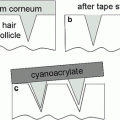
Fig. 17.1
Corneoxenometry. Aspect of a cyanoacrylate skin surface stripping stained by a toluidine blue-basic fuchsine after contact with a penetration enhancer. The staining of corneocytes is uneven and indicates where the damages take place
17.3 Dose-Response Corneoxenometry with Chemical Penetration Enhancers
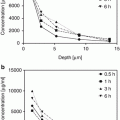
Data from both corneosurfametry and corneoxenometry are reproducible and sensitive enough to frequently disclose significant CIM and RII differences between formulations (Piérard et al. 1995). A dose-response effect was searched for ethanol and laurocapram using the corneoxenometry bioassay (Goffin et al. 2000). In the same study, other assessments were performed using a gel formulation (propylene carbonate, hydroxypropyl cellulose, butylhydroxytoluene, ethanol, glycerol) containing 10 % propylene glycol and a combination of three other enhancers, namely, N-acetyl-L-cysteine (NAC), urea and salicylic acid (SA). The three latter penetration enhancers were present in various proportions with keeping their global concentrations at the 20 % level.
Both the nature and concentration of the respective penetration enhancers affected the RII values. For each formulation, the interindividual variability was reasonably low. Linear dose-effect responses were obtained with ethanol in the range 0–100 %, and laurocapram in the range 0–5 %. The 10 % propylene glycol-based gel exhibited a wide range in RII values when supplemented with NAC, urea and SA. In the bioassay, NAC exhibited a moderate effect on the SC. RII values raised with increasing amounts of urea replacing NAC. The RII worsening was more striking with SA supplementation replacing urea. The combination of SA and urea always proved to be more active than SA alone.
17.4 Corneoxenometry and Organic Solvents
The effects of organic solvents were studied on many occasions (Peck et al. 1994; Garcia et al. 2000). In particular, they were compared using corneoxenometry (Ochalek et al. 2012). Series of CSSS were immersed for 1, 5, 10, 30, 60 or 120 min in vials containing deionized water or an organic solvent including chloroform, ethanol, hexane, methanol, chloroform:methanol (2:1, v/v), hexane:ethanol (2:3, v/v) and hexane:methanol (2:3, v/v). After contact with the selected solvent(s) for the predetermined time, CSSS were thoroughly rinsed for 20 s under running tap water, air-dried and stained for 3 min with toluidine blue-basic fuchsine dyes.
The CIM ranking from the least to the most aggressive product was as follows: hexane (40.7), ethanol (26.5), methanol (23.5), hexane-ethanol (23.3), chloroform (20.8), chloroform-methanol (15.5) and hexane-methanol (7.8). CIM values showed that the effect of hexane-methanol on SC was significantly higher (p < 0.01) than those of all other solvents with the exception of chloroform-methanol. There was no significant difference between ethanol, methanol and hexane-ethanol, but each of them was significantly (p < 0.05) more aggressive than hexane.
The influence of exposure time of solvents with the SC showed some inter-product differences. However, all correlations reached significance (p < 0.01) and best fitted as logarithmic relationships. For each solvent, most of the CIM changes were reached within 10 min.
The organic solvents under consideration are known to extract lipids (Bligh and Dyer 1959; Scheuplein and Ross 1970; Deffond et al. 1986; Imokawa et al. 1986; Abrams et al. 1993; Lavrijsen et al. 1994). In addition, SC alterations other than lipid extraction are likely (Abrams et al. 1993). Large interindividual CIM differences were found for each solvent or mixture (Goffin et al. 1997b) reflecting the variability in the overall lipid extraction by these solvents (Diembeck et al. 1999). The induced alterations on normal human SC by solvents (corneoxenometry bioassay) were indeed reported to be more variable than those induced by diluted surfactants (corneosurfametry bioassay) (Goffin et al. 1998; Xhauflaire-Uhoda et al. 2008b). Despite interindividual inconsistencies in corneocyte alterations, significant differences were reported among solvents using the corneoxenometry bioassay (Goffin et al. 1997b). Hexane-methanol and chloroform-methanol were the mixtures strongly altering the SC structure. Chloroform-methanol is indeed considered to be the most potent extraction mixture for lipids in biologic samples. However, it did not reach the top ranking at the corneoxenometry bioassay (Goffin et al. 1997b). Such a finding further illustrated the fact that organic solvents alter other biologic components (Diembeck et al. 1999), which in turn affect the corneoxenometry data.
The corneoxenometry bioassay allows to assess the influence of the contact time between solvents and the SC. In previous studies (Goffin et al. 1997b), the time range between 1 and 120 min was selected following available information about the kinetics of lipid extraction from human SC [13]. The corneoxenometry data were in line with previous experiments using other methodological approaches (Deffond et al. 1986; Imokawa et al. 1986; Abrams et al. 1993; Lavrijsen et al. 1994). However, it does not explore the effects of solvents on the living epidermis and on the nature and intensity of inflammation that is present in irritant dermatitis.
Conclusion
Corneoxenometry appears as a relevant and predictive bioassay for assessing the overall effect of single and combined penetration enhancers. It is cheap, rapid, minimally invasive and relevant to human skin. In addition, the reproducibility, specificity and sensibility are reasonably high. Corneoxenometry is therefore a valuable screening test proposed as an alternative to animal and in vitro testings.
References
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917CrossRefPubMed
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree