinical settings including abdominal wall repair, hernia repair, facial and eyelid surgery, cleft palate repair, soft tissue augmentation, tendon repair, ulcer repair, vaginal sling repair, and breast reconstruction.13–23 While its use in reconstructive expander and implant breast surgery has become a standard of care, its use in aesthetic revisionary breast surgery has evolved more slowly,24–26 and has only recently gained widespread adoption.24 The senior author has been a pioneer in this field over the last 10 years utilizing virtually every commercially available ADM product in various sizes, shapes, and thicknesses to better clinically define regenerative and biomechanical material properties and refine more clearly the specific clinical indications and techniques of use.
Basic science and disease process
Acellular dermal materials, biologically derived from allograft and xenograft, when placed in the human body, are thought to serve as a regenerative scaffold, promoting the organization of the healing process. These materials have become popular in breast cancer reconstruction, where they are said to serve as a tissue extension or tissue replacement (“soft-tissue patch”) following cancer extirpation of the breast (so-called “sling technique”).22,23,27
Our work with acellular dermal matrix (ADM) began in revisionary aesthetic breast surgery, attempting to prevent capsular contracture, rather than as a tissue replacement in breast reconstruction. Having previously employed “host-compatible” implant surfaces,28 here we have utilized a similar concept in our clinical approach to breast revision: a dermal regenerative interface engaging the surface geometric contour of a breast implant.
Capsular contracture has plagued plastic surgery as the most common complication of aesthetic and reconstructive breast surgery for many years.2,5 The majority of revisionary breast surgeries are performed to correct capsular contracture.2,6 Many etiologies have been proposed for this process, and it is clear that prevention of it in primary cases includes sound techniques – including precise, atraumatic, bloodless dissection; appropriate triple antibiotic breast pocket irrigation; and minimizing any points of contamination during the procedure.4,7 Treatment of an established capsule can be more challenging, and multiple techniques have been utilized for this. The bottom line for any pathophysiological process is to understand the disease at the cellular level. In this case, it is perspicuous that at the cellular level, capsular contracture is most likely caused by any process that will produce increased inflammation, leading to formation of deleterious cytokines within the periprosthetic pocket. Consequently, in addition to all of the techniques for treating and preventing capsular contracture described by many of our colleagues,4,5,8,29–35 we believe that the addition of ADM is another modality in fighting the evolution of the capsule. ADM can counteract the inflammatory process, adding additional availability of tissue in-growth and controlling the interface of the pocket.
Acellular dermal matrix
Use of acellular dermal matrices have been popularized in both breast and abdominal wall reconstructions and has been reported in a range of clinical settings.13–23 In reconstructive cases, it has been used to replace tissue, extend existing tissue, or act as a supplement. In aesthetic cases, it has been used to correct implant rippling and displacement, including symmastia.25,26,36
Immediate breast reconstruction using tissue expanders or implants has become one of the most commonly used surgical techniques, which has made visible rippling and contour deformity a more frequently encountered problem.27 The recent use of allogeneic tissue supplements avoids the problems of autologous tissue coverage and provides camouflage, thus decreasing rippling, and increasing soft tissue padding.27
Acellular dermal matrices can be categorized under either xenograft or allograft in origin. All are produced with a similar objective of removing cellular and antigenic components that can cause rejection and infection. The lack of immunogenic epitopes enables the evasion of rejection, absorption, and extrusion.14,15,23 Production processes allow the basement membrane and cellular matrix to remain intact. This scaffold is left in place to allow in-growth of host fibroblasts and capillaries to eventually incorporate as its own. Much of this scaffold matrix consists of intact collagen fibers and bundles to support tissue in-growth, proteins, intact elastin, hyaluronic acid, fibronectin, fibrillar collagen, collagen VI, vascular channels and proteoglycan – all of which allow the body to mount its own tissue regeneration process.14,15,22,23
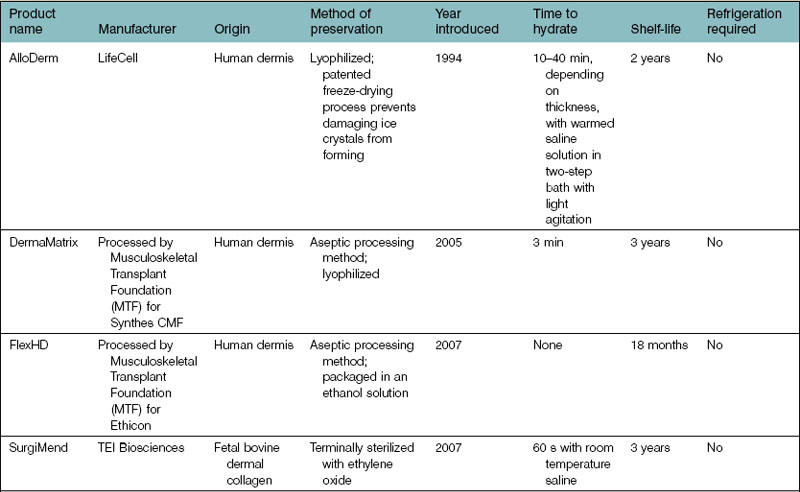
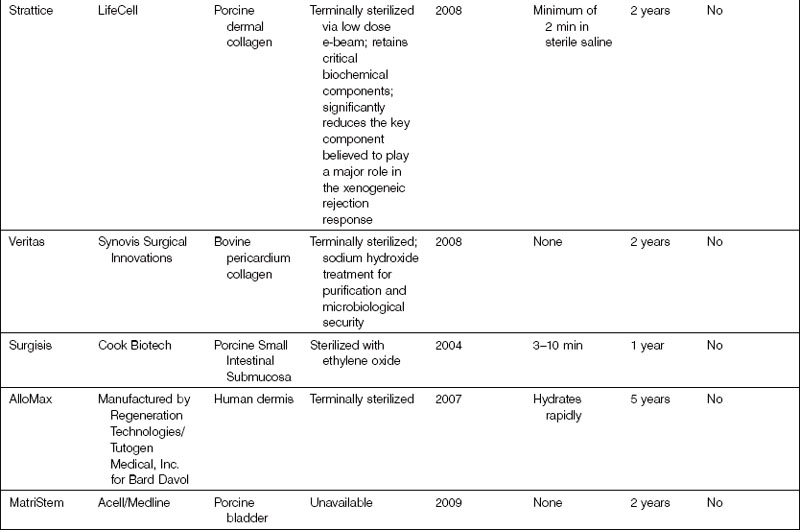
All ADMs are FDA cleared for homologous use only. Although features and processing may vary between ADMs, the success of the product will ultimately depend on its ability to meet the desired characteristics of a model ADM for revisionary breast surgery. A list of ADMs that are available on the market for a variety of applications are included in Table 4.1.
Published literature
AlloDerm is clearly in the forefront regarding published literature concerning ADMs for immediate breast reconstruction. A PubMed search using specific brand names and breast reconstruction reveals the majority of the publications involving AlloDerm. Ten of these papers relate directly to adjunctive ADM treatment of immediate breast reconstruction using AlloDerm,14,15,22,23,27,37–39 Dermamatrix,40 and Neoform.41 All of these papers are recent publications, the oldest dating from 2005.14 All are noncontrolled, retrospective case series. PubMed searches were also performed matching each name of all other human and xenograft ADMs – FlexHD, Allomax, Surgimend, Enduragen, Synovis, Permacol, and Strattice – with breast reconstruction and revealed no listings. It is likely that studies are underway and not yet completed.
Encapsulation
The goal of regenerating tissue is to recapitulate in adult wounded tissue the intrinsic regenerative processes that are involved in normal adult tissue maintenance.42 Scar does not have the native structure, function, and physiology of the original normal tissue. When a wound exceeds a critical deficit, it requires a scaffold to organize tissue replacement. Depending on the type of scaffold that is in place, different processes as described earlier will respond. At this point, the intrinsic factors of the ADM will be important to aid in each specific regenerative or reparative process. While regenerative healing is characterized by the restoration of the structure, function, and physiology of damaged or absent tissue, reparative healing is characterized by wound closure through scar formation.42 All biologic scaffolds are not the same because of differences in the methods used to process them – materials that encapsulate and scar do not offer the benefits of regenerative healing but lead to suboptimal results.
Diagnosis and patient presentation
Acellular dermal matrix is utilized as an adjunct to the sound surgical principles necessary to diagnose and treat the underlying cause(s) necessitating revisionary aesthetic breast surgery. Clinical data shows the four main indications (drivers) for revisionary surgery are capsular contracture, implant malposition, ptosis, and implant visibility or palpability.24–26 Each patient must be individually evaluated regarding concerns, goals, knowledge of previous surgical and implant specifics, and careful evaluation of her breasts – dimensions, quality/quantity of overlying soft tissue, and scarring (critical in planning surgery and maintaining necessary vascularity to manipulated tissues).