Dermal fillers, in particular hyaluronic acid gel (HAG) fillers, are used in the treatment of aging changes in the periocular area. Filler treatment requires in-depth knowledge of specific issues relating to product performance and administration, safety protocols, and recognition and treatment of complications. There are different approaches to treatment of the tear trough. Prior filler treatment must be suspected in patients presenting for aesthetic evaluation, and the possibility of migration with a dysmorphic appearance and/or Tyndall effect appearance always is kept in mind. Treatment with hyaluronidase injection generally is effective in reducing overcorrection or migration of HAG in this area.
Key points
- •
Dermal fillers, predominantly hyaluronic acid gels (HAGs), have great utility in treating aging changes of the periocular area.
- •
Dermal fillers vary in their results and suitability in the periocular area, with generally predictable results and complication rates.
- •
Clinical results and complications are related specifically to biochemical and rheologic characteristics of the product selected, the specific patient, and how the filler is injected.
- •
Hyaluronidase injection is an essential element in the treatment of most HAG dermal filler complications. Complications of other fillers are not treatable in this fashion.
- •
Blindness and neurovascular embolization complications after dermal filler injection is a special topic addressed separately in Catherine J. Hwang and colleagues’ article, “ Blindness After Filler Injection: Mechanism and Treatment ,” in this issue.
Video content accompanies this article at http://www.facialplastic.theclinics.com .
Overview
Periocular aging is characterized by thinning of the tissues encircling the orbit, apparent sagging and redundancy of upper eyelid skin, and pseudoherniation of lower eyelid fat. Surgical approaches may address all these factors, but the use of cosmetic dermal fillers, primarily hyaluronic acid gels (HAGs), is a popular approach to address volume deficit in these aging tissues. More than 1.5 million people undergo facial injection with a synthetic dermal filler yearly in the United States. These products have good utility and an excellent safety profile, even in the periocular area, although undesirable outcomes can occur. ,
The most commonly used dermal fillers are synthetic HAG fillers, with a smaller representation of collagen, calcium hydroxylapatite gel, poly- l -lactic acid, and polymethylmethacrylate beads. Autogenous fat also is employed surgically, which is a different topic, and liquid silicone and other unapproved, imported, or withdrawn products are employed by some injectors.
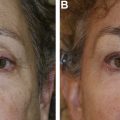
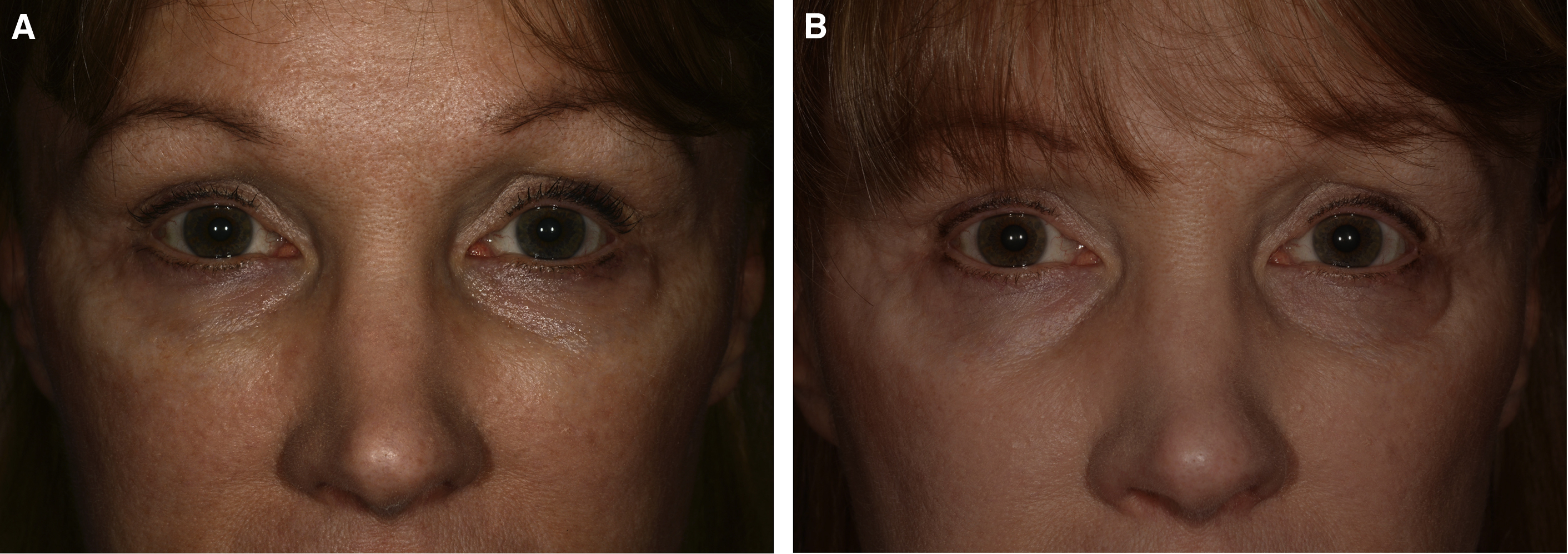
The use of HAG and other fillers in the midface and upper face is associated with several risks, most commonly undercorrection or overcorrection of the soft tissue deficit or the placement of or migration of the filler into an inappropriate location ( Fig. 1 ). Use in the lower eyelid commonly is associated with the Tyndall effect, whose origin is debated.
Additional adverse outcomes include the most feared—vascular or embolization events—or may relate to placement issues, nodules and migration, allergy or hypersensitivity to the product, acute or late chronic infection, granuloma formation, or other chronic edema.
Appropriate patient history and selection, preparation, procedural and product decision making, and aftercare, as needed, result in optimal results. Hyaluronidase injection is a vital tool in managing and reversing most adverse effects of HAG filler treatment, with other approaches needed in the treatment of other complications and classes of fillers.
General comments on dermal filler complications
Patient-Related Issues
Inappropriate patient selection often is the starting point in a complication. In that regard, it is the injecting physician’s role to screen the patient and determine whether the patient is a suitable candidate for the desired treatment. Patients with inappropriate motivation, obvious active social or psychiatric issues, and body dysmorphic syndrome should be rejected, because it is the injector’s role to select the patient and treatment, not vice versa.
A thorough history of skin conditions, allergies, systemic diseases, current medications, and previous procedures is essential. Acne, rosacea, and other infectious or inflammatory skin conditions all compromise the epithelial skin barrier and may promote inflammatory and infectious or biofilm reactions to dermal fillers. A history of cold sore or herpetic outbreaks may mandate pretreatment and post-treatment antiviral prophylaxis.
Patients with active autoimmune disease, including rheumatologic and thyroid disease, must be delayed or rejected. The procedural checklist should include temporal distancing from dental cleaning and procedures, immunizations, other medical and surgical procedures, blood thinners, and treatments or activities that disrupt the epithelial barrier (longer delay for thin or compromised skin). Three patients with a history of HAG filler treatment were noted in the Moderna COVID-19 vaccine trial to have a localized inflammatory reaction at the site of injection. These reactions were self-limited but certainly concerning. Contacting patients a week before treatments to review these checklist items is wise.
Product-Related Issues
There are vital general physical property–related differences between dermal filler products that intuitively provide some guide to their use. There also are properties discovered in the use of products that, in the realm of clinical consensus or expert injector advice, are recommended. In that regard, in the periocular area, the ability to reverse the effects of HAG fillers with hyaluronidase is a vital factor in the selection of those products for most periocular treatments. HAG fillers are hygroscopic in nature, with a variable tendency to pull water into them after injection. Undercorrection of defects generally is desirable, and various products have variable amounts of cross-linking, free or short-chain HAG, and HAG concentration that affect their suitability in the periocular area (and other areas). Related rheologic characteristics include viscosity, G′ (elastic modulus), and Gꞌꞌ or tan delta (viscous modulus). These properties are crucial in determining the appropriate depth and site for the placement of a specific product.
Procedure/Injector-Related Issues
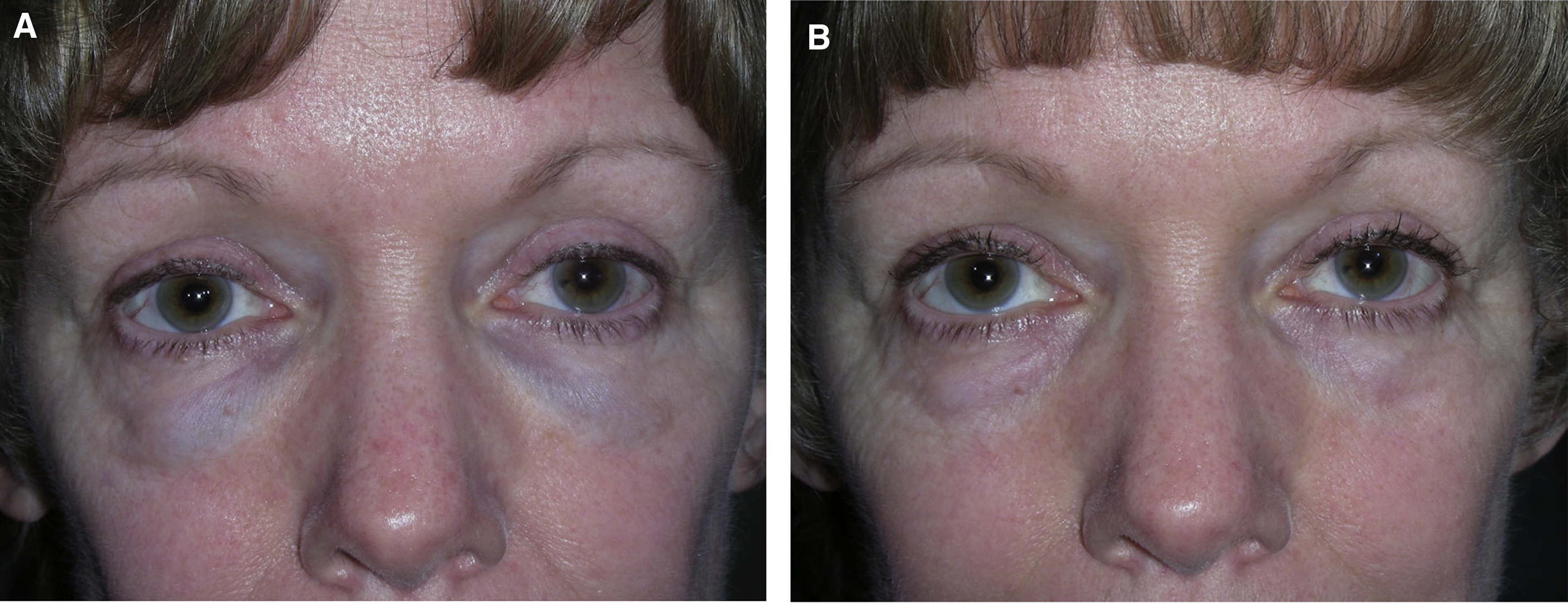
Like all cosmetic injectable products, the results achieved with dermal fillers in the periocular area ultimately largely are up to the treating practitioner’s product selection and placement. Depth, volume, rate of flow, and device used for placement all are injector related. Good placement does not compensate for an inappropriate product selection, and vice versa. Injectable filler products are licensed as implants, and that is a good way to always think of them. The common preconception of many patients and, unfortunately, many injectors, that dermal fillers are injected and completely or largely dissipate, or dissolve over 6 months to 12 months, is false. HAG may persist with some breakdown, migration, and change in effect that goes on for a decade or more ( Fig. 2 ). A thorough knowledge of facial anatomy as well as a history of an individual patient’s previous treatments is essential for injectors. Layering of various products intentionally or unintentionally occurs in many patients over time and has intentional and unintentional effects.
Specific complications
Allergic/Hypersensitivity—Allergy Versus Late Onset
The severity of the allergic reaction is paramount ( Box 1 has a detailed list of soft tissue filler complications). Antihistamines may be effective if the reaction is mast cell–mediated. Oral steroids at doses and duration of treatment determined by the timing and severity of reaction are mainstays for allergic swelling and hypersensitivity not responsive to antihistamines. Delayed hypersensitivity reactions may require oral steroid or other treatment. ,
Early reactions
Vascular infarction/soft tissue necrosis
Inflammatory reactions (acute/chronic)
Infection
Allergic reactions/hypersensitivity
Injection-related events
Pain
Ecchymosis
Erythema
Bruising
Bleeding
Inappropriate/superficial placement
Distant spread
Late reactions
Inflammatory reactions (acute/chronic)
Infection
Granuloma (typically chronic)
Differential diagnosis
Nodules
Dyspigmentation
Displacement of hyaluronic acid (HAG) filler material
Nodules/Granulomas/Inflammation/Infection
Noninflammatory nodules encountered early after hyaluronic acid (HAG) treatment may resolve with time and with judicious application of hyaluronidase. Inflammatory nodules must be assessed and followed to determine if they are due to infection. Some inflammation is common early after treatment with some fillers, but persistence, increasing pain, or erythema or fluctuance should prompt antibiotic therapy (generally, a macrolide and/or tetracycline derivative along with a topical antibiotic, such as mupirocin) and drainage of fluctuance. Treatment may require a biopsy with aerobic and anaerobic culture, holding the cultures for 14 days to 21 days. Dermal fillers form microglobules in the tissue that can support chronic biofilm infections, so chronic inflammatory reactions always must be presumed to be infectious until proved otherwise. Resolution of such infections likely requires a 4-week to 6-week course of antibiotics and multiple hyaluronidase injections to eliminate HAG fillers to the extent possible.
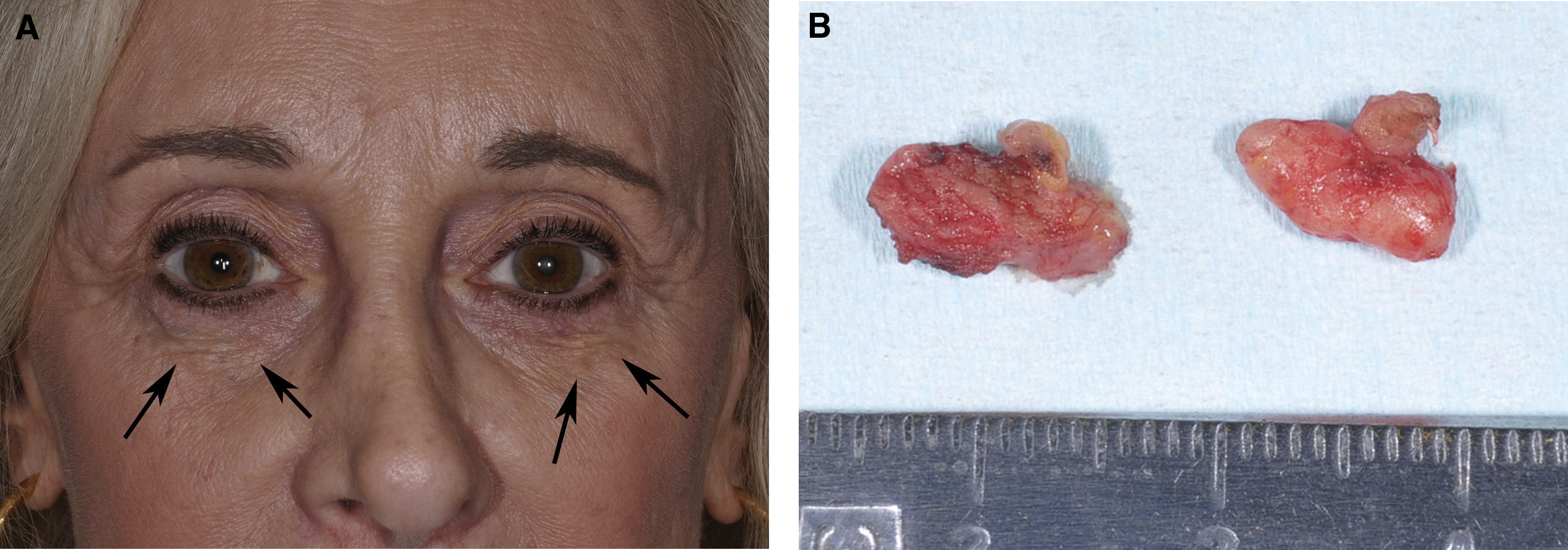
The use of hydroxylapatite gel, poly- l -lactic acid, or polymethylmethacrylate beads in mobile structures, such as the lip, or over thin tissues, such as the tear trough, may result in visible nodules. Hydroxylapatite gel and poly- l -lactic dissipate over some time if the changes are not too severe. Polymethylmethacrylate beads do not resorb, and chronic nodules generally are surgically excised ( Fig. 3 ), although the injection of triamcinolone and 5-fluorouracil is reported in the treatment. Inflammatory reaction to the collagen-containing vehicle of polymethylmethacrylate beads as well as poly- l -lactic acid may respond to injected steroid, and the injection of saline followed by vigorous massage has been performed with poly- l -lactic acid to break up clumps of possibly poorly mixed product causing nodules. 5-Fluorouracil also has been injected to help soften scar tissue surrounding the implant.
Vascular Events
Vascular embolization with distal vascular occlusion and local necrosis is possible in almost any site, but the risk largely is technique related and occurs most commonly over the glabella and nasion. The use of 25-gauge or larger blunt cannulas for injection appears to diminish the risk of arterial embolization. Immediate skin blanching occurs in an arterial distribution with arterial injection, and venous occlusion may occur due to excessive infiltration in a small area.
Rapid recognition and treatment are essential in the treatment of occlusive events. , Stopping injection, aspiration, massage, warm compresses, and hyaluronidase injection in significant doses (150–300 U) are performed, with consideration of the application of 2% nitroglycerine paste. Patients are followed closely with local care to areas of skin necrosis to optimize outcome.
Embolization with visual loss or other neurologic complication is addressed in detail in Catherine J. Hwang and colleagues’ article, “ Blindness After Filler Injection: Mechanism and Treatment ,” in this issue.
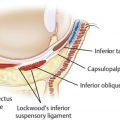
Comments on the treatment of the tear trough area
Undercorrection
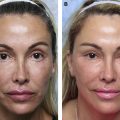
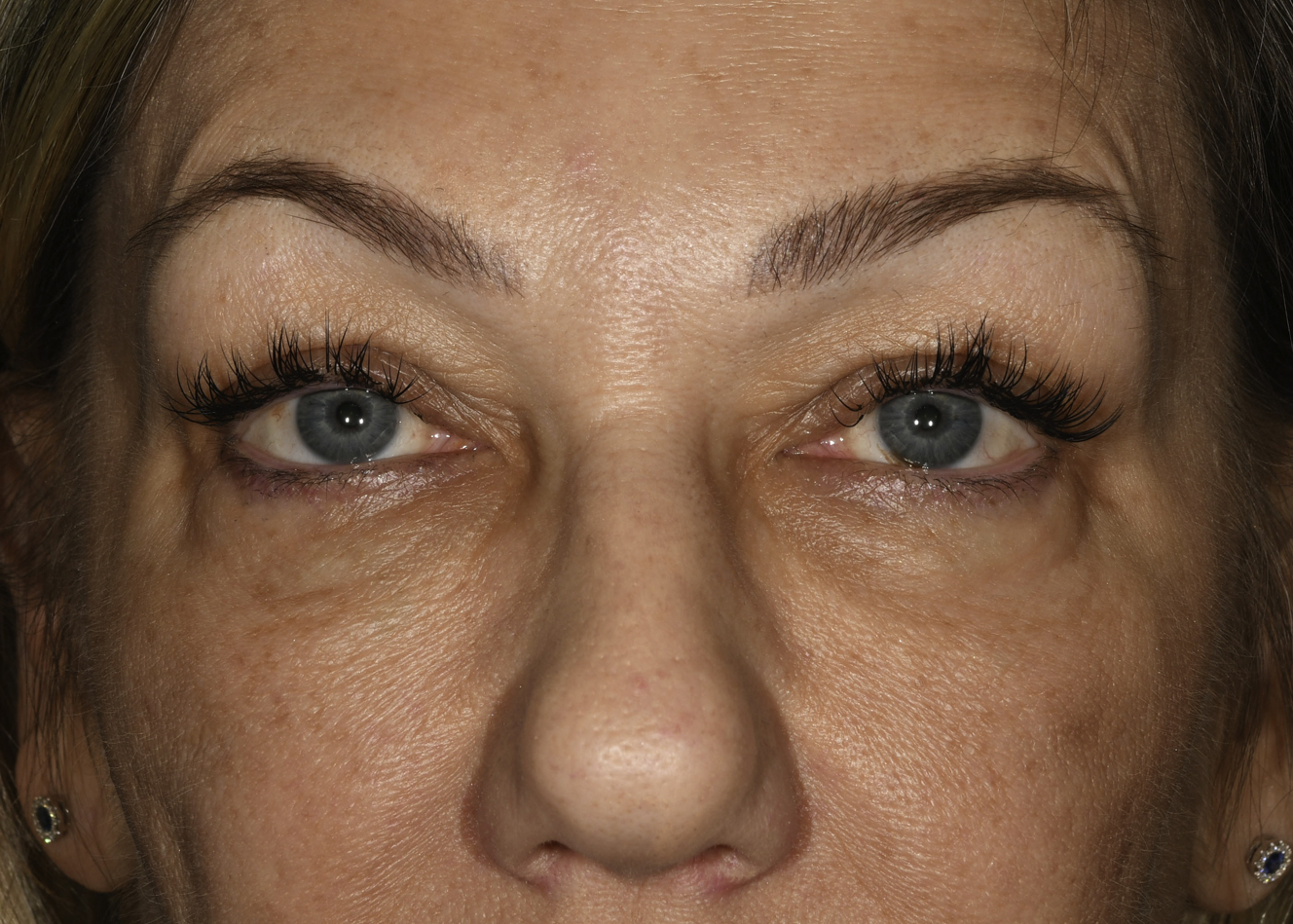
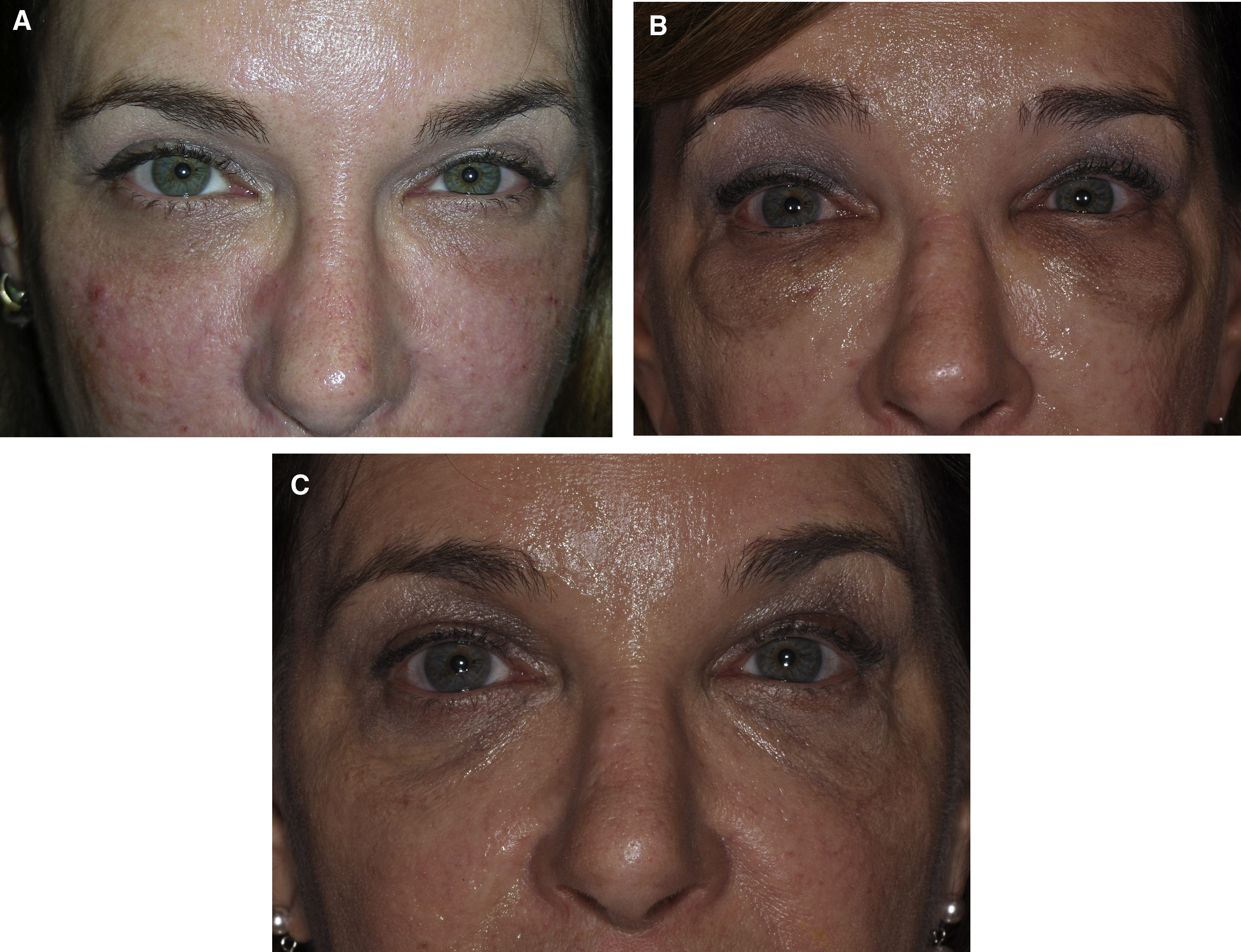
Patients often have filler suggested or request filler injection of the tear trough in situations in which HAG filler injection is not appropriate to this area. Patients with marked tear trough depressions in this area who anticipate outstanding results are sure setups for disappointment. Fig. 4 shows such a patient who experienced dissatisfaction with their tear trough treatment after 2 syringes (2 mL) of HAG filler. Additionally, product selection is paramount in this area, with the Hylacross fillers (Juvéderm Ultra and Juvéderm Ultra Plus, Allergan Aesthetics) particularly problematic and unsuitable to apply in the tear trough area ( Figs. 5 and 6 ). The authors see patients who desire correction and in whom more than 1 syringe of filler clearly is needed to achieve the desired result. The authors’ preferred product in this area is Restylane-L (Galderma, Ft. Worth, TX, USA), and they rarely inject more than 1 mL to 1.5 mL of this HAG into the tear trough on the initial treatment. This and Restylane Silk (Galderma) generally are considered the most suitable products for this area by the most injectors based on physical properties, in particularly G′, and their track record in the tear trough. In the authors’ hands, the incidence of lumps, Tyndall effect, or inadequate clinical effect has been greater when larger amounts are injected, and in most suitable patients the authors begin with part or all of 1 syringe (1 mL) ( Fig. 7 ). The authors additionally prefer an upward-fanning technique utilizing a 25-gauge cannula technique in treating the tear trough to maintain the product in a deep plane and avoid embolic or other complications ( [CR] ). The clinician must have a perspective on what is achievable and know when to tell a patient to stop if an adequate result is not possible.
Overcorrection
A variety of untoward overcorrection consequences occur in the periocular area related to the filler selection; method of administration, including volume, depth, persistence, and inflammation; and ecchymosis induced by the injection. The Tyndall effect, characterized by a bluish discoloration of the treated area, is a common in the tear trough area and appears to arise from the subcutaneous or intramuscular injection or migration of filler. There is some debate as to whether the bluish color is a true Tyndall effect. Overcorrection may occur immediately at placement or months or years later due to migration or presumed chemical changes in the filler.
Product Issues—Author’s Observations
Note that the first author has no financial interest in any company marketing a filler product, except for a small long-term investment in Revance Therapeutics (Revance Therapeutics, Nashville, TN, USA), and has not served as a consultant or taken an honorarium for any company marketing filler products for more than 10 years. Recommendations are based solely on personal experience, discussions with experienced colleagues, and published literature.
Migration of filler may occur months to many years after injection. The authors’ perspective on this has significantly evolved over time. There are treatment issues due to inappropriate placement of products. Different products have different characteristics on initial placement and as they age in position. The authors have seen a wide variety of fillers used in the tear trough, including polymethylmethacrylate beads (Bellafill, Merz Aesthetics, Raleigh, NC, USA), hydroxylapatite gel (Radiesse, Merz Aesthetics, Raleigh, NC, USA), and every HAG filler in common use in the United States. The authors have seen significant aesthetic complications with virtually every product and note the following observations regarding the periorbital use of fillers in the tear trough area:
- 1.
Polymethylmethacrylate bead (Bellafill) complications are severe and persistent. Visible filler requires covering the (hopefully deeply positioned) beads with HAG filler to camouflage it or surgical removal of the beads along with their capsule of fibrous encapsulation. Massage, needling, and injections with 5-fluorouracil and/or triamcinolone are described in the treatment of nodules resulting from this product, although these approaches work only early in the course , (see Fig. 3 ).
- 2.
Hydroxylapatite gel (Radiesse) is noted to be contraindicated in the tear trough by many investigators. Bernardini and coworkers have demonstrated its use favorably in the tear trough, although the authors have not used his technique.
- 3.
HAGs all are prone to the development of an overfilled result, with or without the bluish appearance of the Tyndall effect (see Figs. 1 and 2 ). The authors have examined patients in whom the most recent treatment with a HAG filler was 12 years prior, and the Tyndall effect was first noted 10 years after that, two years prior to presentation. The common presumption and treatment paradigm that these products “go away” in 6-12 months is entirely false and must be rejected and updated. Unfortunately most patients and injectors operate under these false assumptions.
- 4.
The Allergan Pharmaceutical Hylacross family of HAG fillers (Juvéderm Ultra and Ultra Plus) are quite hydrophilic and appear to be especially unsuitable products in the tear trough area. Kami Parsa, MD (personal communication, 2019) has presented results of patients he has treated for aesthetically undesirable results with numerous HAG fillers, including these products. There was 1 patient who required more than 30 hyaluronidase injections to resolve subcutaneous edema associated with a Hylacross filler. Often the skin is stretched and altered by the chronic edema, and aesthetic reconstruction is difficult. The authors have seen several patients with this complication and confirm this observation (see Fig. 5 A,B).
- 5.
The Restylane family of fillers appears most appropriate for use in the tear trough, with the original Restylane or Restylane-L preferred by the most injectors due to its favorable properties, including high Gꞌ. Restylane Silk shares similar properties and is preferred by some injectors. Advantages and properties noted in Restylane include the following:
- a.
Appropriate physical properties of flow, viscosity, high Gꞌ, and moderate hydrophilicity
- b.
Low and slow apparent diffusion through tissues with appropriate placement
- c.
Low risk of autoimmune reaction or nodule formation
- d.
Readily and rapidly removed with low to moderate doses of hyaluronidase (60–100 U/mL) in cases of migration or over-effect
- a.
- 6.
The Vycross family of fillers (Allergan Pharmaceuticals) similarly is prone to migration and Tyndall effect. Although not as hydrophilic as the Hylacross family of fillers, these products (Juvéderm Volbella, Juvéderm Vollure, and Juvéderm Voluma) are more resistant to and harder to remove with hyaluronidase injection than the Restylane (Galderma) family of fillers. As the heaviest of this family with the most persistence, Voluma appears to be the hardest to remove with hyaluronidase injection, with most patients requiring hyaluronidase doses greater than 200 U per 1 mL of filler for adequate resolution of overcorrections (see Fig. 6 ).
- 7.
Suspicion of prior filler placement or migration from the cheek or midface is important in the assessment of patients. Some aesthetic patients are notoriously poor in providing history, especially in regard to spa treatments often not administered by a physician. This is compounded when a treatment may have been many years ago and the problem has been noted recently. A therapeutic trial of hyaluronidase injection is advised whenever there is any suspicion of possible HAG filler migration in the tear trough area. Edematous festoons often have the bluish appearance of a Tyndall effect and are worsened by HAG filler migration. Filler migration always must be suspected in acquired dysmorphia!
