Chapter 5
Complications
5 Complications
5.1 Infection
5.1.1 Introduction
Infections are rare events in hernia surgery but are found with all types of operative treatments. In inguinal hernia surgery, infection rates up to 1.55% were observed in the first quality assurance study by the German North Rhine medical council for the years 1991–2000 (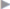 Table 5.1).35 By contrast, surgery of incisional hernia is known to have wound infection rates of up to 18%.13,38 With emergency hernia surgery, there is a substantial increase with infection rates of up to 50%.
Table 5.1).35 By contrast, surgery of incisional hernia is known to have wound infection rates of up to 18%.13,38 With emergency hernia surgery, there is a substantial increase with infection rates of up to 50%.
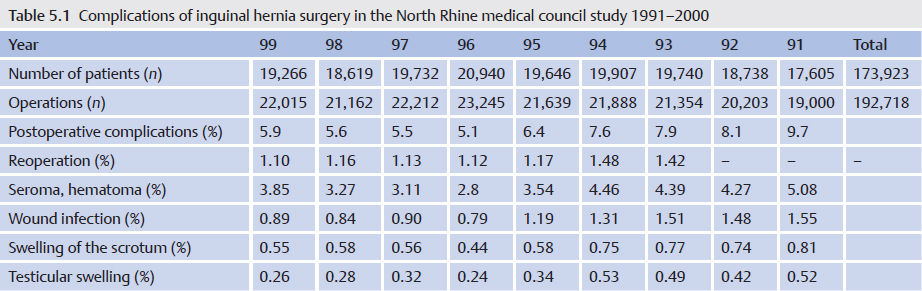
Even in hernia surgery, clean, clean contaminated, and septic procedures are distinguished. In clean procedures such as inguinal hernias, epigastric hernias, or spigelian hernias, wound infection rates of less than 1.5% can be expected under optimal conditions. Infection rates around 10% or more occur with surgery of umbilical or incisional hernias, which are regarded as potentially contaminated. Infection rates over 50% can be anticipated when the hernia surgery is performed acutely for incarceration with gangrene or an infected burst abdomen (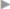 Fig. 5.1).
Fig. 5.1).
The cause of a wound infection in hernia surgery is often multifactorial, involving patient factors, surgical technique, and anesthesia.
It was shown in a meta-analysis that the hernia patient’s age and nutritional status, smoking history, overweight, and immune status have a significant influence on the rate of wound infections. Important aspects as regards the operation in general are the nature and duration of the operation, the care taken with skin antisepsis, including the question of when and how the skin is depilated, duration of preoperative hospitalization depending on the hernia type and choice of procedure, antibiotic prophylaxis, and insertion of wound drains.25 Moreover, in every procedure, meticulous operation technique plays a crucial part in preventing a wound infection. With regards to anesthesia, the oxygen partial pressure, body temperature, fluid balance, and early postoperative pain control have considerable significance.
5.1.2 Definition
Postoperative wound infections were defined in 1999 by the Centers for Disease Control and Prevention (CDC).42 A distinction is made between superficial wound infections, known as A1 or superficial surgical site infections SISSI and A2 or deep ISSI (DISSI).
• A1 infections are limited to the skin and subcutaneous tissue within the first 30 days after an operation.
• A2 infections are deep, subfascial infections within 30 days after operations without implants or within 1 year postoperatively when foreign material has been implanted.
• A3 infections do not play any part in hernia surgery. These refer to postoperative infections of organs and body cavities.
This generally accepted definition of postoperative wound infections can be criticized, as it inadequately describes the phenomenon of late infections after mesh implantation. For instance, bacterial colonization was observed several years after implantation on meshes that were not infected macroscopically, and some mesh infections with abscess formation only become apparent clinically 4 to 5 years after operation.
5.1.3 Infections in Inguinal Hernia Surgery
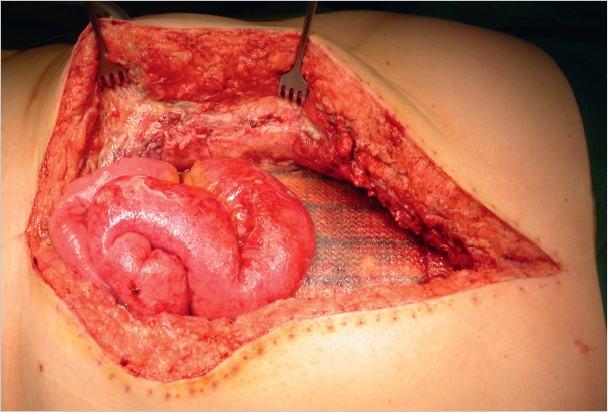
In patients without risk factors for postoperative wound infection, such as obesity, active smoking, age over 70 years, diabetes mellitus, or an otherwise compromised immune system, no significant differences are observed between open suture and mesh repairs in the case of an uncomplicated primary inguinal hernia operation. The infection rate is very low at 0 to 2%. Compared with the open procedures, the infection rate is even lower after endoscopic and laparoscopic repairs (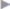 Table 5.2).
Table 5.2).
Against this background, the European Hernia Society (EHS) does not recommend antibiotic prophylaxis with inguinal hernia surgery in its guidelines.53 Patient-related risk factors or complicated repair of incarcerated hernia or recurrent hernia with the use of wound drains can be exceptions. In these patients, the wound infection rate is often more than 5% and can be reduced by a single-shot perioperative antibiotic.
5.1.4 Infections in Incisional Hernia Surgery
The rates of wound infections after incisional hernia repair reported in the literature vary greatly, from 0 to 18%. This is explained by the heterogeneity of the fascial defects and the variety of forms of repair. In general, postoperative wound infections occur much more often with open procedures as compared with laparoscopic repair.8,10,12 Possible causes of this phenomenon are the greater wound area and the greater risk of transferring skin microbes into deeper layers of the wound.
Among the open procedures, the lowest infection rates can be expected with a simple sutured repair. However, since sutured repair is indicated only for small fascial defects less than 1 to 2 cm in size, there are naturally no comparative studies of sutured and mesh repairs.
The mesh position in the abdominal wall also appears to play a part in the development of an infection. Wound and mesh infections occur more often with onlay repairs, where the mesh is placed between the fascia and the subcutaneous tissue, than after sublay operations where the mesh is in retromuscular position on the posterior layer of the rectus sheath. It is assumed that this is due to the greater frequency of seromas and poorer local perfusion after onlay repair.
5.1.5 Prevention of Wound Infections
Uncomplicated inguinal hernia surgery is not a recognized indication for antibiotic prophylaxis. On the other hand, prophylaxis should be considered in patients with typical risk factors (see above), incarceration, and complicated procedures when a drain is placed53 ( Fig. 5.2). Liberal use of antibiotic prophylaxis is recommended for surgery of incisional and umbilical hernias with open mesh insertion. If these operations are performed laparoscopically, however, antibiotic use does not have a significant effect, as the infection rate is very low anyway. Second- and third-generation cephalosporins are the prophylactic antibiotics of choice in hernia surgery, as they encompass the usual skin microorganisms. Erythromycin can be given as an alternative.22 It is important to give it at the correct time, 30 minutes before the skin incision, to ensure an adequate tissue level.57
Fig. 5.2). Liberal use of antibiotic prophylaxis is recommended for surgery of incisional and umbilical hernias with open mesh insertion. If these operations are performed laparoscopically, however, antibiotic use does not have a significant effect, as the infection rate is very low anyway. Second- and third-generation cephalosporins are the prophylactic antibiotics of choice in hernia surgery, as they encompass the usual skin microorganisms. Erythromycin can be given as an alternative.22 It is important to give it at the correct time, 30 minutes before the skin incision, to ensure an adequate tissue level.57
Anesthesia-influenced factors such as body temperature, oxygen partial pressure, fluid balance, and early postoperative analgesia are important, especially when incisional hernia repair is prolonged and involves large wound areas. The aim of all measures must be maintenance of good tissue perfusion with high partial pressure of oxygen. Correct intraoperative and early postoperative fluid balance at all times is ideal, without hypovolemic peripheral vasoconstriction on the one hand or local edema due to “overhydration” on the other hand.28 It is known from colorectal surgery that intraoperative elevation of oxygen delivery to 80% and/or immediate postoperative noninvasive ventilation with concentrated oxygen can reduce the wound infection rate to half.27 There are no corresponding studies on hernia surgery.
From the surgical aspect, a meticulous operation technique with as little tissue trauma as possible provides the foundation for infection-free wound healing. In addition, in open incisional hernia surgery, removal of poorly perfused scar tissue from the subcutaneous space and fascial margins is recommended. The subcutaneous parts of the peritoneal hernial sac should not be left behind as extensive seromas may occur, which later become infected. The mesh must be handled with particular care. It should be removed from its sterile packaging only immediately prior to implantation and after a change of gloves and, if appropriate, repeat disinfection of the operation field, avoiding skin contact. Wound drains are potential portals of infection. If they are regarded as essential, they should preferably not be placed in direct contact with the mesh.
Postoperative seromas at the mesh site are frequent within the first 4 to 6 weeks after the operation; they require treatment only if symptomatic.
5.1.6 Treatment of Wound and Mesh Infections in Hernia Surgery
An infection that occurs in the context of hernia surgery is defined as an emergency. It should be treated promptly, as sepsis may supervene in rare cases, especially after incisional hernia surgery, which can lead to fatal complications.
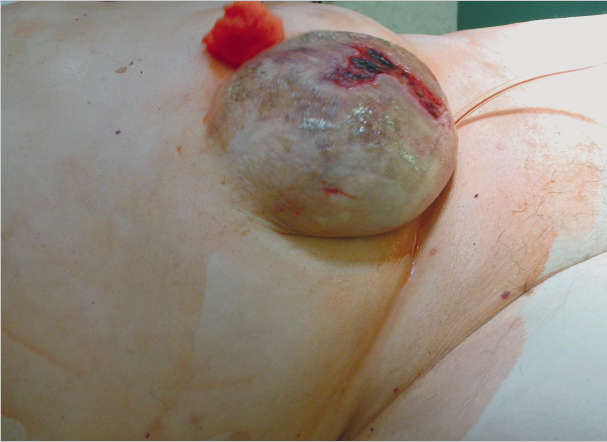
Subcutaneous infections are treated with the usual measures of debridement and open wound treatment or vacuum therapy, always checking whether there is a contact with the mesh (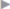 Fig. 5.3). If the implant is involved, the type of material and the timing of the infection essentially determine whether the mesh can be left or should be explanted. An attempt to keep it is always justified with early postoperative infections of large-pore monofilament implants. By contrast, colonization of microporous films (e.g., expanded polytetrafluoroethylene [ePTFE]) can rarely be eliminated and these must usually be removed. Antibiotic therapy alone is usually insufficient. Biofilm-forming microorganisms on alloplastic materials, in particular, are largely resistant to systemic antibiotic therapy. Meshes with metal coatings such as gold or titanium appear to counteract colonization with biofilm producers. If the time of onset and the implanted material allow an attempt at preservation, vacuum therapy for 2 to 3 weeks, possibly accompanied by systemic antibiotic therapy, is often successful.19
Fig. 5.3). If the implant is involved, the type of material and the timing of the infection essentially determine whether the mesh can be left or should be explanted. An attempt to keep it is always justified with early postoperative infections of large-pore monofilament implants. By contrast, colonization of microporous films (e.g., expanded polytetrafluoroethylene [ePTFE]) can rarely be eliminated and these must usually be removed. Antibiotic therapy alone is usually insufficient. Biofilm-forming microorganisms on alloplastic materials, in particular, are largely resistant to systemic antibiotic therapy. Meshes with metal coatings such as gold or titanium appear to counteract colonization with biofilm producers. If the time of onset and the implanted material allow an attempt at preservation, vacuum therapy for 2 to 3 weeks, possibly accompanied by systemic antibiotic therapy, is often successful.19
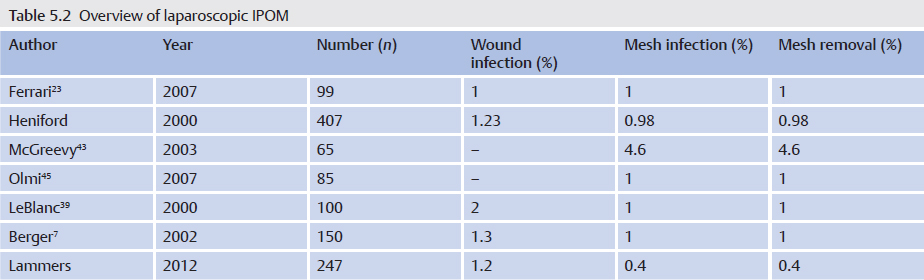
With late infections, which can manifest clinically after months and years, mesh explantation is usually unavoidable. Often there is a thick abscess capsule that cannot be treated and must be removed with the mesh. The same applies for fistulas to hollow organs such as the bowel and bladder. In these cases, the clinical picture is of a septic abdominal wall with an extensive phlegmon (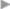 Fig. 5.4).
Fig. 5.4).
The diagnosis can be difficult as the association with a mesh inserted years ago is sometimes not apparent. The first step in treatment consists of mesh removal and treatment of the fistula, preferably by suturing the hernia defect or covering it with a rapidly absorbable mesh (e.g., Vicryl mesh). The incisional hernia can then be scheduled for repeat alloplastic repair 12 months later. Alternatively, “biological” mesh can be used; primary implantation is possible even in contaminated wound areas.
If the clinical picture is of sepsis, the Tarragona strategy applies to the treatment of a wound infection following hernia repair (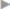 Table 5.3).
Table 5.3).
Table 5.3 Tarragona strategy for antibiotic treatment of sepsis (within 1 h)
Patient’s individual risk factors | “Look at your patient” |
Local epidemiology | “Look at your hospital” |
Broad initial antibiotic administration | “Hit hard and early” |
Which bacteria may be present? | “Get to the point” |
Reevaluation within 48–72 h | “Focus?” |
Fig. 5.4 Recurrent skin fistulas after mesh implantation (polyester) to repair an incisional hernia.
5.2 Incarceration
Incarceration (cancer in Latin = enclosure, prison) signifies the entrapment of tissue in the hernia. Every hernia can become incarcerated in principle, regardless of its size and location. An incarcerated hernia is always an emergency, as it may strangulate, when the blood supply to its contents is cut off. “The sun must not rise or set on an incarcerated hernia.”
An irreducible hernia should be distinguished from an incarcerated one. Unlike an incarcerated hernia, an irreducible hernia can be asymptomatic and present for a long time. An irreducible hernia is an urgent but not an emergency situation.
5.2.1 Etiology
By definition, the orifice of a hernia is a narrow opening in the abdominal wall. Tissue that slides through the hernial opening can swell as a result of impaired venous drainage and can then no longer slide back into the abdomen. The swelling can also interfere with the blood supply and the incarcerated tissue can become ischemic or strangulated. This vicious circle ultimately leads to tissue necrosis. Incarceration of small intestine with its short ischemia time is therefore a particular risk with incarcerated hernia and requires rapid treatment.
The Littré-Richter hernia is a special form of incarcerated hernia in which only part of the bowel wall is incarcerated; so, clinical presentation is often late in the form of bowel wall necrosis with peritonitis. The causes for the sliding of tissue into the hernia sac can be sudden rise in intra-abdominal pressure (coughing, sneezing) and also increased peristalsis or meteorism. There is an obvious association between the size of the hernial orifice and the risk of incarceration.
The hernia can contain a variety of tissues:
• Preperitoneal fat.
• Greater omentum.
• Small or large intestine.
• Abdominal organs.
5.2.2 Clinical Features
An incarcerated hernia usually presents clinically as an acute condition, classically with the symptom triad of:
• Pain.
• Swelling.
• Irreducibility.
If there is also inflammatory erythema over the hernia, immediate surgery is indicated. Nausea and vomiting can indicate intestinal obstruction and should be further investigated. Other clinical signs indicating the need for immediate operation are overt intestinal obstruction and an acute abdomen with signs of peritonitis.
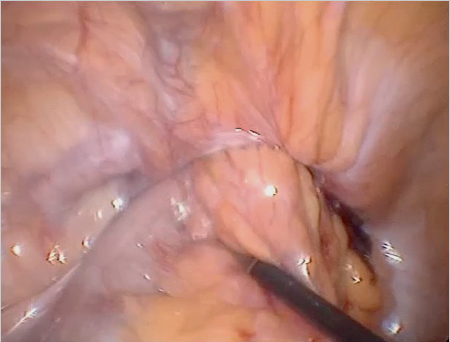
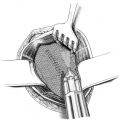
If these clinical signs are absent, the diagnosis can sometimes be difficult. Femoral hernias in women, with an incarceration rate of up to 30% at diagnosis, are often not easy to identify clinically. Internal hernias, too, are detected clinically more from the secondary symptoms such as obstruction and acute abdomen (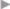 Fig. 5.5).
Fig. 5.5).
5.2.3 Diagnosis
The diagnosis of an incarcerated hernia is typically clinical. However, there are forms of hernia (femoral hernia, internal hernia, the Littré hernia, spigelian hernia), where additional diagnostic aids may be required. When the situation is unclear, ultrasonography or an abdominal CT may be helpful in making the diagnosis. Since the vitality of bowel in an incarcerated hernia cannot be determined clinically, operation should be resorted to readily.
5.2.4 Treatment
Immediate surgery is indicated in all cases where the incarcerated hernia is associated with an acute abdomen or obstruction. A closed attempt at reduction of an incarcerated hernia can be made when these secondary signs are absent and when it has been irreducible for only a short time. As far as possible, reduction should be attempted by an experienced doctor with appropriate analgesia. Spasmolytic agents may be used in addition.
Fig. 5.5 Incarcerated inguinal hernia (laparoscopic view, with the kind permission of Dr René H. Fortelny, Wilhelminenspital, department of general, visceral and tumor surgery, Vienna, Austria).
The patient should be as relaxed as possible. The doctor uses both hands to prevent the hernia from slipping away and to achieve even pressure on it as far as possible. A cautious reduction attempt can then be made under steady pressure. If this is unsuccessful, operation is indicated. If hernia reduction is successful, the patient should be admitted to hospital and observed, and the hernia should be repaired surgically immediately. Reduction of the complete hernia is called en bloc reduction. Reduction of the hernia by tearing the hernial ring is called pseudotaxis. If either of these events takes place, the actual problem of bowel entrapment has not been eliminated, and the symptoms persist so that the intestinal ischemia progresses.
When surgical reduction of the hernia is indicated, this should be done promptly. When choosing the procedure, the hernia contents must always be assessed. When an incarcerated inguinal hernia is treated under general anesthesia, for example, spontaneous reduction may occur, and in this case, the entire small intestine must be examined for vitality. It is not sufficient simply to repair the hernia.
As regards open versus laparoscopic hernia repair, there is no generalized recommendation. If the treating institution has the appropriate expertise in laparoscopy, the procedure can be performed laparoscopically or endoscopically.14,24,58 Moreover, there are no clear recommendations on whether to repair the hernia with a mesh. However, there do not appear to be any contraindications to the use of polypropylene mesh.6,9,17,21,44
5.2.5 Incarceration Rates and Risk
It is not possible to make a general statement about the risk of hernia incarceration. Many factors play a role in incarceration, including hernia location, hernial orifice size, nutritional status, and the patient’s age and sex. An incarceration rate of 0.3 to 3% per year is reported for inguinal hernia. Indirect inguinal hernias become incarcerated about 10 times more often than direct hernias.26,52 After adhesions, hernias appear to be the most frequent cause of small bowel obstruction.29
An eventual incarceration rate of up to 30% is reported for umbilical hernias.51 There is little information in the literature for ventral or incisional hernia but the rate of incarceration is markedly increased at a body mass index (BMI) of over 40 kg/m.2,37 The Herniamed database currently describes 764 incarcerations with 19,518 recorded incisional hernias (3.9%).
5.3 Postoperative Pain
Postoperative pain after a hernia operation is classified as acute wound pain and chronic postoperative pain persisting beyond 3 months.
5.3.1 Acute Postoperative Pain
Acute postoperative pain differs according to:
• The type of repaired hernia.
• The access route.
• The operation technique.
In inguinal and femoral hernia surgeries, acute postoperative pain is greatly determined by the access route. Pain along the course of the incisions predominates after open transinguinal and preperitoneal procedures, whereas pain is reported in the area of the dissected mesh site following endoscopic and laparoscopic operations. These symptoms are mild and are often perceived more as a type of extensive “hypersensitivity” than as wound pain. Overall, symptoms after endoscopic inguinal and femoral operations are less in the first 3 to 5 days than after open operations. The differences disappear after this time period.
Major differences in pain perception within the first 3 to 6 hours after the operation are also found with different methods of anesthesia. More patients are pain-free up to 8 hours postoperatively after open repair under local anesthesia with long-acting anesthetics such as ropivacaine.46 Significantly less pain is reported within the first 3 to 4 hours postoperatively after operations performed under spinal anesthesia compared with procedures under general anesthesia with laryngeal mask or endotracheal intubation.31
Treatment of acute postoperative pain begins during the operation (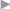 Table 5.4). In open surgery of inguinal and femoral hernias, field block of the ilioinguinal and iliohypogastric nerves close to the anterior superior iliac spine and wound edge infiltration at the conclusion of the operation provide effective analgesia for up to 8 hours postoperatively. Long-acting drugs, such as bupivacaine or ropivacaine, in a concentration of 0.5 to 1% are used. Lower concentrations are ineffective. Continued infiltration treatment is useful only until the first postoperative day. Symptoms subsequently are usually so mild that patients no longer benefit from further infiltration.31
Table 5.4). In open surgery of inguinal and femoral hernias, field block of the ilioinguinal and iliohypogastric nerves close to the anterior superior iliac spine and wound edge infiltration at the conclusion of the operation provide effective analgesia for up to 8 hours postoperatively. Long-acting drugs, such as bupivacaine or ropivacaine, in a concentration of 0.5 to 1% are used. Lower concentrations are ineffective. Continued infiltration treatment is useful only until the first postoperative day. Symptoms subsequently are usually so mild that patients no longer benefit from further infiltration.31
Table 5.4 Recommendations for perioperative pain management after inguinal hernia repair in adults
Phase | Measures |
Pre- and intraoperative | Local anesthesia with long-acting drugs (e.g., ropivacaine) Field blocks of the ilioinguinal/iliohypogastric nerves Wound edge infiltration |
Early postoperative (6–24 h) | Fixed schedule: NSAID/COX-2 inhibitor |
Postoperative (> 24 h) | Fixed schedule: NSAID/COX-2 inhibitor |
Abbreviations: COX-2, cyclooxygenase-2; NSAID, nonsteroidal anti-inflammatory drug. | |
Oral nonsteroidal anti-inflammatory drugs (NSAID) or cyclooxygenase-2 (COX-2) inhibitors are usually suitable for further analgesia. Paracetamol (acetaminophen) alone is often insufficient and should be combined with an NSAID/COX-2 inhibitor. Medication on a fixed schedule is recommended for the first 2 days after the operation, and it is then taken as needed (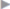 Table 5.4). Nonsteroidal anti-inflammatory drugs and paracetamol are ineffective for treatment of early postoperative neuropathic pain and symptoms should be managed with, for example, tramadol or oxycodone (
Table 5.4). Nonsteroidal anti-inflammatory drugs and paracetamol are ineffective for treatment of early postoperative neuropathic pain and symptoms should be managed with, for example, tramadol or oxycodone (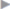 Table 5.4).
Table 5.4).
Early postoperative pain after endoscopic or laparoscopic procedures and all other hernia repairs of the abdominal wall can be reduced by wound edge infiltration.
Extensive incisional hernia repair, especially above the umbilicus, produces considerable pain and corresponding interference with the accessory respiratory muscles. The risk of pulmonary complications is further increased by postoperative atony after the adhesiolysis that is usually necessary. In this situation, patients benefit from an epidural catheter inserted preoperatively. As a result of the very effective analgesia, pulmonary complications are observed less often and the goal of early mobilization is supported.
Giving 4 to 8 mg of dexamethasone is another effective measure in the multimodal concept for ensuring immediate postoperative mobilization. A single dose of dexamethasone with the premedication immediately before the start of the operation improves general postoperative wellbeing and significantly lowers the risk of nausea, vomiting, dizziness, and fatigue. Medication with NSAID/COX-2 inhibitors on a fixed schedule, possibly in combination with paracetamol, can be used for analgesia after the first 2 to 3 postoperative days.
5.3.2 Chronic Postoperative Pain after Inguinal and Femoral Hernia Operation
Chronic postoperative pain refers to the pain that persists beyond a period of 3 months postoperatively. Depending on the definition of pain intensity, up to 60% of patients are affected after inguinal hernia repair. A severe and disabling pain syndrome occurs in 2 to 6% of cases. Affected patients suffer from permanent or frequently recurring pain that requires treatment and interferes with their daily activities. The outcomes in about half of these patients include incapacity for work and depression.
From experience to date, endoscopic and laparoscopic procedures are involved more rarely than open sutured or mesh repairs.20 However, chronic pain was described in up to 13.8% of patients after TEP or TAPP repair.18 Patient-related risk factors for developing a disabling inguinal pain syndrome were often identified as surgery of recurrence through the same access,32 age under 65 years,54 a pessimistic preoperative attitude to the operation,47 severe pain immediately postoperatively,54 and female sex. As regards the operation technique, failure to expose potentially affected nerves, use of small-pored and heavyweight mesh,56 and certain mesh fixation techniques with tacks and staples were shown to be detrimental.
There are extensive data on the chronic pain syndrome especially after Lichtenstein repair. It was shown that chronic pain correlates with failure to identify nerves at operation.2 Only 3 minutes are required for specific nerve exposure and this has an average success rate of 75%.36 Direct contact between nerves mobilized from their enveloping fascia and suture and mesh materials leads to irreversible damage to the myelin sheath and axon.16 Accordingly, an operation technique that includes ample neurolysis, preferably with nerve preservation, predisposes more to chronic pain.49 Nerves that cannot be moved from the operation field with their enveloping fascia should be resected.30 The same applies for nerves that are damaged intraoperatively. In case of doubt, resection should be preferred to an attempt at preservation.1
As regards mesh fixation, fibrin glue and self-adhesive grafts for Lichtenstein repair were associated with a lower rate of chronic pain syndrome than classic sutured fixation.11,48 The recommendations for preventing chronic groin pain are:
• Awareness of the patient’s risk factors, such as age, and sex.
• Alternative access route for surgery of recurrence: endoscopic after open operation and vice versa.
• Use of lightweight large-pored mesh.
• Reduction of mesh fixation to a minimum, possibly no mesh fixation in TEP, possibly mesh gluing in the Lichtenstein repair or loosely suturing the mesh cranially.
• Identification of the nerves.
• Avoidance of direct mesh-nerve contact.
• Facultative pragmatic neurectomy.
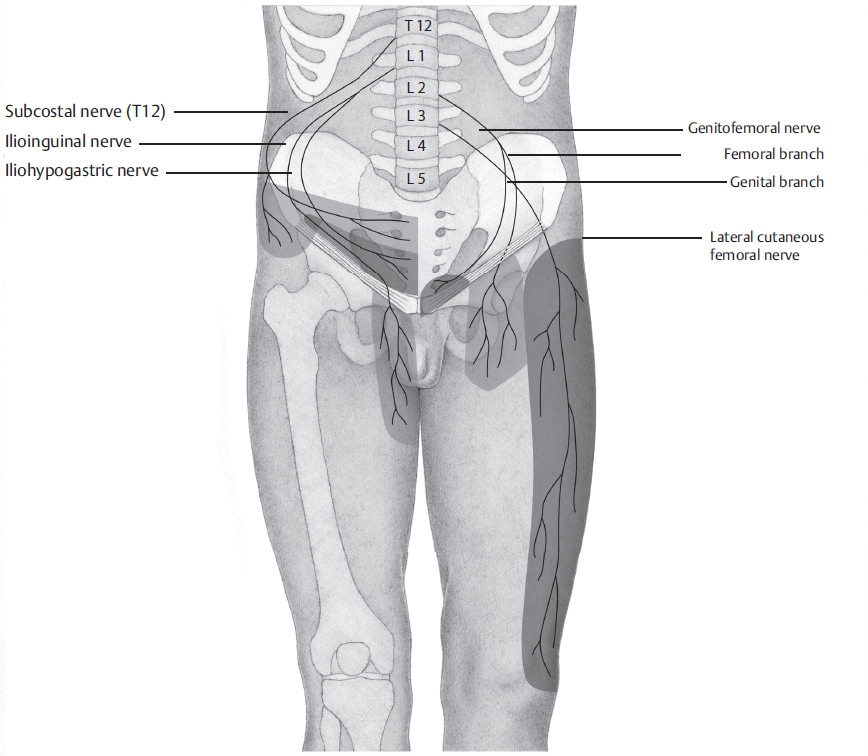
The treatment of chronic groin pain comprises a number of methods. An attempt should always be made first to identify the possible cause of the pain. The character of the pain may provide clues. In theory, neuropathic pain as evidence for a nerve injury is distinguished from somatic pain (caused by staples, sutures, or hernia recurrence) and from visceral pain (testicular pain, painful ejaculation). In practice, these distinctions are often not possible from the different pain character alone. It is worthwhile, however, to distinguish neuropathic pain after nerve injury from non-neuropathic or nociceptive pain as the treatment approaches differ. Immediately after injury, the affected nerve can be distinguished by its typical distribution pattern (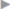 Fig. 5.6). As a result of the peripheral nerve injury, however, structural changes take place in the nervous system after a short time, which can be summarized as peripheral and central sensitization (
Fig. 5.6). As a result of the peripheral nerve injury, however, structural changes take place in the nervous system after a short time, which can be summarized as peripheral and central sensitization (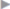 Table 5.5). This influences both the distribution pattern and the pain character.
Table 5.5). This influences both the distribution pattern and the pain character.