- 8.
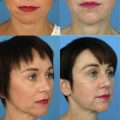
Patients presenting for lower eyelid evaluation in which dysmorphia related to HAG filler treatment is suspected require careful counseling and planning of any intervention, including hyaluronidase treatment. As bad as the swollen blue lower eyelids may look to a surgeon, it is the aesthetic the patient has lived with for some time, and the real upset may begin when the filler is dissolved and the tear trough change, lower lid bag, and skin laxity reassert themselves. These consequences must be explained to the patient, and, if the plan is to pursue surgery after hyaluronidase treatment, it should be scheduled in the near future before proceeding. The surgeon may wish to leave enough time interval for at least 1 more round of hyaluronidase before the surgery date arrives. It is a bad idea to dissolve filler extensively at the time of surgery if the patient no longer is aware of their native anatomy. It is better for them to have a few days to absorb that picture before blepharoplasty surgery. In some cases, the patient may insist on additional filler treatment to the tear trough, which may be reasonable as an interim treatment or if the surgeon can achieve a more favorable result.
The authors’ experience with patients presenting with dysmorphic filler is that a majority wish to pursue lower blepharoplasty and proceed with surgery after hyaluronidase treatment of the filler that they are aware is present (30%) or have filler unknown to them camouflaging their native anatomy and desire surgery, with hyaluronidase treatment performed preoperatively to unmask their native anatomy (30%). Approximately 75% of patients receive more than 1 hyaluronidase treatment. Skippen and colleagues report a series in which 92% of patients received a single hyaluronidase treatment; only 10% of patients proceeded with lower blepharoplasty after hyaluronidase treatment; and 80% of patients were treated again with HAG fillers after hyaluronidase treatment. Those investigators are an expert group whose capabilities are not questioned, but they appear to have a different patient type, practice pattern, and appropriate approaches to therapy.
- 9.
The authors’ preferred technique for tear trough treatment (see [CR] ) generally is as follows:
- a.
Upward fanning technique using a 25-gauge cannula advanced superiorly from a needle stab incision 3 cm below the orbital rim in line with the pupil
- b.
Initial infraorbital block with 0.3% lidocaine with epinephrine 1:600,000.
- c.
With the cannula deep on the periosteum and the side port up, small aliquots (0.02–0.03 mL) of Restylane-L are placed in the tear trough. As treatment proceeds, subtle tear trough elevation is apparent, arcing across the tear trough with multiple advancements.
- d.
Aiming for a modest undercorrection of the tear trough defect, because there is some swelling of the HAG after treatment, and overcorrection is undesirable.
- e.
A separate lateral treatment pocket may be undertaken for the temporal hollow, but careful treatment with small aliquots across the area is needed, and again undercorrection is advisable.
- f.
Some icing, short-term avoidance of blood thinners, and limited massage and activity for a day are advisable. Bruising generally is minimal with a 25-gauge cannula technique.
- a.
- 10.
Tear trough patients are followed-up at 1 week to 2 weeks, with additional treatment or touch-up as desired. The authors use half-inch 29-gauge to 30-gauge standard hypodermic needles for limited touch-up treatments.
Summary
Dermal fillers, in particular HAG fillers, are used in the treatment of aging changes in the periocular area, especially the tear trough. Filler treatment requires in-depth knowledge of specific issues relating to product performance and administration as well as knowledge of safety protocols and recognition and treatment of complications.
There are different approaches to treatment of the tear trough, with the authors favoring an approach from below, with a 25-gauge blunt cannula injecting Restylane-L as the preferred HAG. Prior filler treatment must be suspected in patients presenting for aesthetic evaluation, and the possibility of migration with a dysmorphic appearance and/or Tyndall effect appearance in the tear trough always is kept in mind. Treatment with hyaluronidase injection generally is effective in this area, although the aesthetic change may be drastic and a follow-up plan must be discussed with the patient before proceeding with this therapy.
Clinics care points
- •
Proper patient selection, product selection, and placement are paramount in achieving ideal results with dermal fillers.
- •
HAG fillers persist long term, even if the desired clinical effect dissipates, creating the frequently encountered Tyndall effect in the tear trough area.
- •
Inflammation in areas of filler injection must be treated as infectious in nature until proved otherwise.
- •
Hyaluronidase injection is a vital tool in treating many complications and overcorrection using HAG fillers
- •
Before hyaluronidase injection of the tear trough area, patient counseling must occur, discussing the patients’ options and planning if the patient finds recurrence of the tear trough to be aesthetically undesirable.
- •
An upward fanning injection technique with Restylane-L and a 25-gauge cannula is recommended by the authors in treating the tear trough area.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree