Chapter 3
Special Hernias
3 Special Hernias
3.1 Female Hernias
Only about 5 to 8% of all hernias in the inguinal region occur in women;5,48,77 approximately 20 to 30% are femoral hernias and the other two-thirds are predominantly indirect inguinal hernias. Direct inguinal hernias are rare in women.127 In major clinical studies, direct inguinal hernias amount to only a little over 10%. Unlike indirect hernias, the incidence of bilateral direct inguinal hernia is comparatively higher in women at 40 to 50%.48
The incidence and distribution of female groin hernias differ completely from male hernias, primarily because of the anatomical differences. The female pelvis is shallower and wider and the area of the muscle-free Hesselbach triangle is correspondingly smaller. Moreover, the structures that pass through the inguinal canal are rather narrow and string-like. These factors make women less prone to develop an inguinal hernia but promote the development of femoral hernias. Although direct inguinal hernias are rare, when they occur, they are very frequently bilateral, indicating a systemic disorder, e.g., collagen metabolism.
Data regarding the timing of operation in pregnant women are very sparse. There are no publications about a particular risk of incarceration during pregnancy or problems during childbirth. In advanced pregnancy, the enlarged uterus covers the hernial orifice and so possibly protects against incarceration. The current recommendation is to observe a hernia diagnosed during pregnancy and perform elective surgery following delivery.13
3.1.1 Operation Tactics
The choice of access and operative procedure for female hernias in the inguinal region is currently undecided. Good results with long-term recurrence rates of 2.5 to 4% up to 10 years have been described for open transinguinal repair of a groin hernia, using both sutured and mesh methods.5,122 Recurrent hernias, especially when observed in the early postoperative period, are found much more often in the femoral region in women. This suggests that they are not genuine recurrent hernias but rather an initially overlooked primary femoral hernia. In a clinical study of the groin region, about 20% of femoral hernias were overlooked.102 In addition to clinical examination, ultrasonography can help to distinguish preoperatively between an indirect inguinal and a femoral hernia and should therefore be employed readily. Moreover, intraoperative assessment by palpation of the femoral compartment in women is an essential component of a successful open operation.
If no evidence for a femoral hernia is found during surgery of an indirect inguinal hernia, a sutured repair with complete closure of the deep inguinal ring can be used. When an additional defect in the femoral compartment is found, TIPP repair (See section  2.3.2) or a modification of the Lichtenstein repair is the option during open surgery. Mesh repair should be the standard treatment of the rare direct female inguinal hernia. All potential hernial orifices are treated by endoscopic TEP repair, thus eliminating the problem of an overlooked femoral hernia. This argument is used to promote TEP as the most suitable method of inguinal hernia repair in women.102 However, mesh repair is often not necessary for an isolated indirect inguinal hernia, especially in younger women,122 and can therefore be regarded as “overtreatment.”
2.3.2) or a modification of the Lichtenstein repair is the option during open surgery. Mesh repair should be the standard treatment of the rare direct female inguinal hernia. All potential hernial orifices are treated by endoscopic TEP repair, thus eliminating the problem of an overlooked femoral hernia. This argument is used to promote TEP as the most suitable method of inguinal hernia repair in women.102 However, mesh repair is often not necessary for an isolated indirect inguinal hernia, especially in younger women,122 and can therefore be regarded as “overtreatment.”
3.1.2 Operation Techniques
The operation technique differs from the procedure in men as round ligament of the uterus is in the inguinal canal instead of the spermatic cord. Injuries of the round ligament are not dangerous and it can be divided and resected without significant consequences.
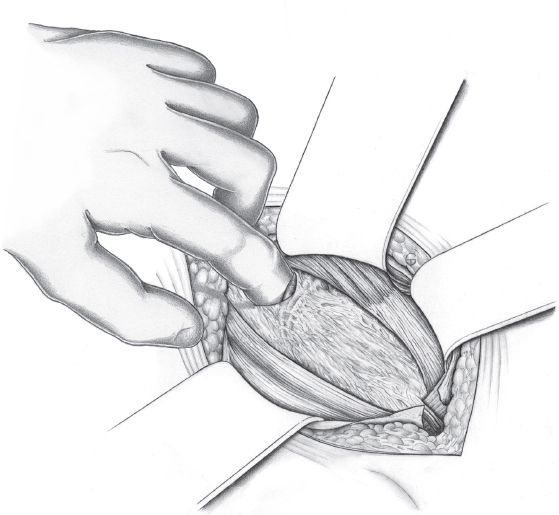
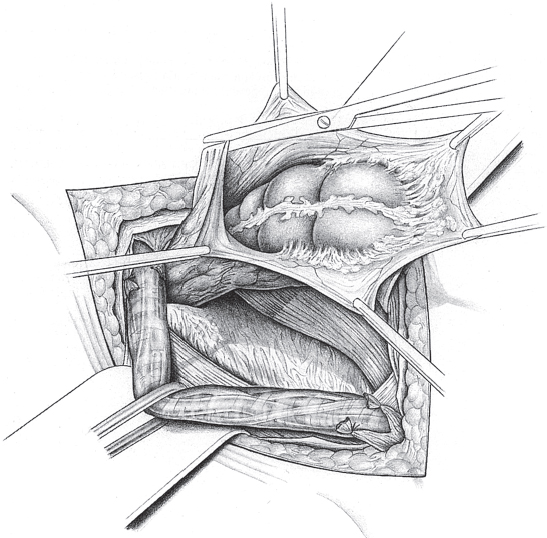
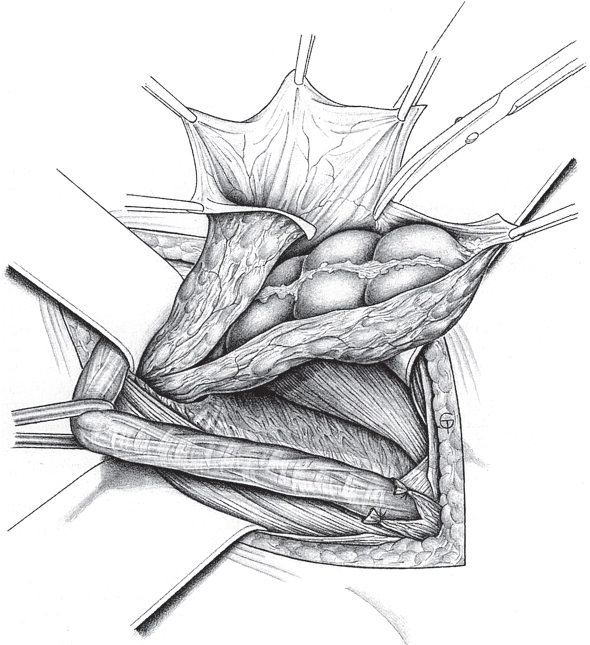
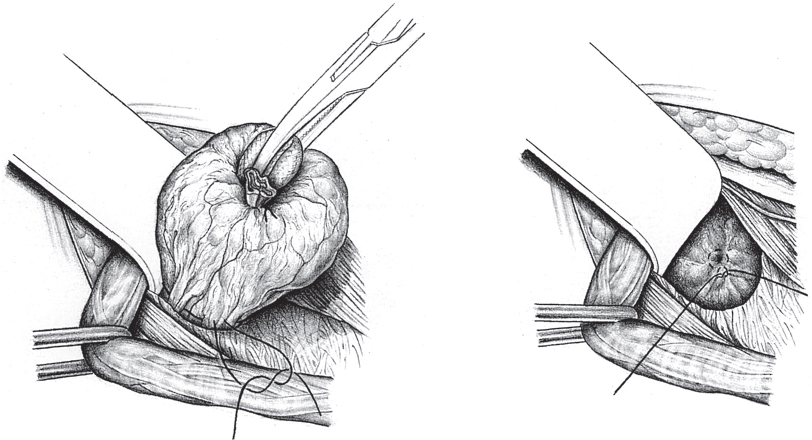
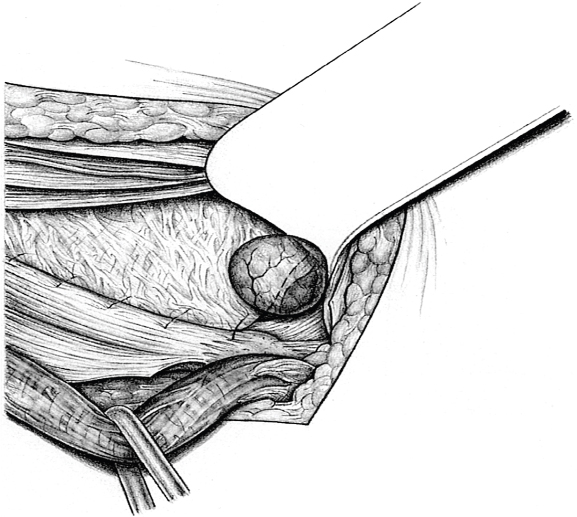
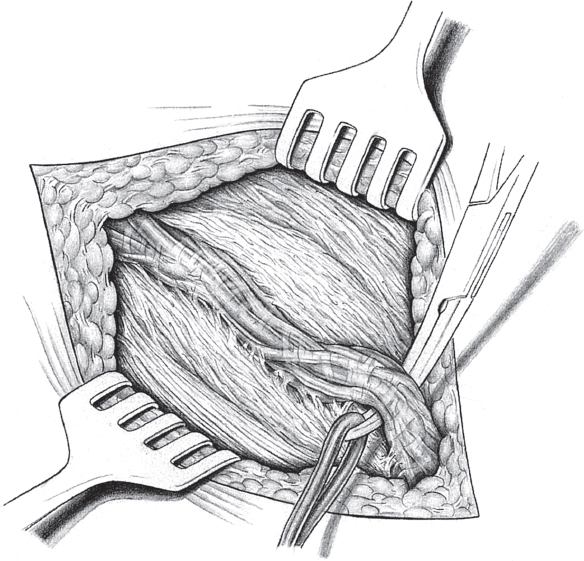
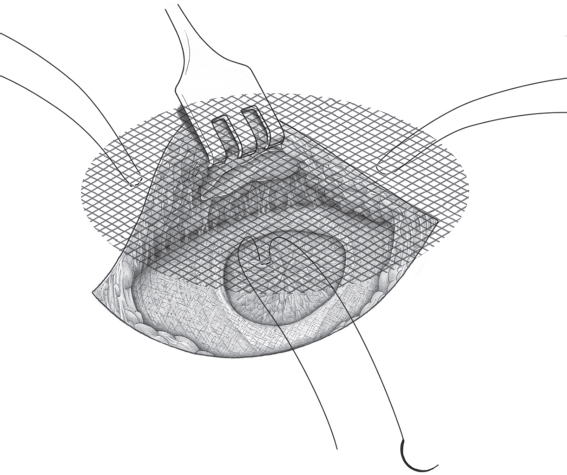
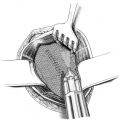
When the hernia is indirect, the sac is treated and the femoral compartment is then explored digitally through the expanded deep inguinal ring ( Fig. 3.1).
Fig. 3.1).
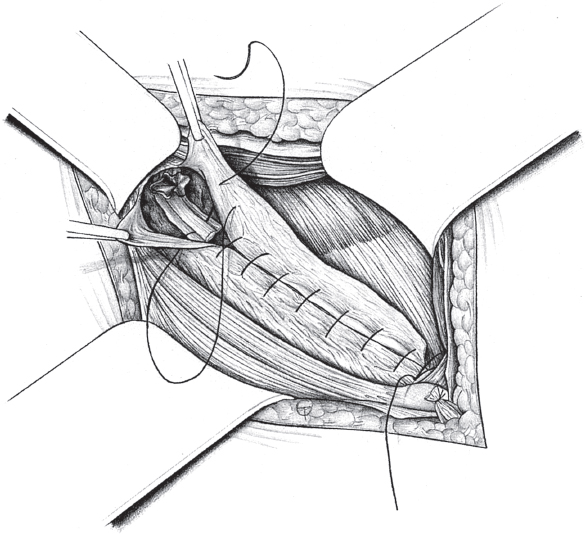
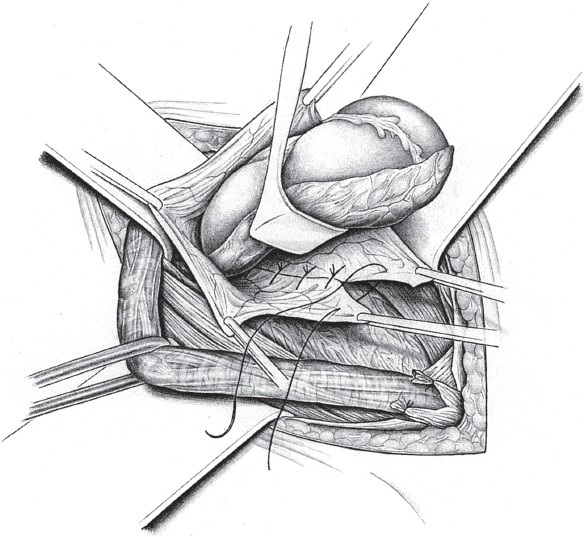
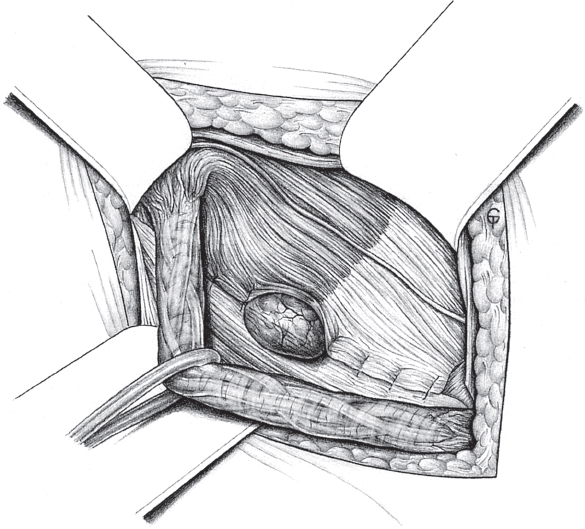
Sutured repair, e.g., by the Shouldice method (see section  2.2.2.4), can then be carried out, especially in younger patients. Unlike in men, the deep inguinal ring is closed completely. The proximal stump of the round ligament can be included in the lateral part of the Shouldice suture (
2.2.2.4), can then be carried out, especially in younger patients. Unlike in men, the deep inguinal ring is closed completely. The proximal stump of the round ligament can be included in the lateral part of the Shouldice suture (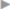 Fig. 3.2).
Fig. 3.2).
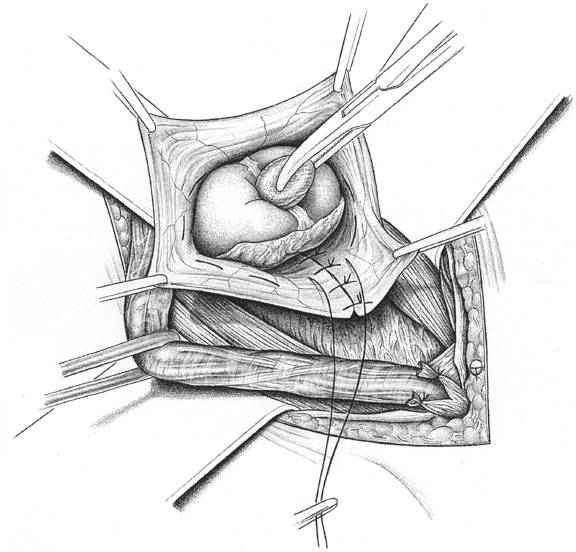
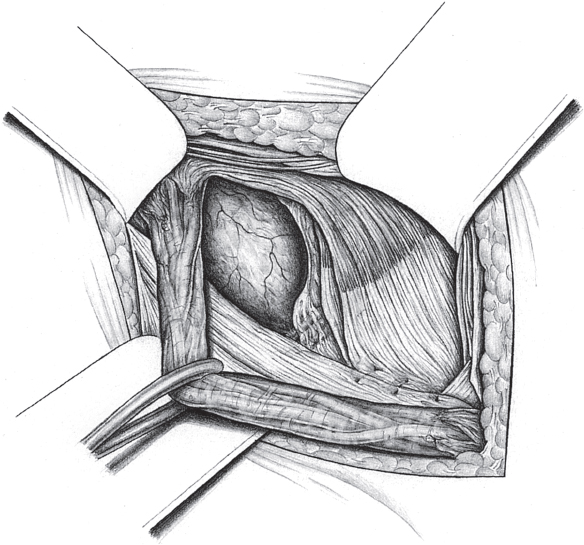
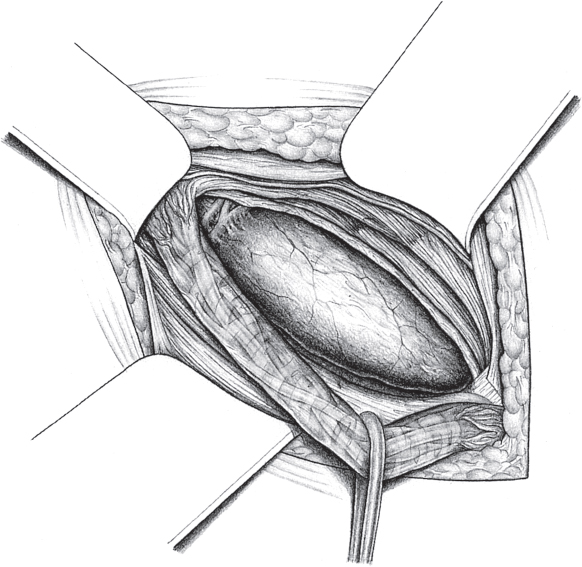
When the hernia is direct, the posterior wall of the inguinal canal may have to be incised to allow palpation of the femoral orifice. Mesh repair is recommended, e.g., with the Lichtenstein procedure. Unlike the operation in men (see section 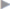 2.3.1), the mesh is not slit laterally.
2.3.1), the mesh is not slit laterally.
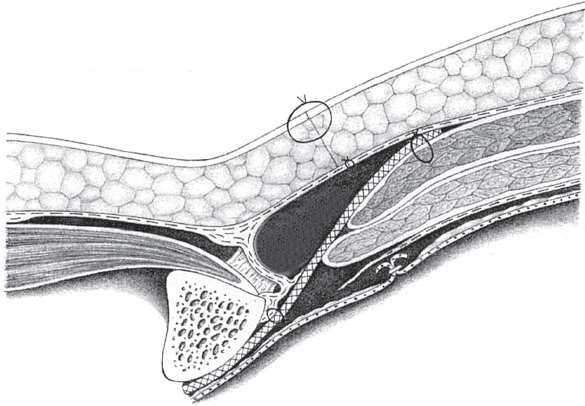
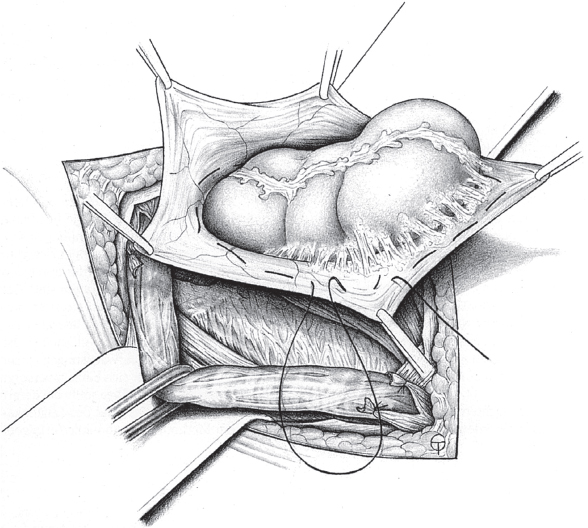
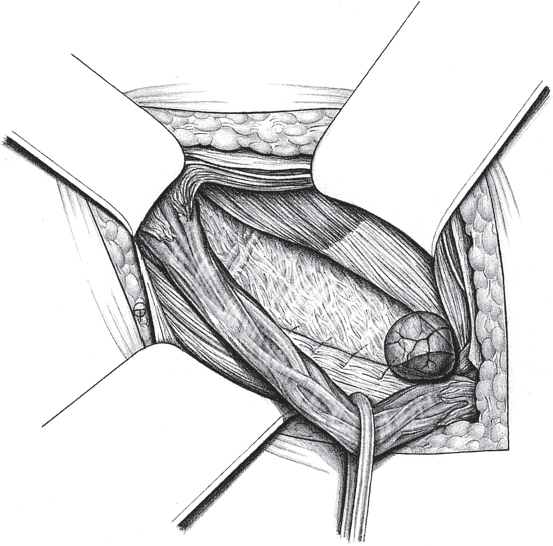
If a femoral hernia is found in addition during open surgery, TIPP repair can be used without slitting the mesh laterally (see section 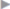 2.3.2) or the Lichtenstein repair can be modified by fixing the medial part of the mesh to the Cooper ligament rather than to the inguinal ligament (
2.3.2) or the Lichtenstein repair can be modified by fixing the medial part of the mesh to the Cooper ligament rather than to the inguinal ligament (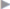 Fig. 3.3). Suture of the mesh starts as usual over the pubic bone, without picking up the periosteum. From there, the suture passes from medial to lateral between the Cooper ligament and the inferior border of the mesh as far as the femoral vein. The suture ascends here to the base of the inguinal ligament and continues beyond the deep inguinal ring, as in men. The mesh is fixed cranially by the usual method of tying it loosely to the internal oblique muscle. At the deep inguinal ring, the proximal stump of the round ligament can be fixed to the mesh.
Fig. 3.3). Suture of the mesh starts as usual over the pubic bone, without picking up the periosteum. From there, the suture passes from medial to lateral between the Cooper ligament and the inferior border of the mesh as far as the femoral vein. The suture ascends here to the base of the inguinal ligament and continues beyond the deep inguinal ring, as in men. The mesh is fixed cranially by the usual method of tying it loosely to the internal oblique muscle. At the deep inguinal ring, the proximal stump of the round ligament can be fixed to the mesh.
The operative technique of laparoscopic (TAPP) or endoscopic (TEP) inguinal hernia repair in women does not differ from that in men (see section  2.4.1 and
2.4.1 and  2.4.2). In case of doubt, the round ligament can be divided at the level of the deep inguinal ring to optimize mesh position.
2.4.2). In case of doubt, the round ligament can be divided at the level of the deep inguinal ring to optimize mesh position.
Fig. 3.2 The Shouldice repair of an indirect inguinal hernia in women. The round ligament is included in the lateral region of the suture.
Fig. 3.3 Lichtenstein repair of a combined hernia with a femoral hernial orifice. Schematic cross-section of mesh position medial to the femoral vein.
3.2 Sportsman’s Groin
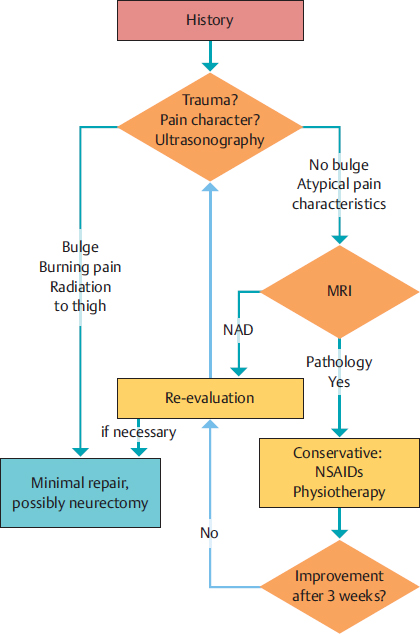
Sportsman’s groin (sportsman’s hernia) is an interesting condition common in top athletes, which has received much attention only in the past 10 years. It is defined by chronic and usually exercise-related pain in the groin region. In our routine practice, we prefer the term sportsman’s groin to sportsman’s hernia, because patients with this syndrome rarely have a genuine hernia but rather have weakness of the posterior wall of the inguinal canal with nerve irritation. The term “soft groin” is often used to describe this condition but this rather general diagnosis does not adequately reflect the complexity of the disorder. This section presents the typical symptoms, the required diagnostics, and recommended treatment of sportsman’s groin.
Pain in the groin region is not rare; the annual incidence reported in active footballers is 8 to 15%.26 This is easily understandable because of the stress on this region of the body, especially in athletes. The muscles of the anterior abdominal wall play a key part in stabilizing the pelvis. Considerable forces act on muscle origin and insertion sites during eccentric rotating movements, running and sprinting, and on impact and rotation of the upper body. In addition, the multilayered nature of the matrix of muscles and tendons causes these structures to slide over each other. Important sensory nerves, the ilioinguinal nerve and the genital branch of the genitofemoral nerve, pass through this sliding matrix. The abductors and adductors of the thigh act as antagonists of the abdominal muscles in stabilizing the pelvis and must always be considered when the cause of groin pain is sought, especially in athletes. The pubic bone, where the abdominal muscles are inserted, is especially important.
This is not a classic inguinal hernia but rather a circumscribed weakness of the posterior wall of the inguinal canal, which leads to circumscribed protrusion. Since there is no hernia, the term “sportsman’s hernia” or “sports hernia” should be avoided. The leading problem resulting from this site of lower resistance is sudden irritation of the genital branch of the genitofemoral nerve, which then becomes chronic. The typical pain is burning, stabbing, and/or radiating to the inner thigh, testis, and scrotum, and also to the back. The circumscribed expansion of the inguinal canal also leads to medial and cranial retraction of the rectus muscle. This causes increased tension on the pubic bone with a sustained increase in stress on the fan-like muscle insertion, resulting in sometimes burning pain in the pubic bone, known as “osteitis pubis” or “pubalgia.” This painful stimulus produces reflex tension of the rectus and therefore further tension on the pubic bone. The dull pain at rest and sudden shooting pains with exercise are very troublesome for top athletes and can drastically reduce their performance. There is also a mental component. By trying to avoid the symptoms, the athlete is much more cautious and limited in his mobility and radius of action. Consequently, his form falls off.
Besides the acute and readily explained pain in the groin area, further differential diagnoses should be considered when the details of the pain are less obvious.41
3.2.1 History
Knowing the history is the first important step in establishing the correct diagnosis. Essential points in the history are the time of onset of the groin pain, prior trauma, or wrong movements during training, and the pediatric history in case of any previous damage, such as the Perthes disease or hip dysplasia.
In the trauma history, a distinction should be made between impact injury, extreme movements (shooting at goal, skiing injury etc.), dislocations, and chronic overuse.
For example, the type of sport can provide important evidence about the nature of the strain. Golfers are exposed to rotation of the pelvis with the leg fixed, skiers are more at risk of sudden extreme movements or collisions, and footballers are subject to recurrent twisting and shooting stresses and stresses on the adductor muscles.
The characteristics of pain can help lead to the right diagnosis:
• Is the pain load-dependent?
• Is it unilateral or bilateral? Can the pain be provoked?
• Does it occur when the patient is lying or standing?
A typical and therefore indicative pain as the cause of sportsman’s groin has a burning and sometimes sporadic character radiating to the inside of the thigh, into the scrotum, and to the back.
Infiltration of a local anesthetic at the pain point may sometimes help to elucidate the cause of an insertion tendinopathy.
3.2.2 Diagnosis
Clinical Examination
Operation must always be preceded by an accurate record of the symptoms. Athletes typically report a sudden, shooting, sharp, and sometimes burning pain in the groin region, radiating to the inside of the thigh, perineum, and scrotum. The typically unilateral pain can be provoked by sit-ups and sudden movement with torsion of the hip region. It is particularly typical that the pain sensation also occurs with the same intensity after adequate rest. Examination reveals a tender inguinal canal on standing and especially with the Valsalva maneuver, and the pubic bone is often tender too. An actual hernial orifice is not palpable as with a typical inguinal hernia. The clinical examination always includes the musculoskeletal system and palpation of the abdomen.
The musculoskeletal structures to consider in the differential diagnosis are:
• The sacroiliac joint.
• The hip:
◦ Osteoarthritis.
◦ Coxa vara, coxa valga.
◦ Slipped epiphysis and snapping hip.
• The muscles of the pelvic region, especially the adductors, which can be affected by insertion tendinopathies on the pubic bone.
From the neurological point of view, it is important to identify or exclude the ilioinguinal nerve entrapment syndrome. A slightly flexed and internally rotated hip is a frequent clinical sign. Pressure injury of the genital branch of the genitofemoral nerve and/or ilioinguinal nerve often produces sensory deficits in the groin itself and on the inside of the thigh. Radiation of the pain along the spermatic cord (spermatic cord neuralgia) as far as the testis is a typical symptom of such nerve irritation.
Radicular syndromes due to degenerative disease of the spine or intervertebral discs can also involve the groin in the area of pain radiation, usually unilaterally. Radiography may aid diagnosis.
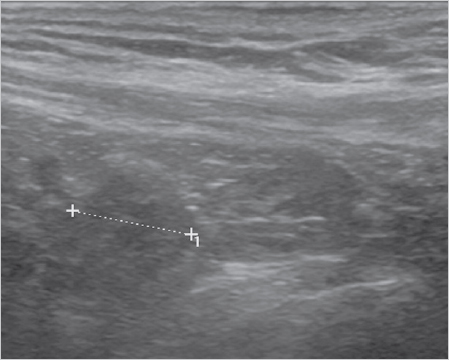
Fig. 3.4 Dynamic ultrasound scan with typical appearance of the bulge, which is diagnostic of sportsman’s groin.
Ultrasonography
In addition to an accurate history and physical examination, ultrasonography plays a very important role. The sliding layers in the groin region can be examined and the local bulge (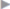 Fig. 3.4) of the posterior wall of the inguinal canal during static and dynamic examination (when the patient strains) can be detected.80 This finding is diagnostic in conjunction with the clinical symptoms. Correctly performing and interpreting an ultrasound of the groin requires much experience and practice.
Fig. 3.4) of the posterior wall of the inguinal canal during static and dynamic examination (when the patient strains) can be detected.80 This finding is diagnostic in conjunction with the clinical symptoms. Correctly performing and interpreting an ultrasound of the groin requires much experience and practice.
Magnetic Resonance Imaging and Computed Tomography
Computed tomography (CT) is rarely required to diagnose sportsman’s groin. Magnetic resonance imaging (MRI) plays a pivotal role in its diagnosis. For patients with a normal clinical examination and unclear ultrasound findings, MRI of the region including the spine is indicated, especially to detect conditions of bone and cartilage that cannot be seen directly on conventional X-ray. MRI may show, for example, an avulsion fracture or insertion tendinopathy (“conjoint tendinopathy”), that is, conditions that necessitate mainly conservative management.55 However, the protrusion of the posterior wall of the inguinal canal can never be seen on MRI, as the examination is performed with the patient lying down and the circumscribed defect is too small.
Herniography
Herniography is still quite widely used in the Anglophone countries.29 In our experience, however, it can hardly be recommended on account of the radiation exposure and the test can easily be replaced by ultrasonography performed by an experienced examiner.
3.2.3 Treatment
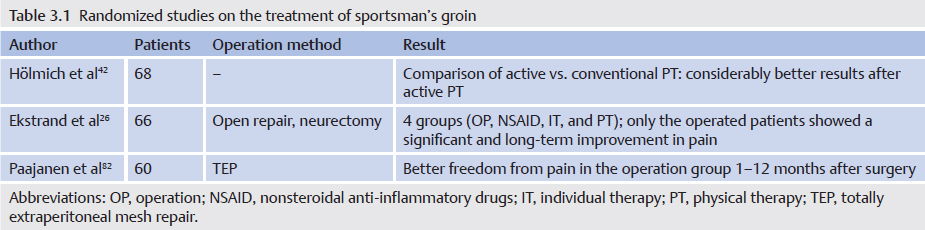
Unlike the treatment of inguinal hernia, which does not include any conservative component, sportsman’s groin is always treated individually, considering all therapeutic modalities. Many of the affected patients are treated exclusively conservatively for a long time.29 The achievable rates of freedom from pain until the next playing season125 are unsatisfactory at only 41%. There are hardly any randomized studies in the literature addressing the question of optimal therapy (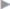 Table 3.1). It can be deduced from the available studies that operative therapy appears to be superior to conservative therapy alone or physical therapy. In the absence of ultrasound evidence of abdominal wall instability, some research groups recommend invasive abdominal wall procedures such as anterior pelvic floor repair or adductor tenotomy. In addition, procedures such as neurectomy on its own in the inguinal canal and transabdominal preperitoneal (TAPP) mesh repair are used (for a summary, see31 and70). Laparoscopic TAPP is used to treat sportsman’s groin especially in America.
Table 3.1). It can be deduced from the available studies that operative therapy appears to be superior to conservative therapy alone or physical therapy. In the absence of ultrasound evidence of abdominal wall instability, some research groups recommend invasive abdominal wall procedures such as anterior pelvic floor repair or adductor tenotomy. In addition, procedures such as neurectomy on its own in the inguinal canal and transabdominal preperitoneal (TAPP) mesh repair are used (for a summary, see31 and70). Laparoscopic TAPP is used to treat sportsman’s groin especially in America.
Our research group has developed the minimal repair technique to treat sportsman’s groin and has used it in over 3,500 patients since 2002. It is used in all athletes in whom posterior weakness of the inguinal canal can be demonstrated clinically and on ultrasound and in whom a purely bony or ligamentous origin of the pain is unlikely. The minimal repair mechanism involves refixation of the rectus muscle with a resulting reduction in the tension on the pubic bone (see operation techniques in section 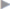 2.2.2). When adductor problems are also present, we recommend physical therapy in addition.
2.2.2). When adductor problems are also present, we recommend physical therapy in addition.
Neurectomy is combined only as needed (macroscopic abnormality or unfavorable anatomical course), usually by resection of the genital branch of the genitofemoral nerve, and occasionally of the ilioinguinal nerve. In case of doubt, we currently tend to resect a nerve in the painful groin region rather than leave it.
With the minimal repair technique, we deliberately decide against mesh implantation. In our view, the size of the defect in the posterior wall of the inguinal canal, which is not more than 2 to 3 cm in most patients, does not require mesh. Moreover, an implanted mesh graft would interfere with the muscular sliding of the athlete’s abdominal wall or even abolish it.
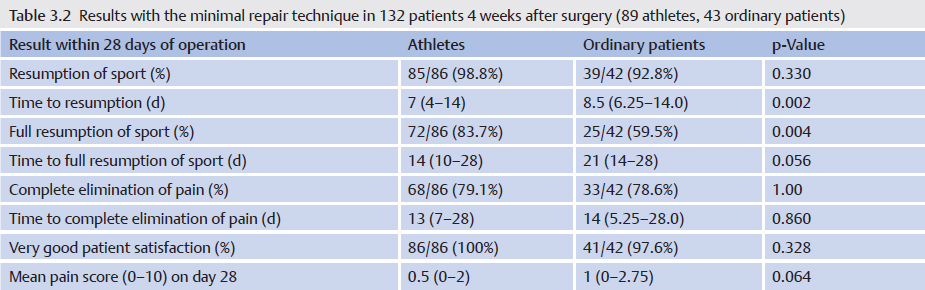
The most important result for all affected patients is rapid resumption of their high-performance sport. We investigated this in a recently published study and we found that 83.7% of the athletes were able to return fully to their sport after a median 14 days (range: 10–28 days) with an outstanding improvement in their symptoms (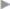 Table 3.2). Patient satisfaction is excellent. The flow sheet in
Table 3.2). Patient satisfaction is excellent. The flow sheet in 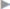 Fig. 3.5 summarizes our recommended treatment. An important aspect of rehabilitation is a training plan tailored to the patient’s sport. We support the patient’s wish for early exercise: sports such as jogging, cycling, and training can start between days 2 and 5, increasing to full loading between days 6 and 10. The aim is defined as full loading after 14 days.
Fig. 3.5 summarizes our recommended treatment. An important aspect of rehabilitation is a training plan tailored to the patient’s sport. We support the patient’s wish for early exercise: sports such as jogging, cycling, and training can start between days 2 and 5, increasing to full loading between days 6 and 10. The aim is defined as full loading after 14 days.
3.2.4 Summary
Sportsman’s groin is a new challenge for the hernia surgeon as sports challenges in adolescence are growing and professional sportsmen (footballers, ice hockey players) are suffering increasingly often from inguinal pain syndromes that cannot be managed conservatively. Careful clinical examination and ultrasonography by an experienced examiner can regularly demonstrate weakness of the posterior wall of the inguinal canal. The minimal repair technique is a gentle operative therapy with excellent short- and long-term results and high patient satisfaction.
Fig. 3.6 Sliding hernia. It is repaired by creating a new peritoneal ring, incising the free peritoneum.
3.3 Sliding Hernia
The presence of a sliding hernia must be suspected if a greatly thickened hernia swelling is found when the external oblique aponeurosis is divided. The hernial sac is dissected in the usual way. It is not necessary to open the sac prior to complete dissection. Adhesions to the spermatic cord can usually be divided readily. The sac of an indirect hernia should be dissected as far as the deep inguinal ring and that of a direct hernia to beneath the level of the transversalis fascia.
If a sliding hernia is suspected based on the consistency of the hernial sac, the sac is opened between clamps on the anterior wall (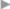 Fig. 3.6). It must be ensured that only the anterior peritoneal layer is divided; so, no part of the bowel is injured. Opening the sac of small medial sliding hernias can be omitted when the bladder forms part of the wall. In these cases, a superficial purse string suture suffices to bury the sac.
Fig. 3.6). It must be ensured that only the anterior peritoneal layer is divided; so, no part of the bowel is injured. Opening the sac of small medial sliding hernias can be omitted when the bladder forms part of the wall. In these cases, a superficial purse string suture suffices to bury the sac.
In the case of larger sliding hernias, the sac must be opened at the lateral and medial border of the sliding component as far as the deep inguinal ring (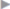 Fig. 3.7). This enables the sac contents to be reduced. Reperitonealization should not be attempted because of the risk of injury.
Fig. 3.7). This enables the sac contents to be reduced. Reperitonealization should not be attempted because of the risk of injury.
After complete excision of the wall of the hernia bearing the bowel, the free peritoneal margins behind the loop of bowel are sutured to create a new deep inguinal ring (interrupted 2/0 silk or PDS suture; 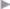 Fig. 3.8). The hernia contents can then be reduced.
Fig. 3.8). The hernia contents can then be reduced.
Fig. 3.8 Sliding hernia. The peritoneal leaf behind the hernial sac is folded and sutured to create a new peritoneal funnel.
After complete reduction, the peritoneum is closed by an additional purse string suture in the newly created peritoneal funnel (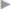 Fig. 3.9).
Fig. 3.9).
Another technique for burying the sac is resection of the free peritoneal covering to leave a narrow margin, which is closed by an internal purse string suture (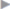 Fig. 3.10).
Fig. 3.10).
Fig. 3.9 Sliding hernia. The sac is buried in the newly created inguinal ring by an external purse string suture.
Fig. 3.11 Sliding hernia. Repair by an external purse string suture. After inspection, the sac is closed and buried by an external purse string suture.
The hernia swelling is buried beneath the deep inguinal ring by an external purse string suture (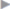 Fig. 3.11).
Fig. 3.11).
It can be assumed overall that complete reduction and adequate closure of the hernial orifice are more important than anatomical restoration of the peritoneal covering. Reduction using the Hoguet maneuver has proved effective for medial sliding hernias and sliding femoral hernias. This can convert the hernia to a lateral inguinal hernia, which can be repaired as described above.
3.4 Recurrent Hernia
“The most common cause of recurrent hernia is poor technique by the surgeon at the first operation.”
(Lloyd M. Nyhus 1989)
“It is the pathological changes in collagen that set the stage for the development of a recurrent hernia.”
(R. Bendavid 2004)
3.4.1 General Remarks/Epidemiology
The touchstone of every hernia repair procedure is the recurrence rate. Therefore, critical examination of the employed method can only be achieved by consistent follow-up of one’s own patients. According to the literature, excellent recurrence rates can be achieved in specialized centers with nearly all inguinal hernia repair procedures. A meta-analysis of prospective randomized studies that included all procedures showed that open or laparoscopic mesh procedures have an advantage over sutured methods.65 However, the recurrence rate varied considerably among the individual studies. These results are supported by large population-based analyses, e.g., the Danish Hernia Database, which show significantly lower reintervention rates for primary operations using mesh implantation.106 Thus, as in incisional hernia repair, mesh implantation appears at least to postpone the onset of a recurrent hernia.28 Since recurrent hernias will never be entirely a thing of the past, they demand detailed consideration.
3.4.2 Risk Factors
For a long time, it was generally assumed that poor surgical technique is the primary cause of hernia recurrence.1 It is also certain that inadequate technique (e.g., insufficient mesh size in TEP/TAPP, failure to expose and double the entire transversalis fascia in the Shouldice procedure) predisposes to recurrence. However, scientific insights today suggest that impaired collagen metabolism and other endogenous or exogenous factors are also of great importance with regard to the development of a recurrent inguinal hernia.46,51,96 The endogenous risk factors can include the patient’s age, while smoking and various medications are among the exogenous risk factors.115 Lau et al. describe a potential genetic component in a study of 1418 men in whom a positive family history causes an eightfold increase in the risk of having an inguinal hernia.54
3.4.3 Forms and Location
Genuine recurrences must be distinguished from false ones. While a genuine recurrence develops at the operation site, a false recurrence appears in immediate proximity or is an additional hernia (e.g., femoral hernia) overlooked at the initial operation. As with primary hernias, recurrent hernias can be classified as direct, indirect, and combined. Protrusion of an elastic mesh through the former hernial orifice after preperitoneal repair of large defects without closure of the orifice can be defined as bulging.
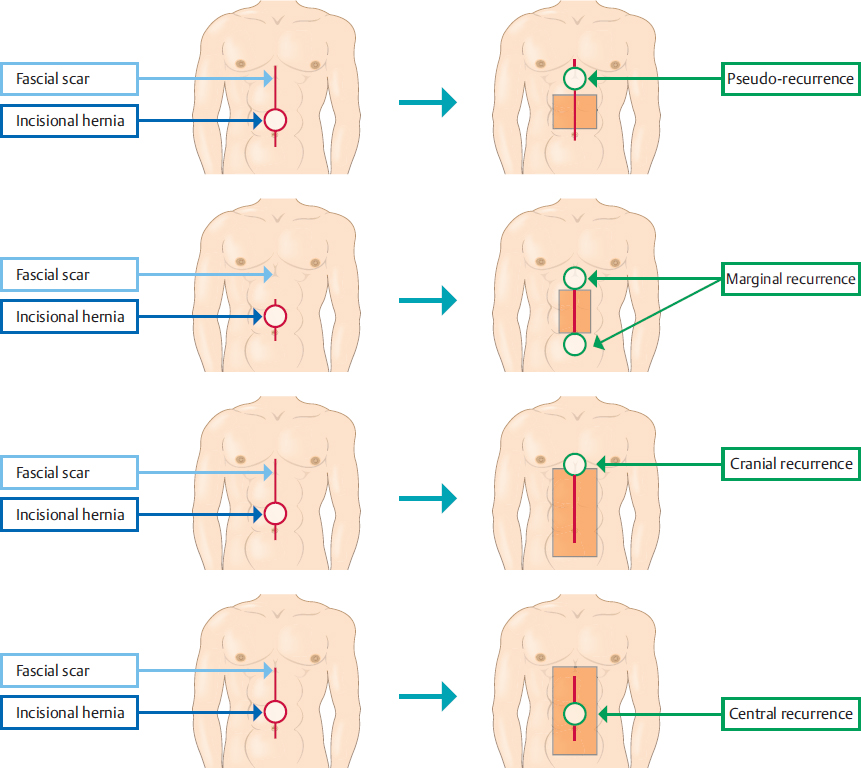
Recurrence can be expected with up to 50% of incisional hernias if mesh procedures are not used. They are usually located at the center of the repair and only rarely at the periphery. Recurrent hernias after technically insufficient operations using mesh appear in typical locations (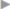 Fig. 3.12). For instance, pseudorecurrence is found after inadequate excision and stabilization of the previous laparotomy scar (
Fig. 3.12). For instance, pseudorecurrence is found after inadequate excision and stabilization of the previous laparotomy scar (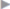 Fig. 3.12a), marginal recurrence in the absence of cranial and caudal overlapping (
Fig. 3.12a), marginal recurrence in the absence of cranial and caudal overlapping (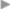 Fig. 3.12b), the typical subxiphoid cranial recurrence when the subxiphoid space is not dissected (
Fig. 3.12b), the typical subxiphoid cranial recurrence when the subxiphoid space is not dissected (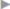 Fig. 3.12c), and central recurrence (
Fig. 3.12c), and central recurrence (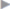 Fig. 3.12d) when the anterior layer of the rectus sheath is not closed sufficiently.
Fig. 3.12d) when the anterior layer of the rectus sheath is not closed sufficiently.
Recurrent inguinal hernias include indirect hernias (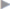 Fig. 3.13) and different forms of direct hernias (medial, suprapubic, intermediate, complete,
Fig. 3.13) and different forms of direct hernias (medial, suprapubic, intermediate, complete, 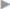 Fig. 3.14,
Fig. 3.14, 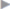 Fig. 3.15,
Fig. 3.15, 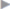 Fig. 3.16,
Fig. 3.16, 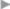 Fig. 3.17).
Fig. 3.17).
Fig. 3.12 Location of typical recurrent hernias due to technical errors during the previous operation.
3.4.4 Choice of Procedure
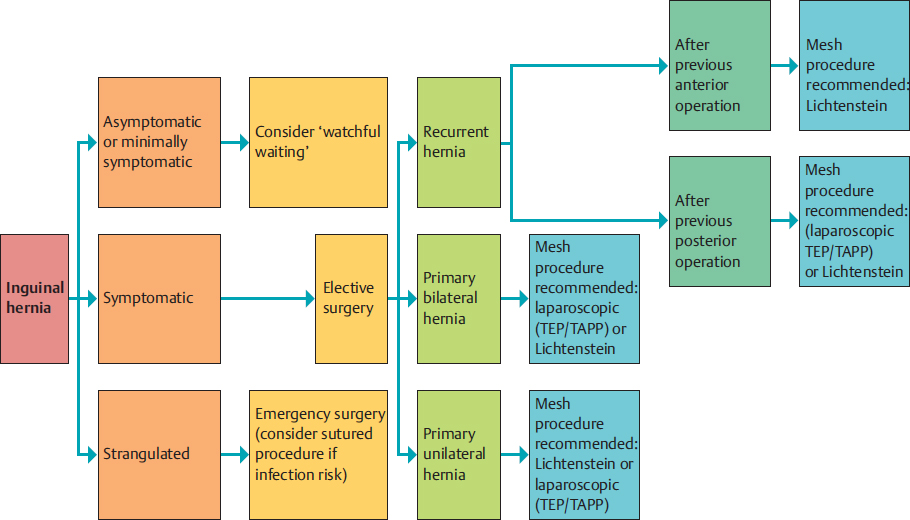
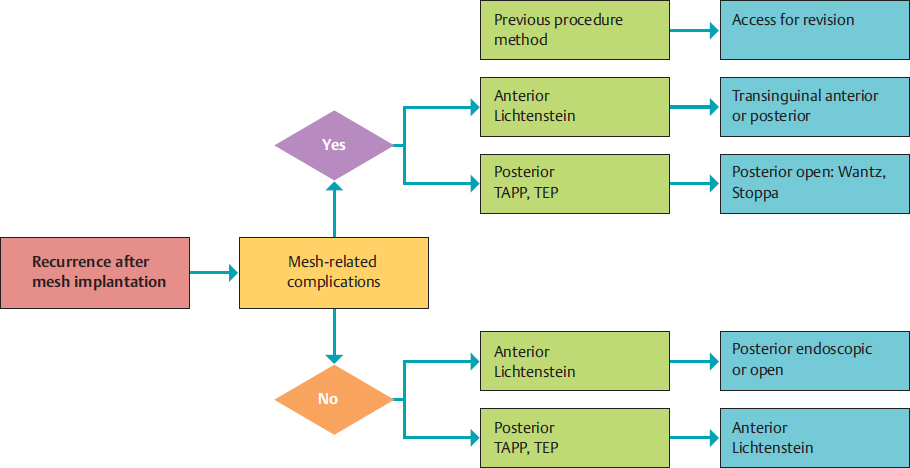
The European Hernia Society guidelines on choice of procedure recommend a change of access route when treating a recurrent hernia. The approach to recurrent hernias following previous anterior repair should be laparoscopic and posterior if possible, and an anterior approach should be used for recurrences after previous posterior surgery (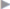 Fig. 3.18).
Fig. 3.18).
Fig. 3.19 Choice of procedure to treat recurrent inguinal hernia depending on the presence of mesh-related complications and type of previous surgery.
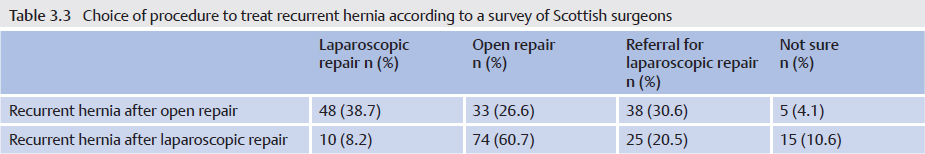
The guidelines of the Danish Hernia Database published in 2011 also include a change of access route in their recommendations.97 This procedure has become partly established clinically. In a survey of compliance with National Institute of Clinical Excellence (NICE) guidelines from 2004 recommending a laparoscopic approach for recurrent hernia, Shaikh110 found in 2011 that the access route for recurrent hernia repair had already changed (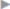 Table 3.3).
Table 3.3).
There are no comparative studies on the change of access route. Apart from the actual recurrence, however, chronic pain and a foreign body sensation are often the primary indication for revision of a previous inguinal hernia repair using mesh. In these patients, additional neurectomy and/or mesh removal may be indicated for pain therapy. For these revision operations, Schwab and Klinge in 2007 proposed the algorithm shown in 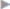 Fig. 3.19, which corresponds to the EHS proposal in the absence of mesh-related complications but suggests repeating the primary access route when mesh revision is indicated.107
Fig. 3.19, which corresponds to the EHS proposal in the absence of mesh-related complications but suggests repeating the primary access route when mesh revision is indicated.107
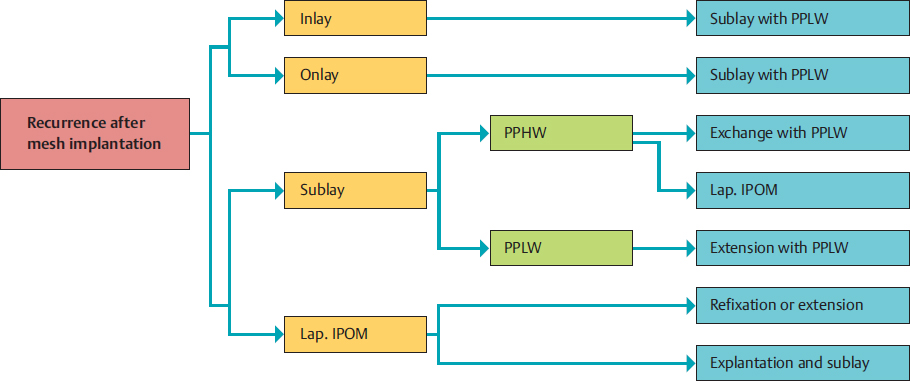
All mesh implantation procedures can be used to manage a recurrent incisional hernia after primary sutured repair. For recurrences following mesh implantation, Conze, Binnebösel, and Klinge21 suggest the following algorithm for managing a recurrent incisional hernia. The authors regard repair by retromuscular sublay mesh augmentation as the reference procedure, and recurrence after inlay or onlay incisional hernia repair is treated by sublay with a lightweight mesh. After sublay repair with lightweight mesh, the previous repair is extended and after previous surgery using heavyweight mesh, this is exchanged for lightweight mesh or the procedure is changed to laparoscopic IPOM. After a previous laparoscopic IPOM procedure, the mesh can be either refixed or extended or the mesh can be explanted, with a switch to open sublay repair (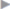 Fig. 3.20).
Fig. 3.20).
Overall, there is no gold standard procedure for repairing recurrent inguinal and incisional hernias. The choice of procedure must be guided by the patient and the hernia itself.
3.4.5 Preparation
The diagnosis is made preoperatively by clinical examination, ultrasonography of the groin or abdominal wall to determine the defect size, and Doppler ultrasound of the testicular vessels in the case of recurrent inguinal hernia. Detailed knowledge of the previous operation can considerably simplify the procedure (type, size of the implanted mesh, location of the mesh materials, mesh fixation, location of the spermatic cord, 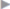 Fig. 3.21).
Fig. 3.21).
Fig. 3.20 Choice of procedure to treat recurrent inguinal hernia depending on the type of previous surgery and mesh material.
PPHW: heavyweight polypropylene
PPLW: lightweight polypropylene
Fig. 3.21 Recurrent hernia. Scarred spermatic cord moved to subcutaneous position after previous Bassini–Kirschner operation.
3.4.6 Dissection and Repair
Because of the large number of possible previous operations including their technical variants (mesh type and size, mesh location, mesh fixation) for both inguinal and incisional hernias, a general detailed description of the dissection and repair of a recurrent hernia is not feasible and the repair must be guided by the patient and the hernia. The aim must be long-term stabilization of the groin or abdominal wall with alloplastic mesh material.
3.4.7 Complications
Compared with the primary operation, significantly increased complication rates are found, both intraoperative (nerve, vas, and bowel injury) and early and late postoperative (hematoma, wound infection, chronic pain, re-recurrence).
3.5 Umbilical Hernia
3.5.1 Epidemiology and Pathogenesis
Five percent of all hernias are umbilical hernias.105 There is a high incidence in premature infants and babies with birth defects with a birth weight below 1,500 g. With a few exceptions, healing to a stable umbilical scar can be expected by the age of 2 years following obliteration of the umbilical vessel.38,39 Surgical treatment is not required in 98% of cases.104,126 Unlike omphalocele, a congenital malformation of the abdominal wall with protrusion of abdominal viscera, the abdominal wall is otherwise well developed with an umbilical hernia. In adults, development of an umbilical hernia is promoted by risk factors such as pregnancy, obesity, disorders of connective tissue metabolism due to endogenous (genetic) and exogenous (smoking, corticosteroids) factors, hepatic cirrhosis, and malignant disease. Spontaneous healing cannot be assumed apart from in pregnant women, in whom recovery of the abdominal wall can be anticipated after delivery. In adulthood, the incarceration rate is up to 30% with mortality of 10 to 15%.104
3.5.2 Diagnosis and Classification
A palpable, painless protrusion in the umbilical region is typical. In many patients, the umbilical hernia has been present for years without significant changes and with only slight symptoms. If it increases rapidly in size, intra-abdominal pathology must be excluded. Clinical examination should be supplemented by ultrasonography of the abdominal wall. The ultrasound scan should extend over the entire midline from the xiphoid to 3 cm below the umbilicus. It allows assessment of the linea alba for other gaps and the presence of rectus diastasis.
Classification follows EHS criteria (European Hernia Society). Hernias are classified as small up to 2 cm, medium defects of 2 to 4 cm, and large defects with a diameter over 4 cm. Small umbilical hernias are further classified as minimal defects below 0.5 cm.
3.5.3 Indications
Incarceration and persistent symptoms are absolute indications. Diagnosis after the age of 6 years with minor symptoms is a relative indication. Surgery in infancy should be performed only in exceptional cases, e.g., because of recurrent incarceration. The choice of an appropriate operative procedure is crucial for the recurrence rate and complications. Fascial suture of large hernias on its own leads to recurrence rates of 20 to 45%, but with small defects up to 2 cm recurrence occurs in only 4.1 to 6.6% of cases.103
Surgical treatment is not necessary for asymptomatic umbilical hernias < 0.5 cm. Only recurrent symptoms justify repair of small umbilical hernias. Spitzy sutured repair generally suffices for fascial defects < 2 cm. The recurrence rate increases markedly with 2 to 4 cm umbilical hernias and mesh insertion is recommended. This can be by preperitoneal umbilical mesh plasty (PUMP), retromuscular mesh in the classic onlay technique, or intraperitoneal technique (open IPOM) in an open operation, or by a laparoscopic procedure (lap. IPOM). Mesh repair is regarded as obligatory for umbilical hernias with a defect greater than 4 cm. There are no limits to laparoscopic and open IPOM insertion as regards hernia size. PUMP repair is limited by the preperitoneal space that can be dissected roughly 2 to 3 cm in all directions from the fascial border. Defects greater than 4 cm may not be suitable for PUMP repair and require retromuscular mesh placement, such as midline incisional hernias.
3.5.4 Complications
With open repair, impaired wound healing due to infection and disturbed blood supply is caused by division of the base of the umbilicus. Perioperative antibiotic prophylaxis is often recommended but is not evidence-based. As regards the impaired perfusion of the umbilical skin, this should be minimized by an appropriate incision with little or no dissection of the sac from the skin. Bowel injuries are rare but have been described with both open and laparoscopic procedures and must be included in the informed consent. Intra- and postoperative bleeding results from the anatomy of the abdominal wall with vessels in the median umbilical fold. Umbilical hernias in patients with cirrhosis and ascites present particular challenges. The tendency to incarceration is greater and skin ulceration and postoperative ascites leaks occur more often. Besides attempting to correct the cirrhosis preoperatively to reduce ascites, an abdominal drain is recommended for 10 to 14 days until the wound heals.
3.5.5 Operation Technique
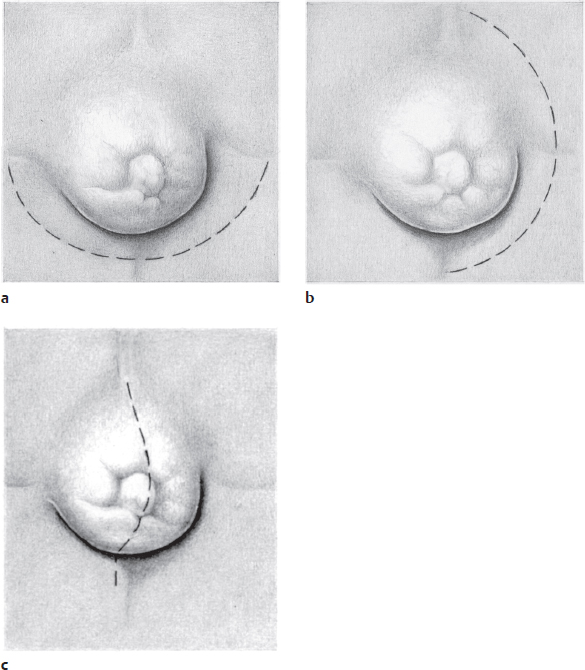
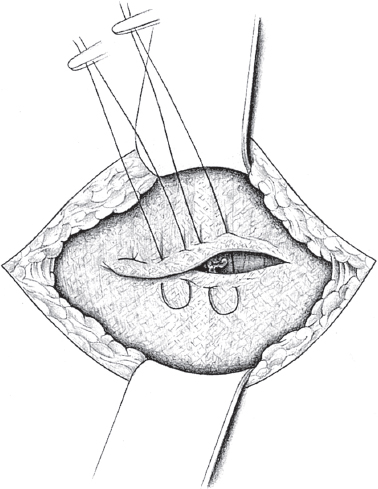
The Spitzy semicircular infraumbilical incision, the Drachter semicircular left lateral incision, and the vertical transumbilical incision can be considered for open operation (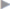 Fig. 3.22). The latter provides almost “scar-free” repair69 but should be used only when the skin has no deep and inaccessible creases and can be disinfected reliably because of the potential risk of infection. If the umbilical skin is inflamed or necrotic, the umbilicus is excised and the procedure concludes with reconstruction of an umbilical depression.
Fig. 3.22). The latter provides almost “scar-free” repair69 but should be used only when the skin has no deep and inaccessible creases and can be disinfected reliably because of the potential risk of infection. If the umbilical skin is inflamed or necrotic, the umbilicus is excised and the procedure concludes with reconstruction of an umbilical depression.
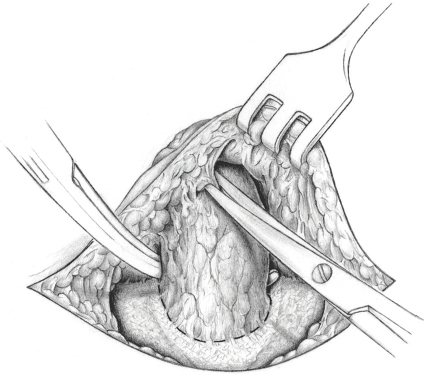
Following the skin incision, dissection continues down to the fascia, and the hernial sac is bypassed at fascial level, detached or ideally divided at the base of the umbilicus, and buried with a purse string suture (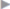 Fig. 3.23,
Fig. 3.23, 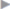 Fig. 3.24). Alternatively, a circular opening is made in the sac at fascial level, the contents are reduced, and the peritoneum is sutured after mobilization from the posterior layer of the rectus sheath (e.g., 2/0 or 3/0 PGA).
Fig. 3.24). Alternatively, a circular opening is made in the sac at fascial level, the contents are reduced, and the peritoneum is sutured after mobilization from the posterior layer of the rectus sheath (e.g., 2/0 or 3/0 PGA).
Fig. 3.22 Incisions.
a Spitzy inferior incision.
b Drachter lateral incision.
c Transumbilical incision.
Fig. 3.23 Dissection of the sac, bypassing of the neck of the sac at fascial level with the Overholt forceps.
Spitzy Repair
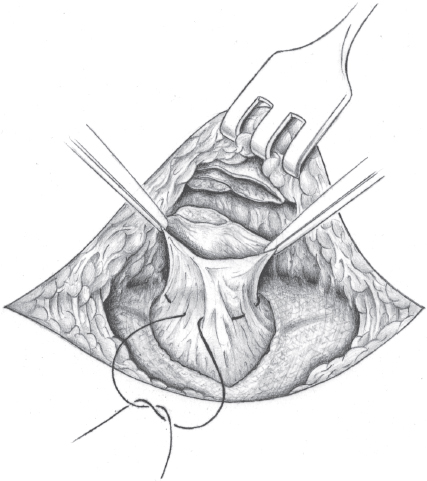
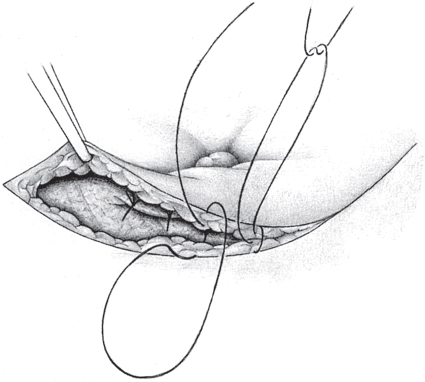
Hernias up to 2 cm are closed, preferably transversely, by a continuous suture of the fascial margins (e.g., polypropylene 0 or 2/0). The peritoneum should be dissected from the posterior layer of the rectus sheath to ensure that the suture is placed under vision and interposition of the peritoneum in the fascial defect is avoided (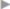 Fig. 3.25). Both permanent (polypropylene) and slowly absorbable (PDS) sutures can be used. An alternative is the Mayo “vest-over-pants” repair with a transverse row of U sutures to close the fascia (
Fig. 3.25). Both permanent (polypropylene) and slowly absorbable (PDS) sutures can be used. An alternative is the Mayo “vest-over-pants” repair with a transverse row of U sutures to close the fascia (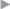 Fig. 3.26). However, better long-term results have not been achieved with the overlapping fascia. In any case, tension-free approximation of the fascial edges is desirable. In case of doubt, alloplastic material must be used. The operation concludes with fixation of the base of the umbilicus at fascial level (
Fig. 3.26). However, better long-term results have not been achieved with the overlapping fascia. In any case, tension-free approximation of the fascial edges is desirable. In case of doubt, alloplastic material must be used. The operation concludes with fixation of the base of the umbilicus at fascial level (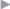 Fig. 3.27) and intracutaneous suture (
Fig. 3.27) and intracutaneous suture (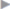 Fig. 3.28).
Fig. 3.28).
Preperitoneal Umbilical Mesh Plasty
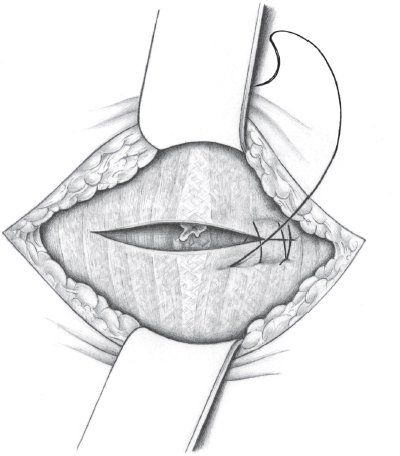
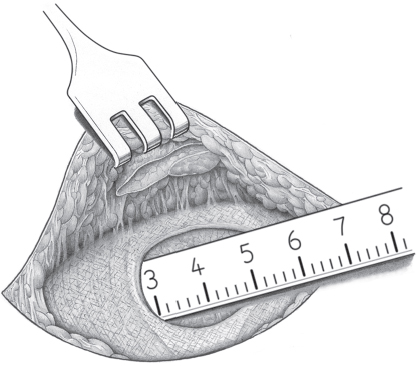
Preperitoneal umbilical mesh plasty (PUMP) repair is suitable for 2 to 4 cm fascial defects. The advantages are that the technique is easy to learn and uses an individually tailored, economical flat mesh; direct contact between mesh and bowel is reliably avoided. After treating the hernial sac and peritoneal defect, the peritoneum is dissected bluntly from the edges of the defect in all directions for a distance of 3 cm (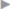 Fig. 3.29). A circular or oval implant at least 6 cm in diameter is cut from a lightweight large-pored mesh and two to three fixing sutures (e.g., PGA 2/0) are placed in it (
Fig. 3.29). A circular or oval implant at least 6 cm in diameter is cut from a lightweight large-pored mesh and two to three fixing sutures (e.g., PGA 2/0) are placed in it (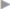 Fig. 3.30). The mesh is then spread out flat in the preperitoneal space and fixed by loose sutures through the posterior wall of the fascia (
Fig. 3.30). The mesh is then spread out flat in the preperitoneal space and fixed by loose sutures through the posterior wall of the fascia (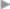 Fig. 3.31). The fascia is closed transversely with a continuous suture (e.g., polypropylene 0 or 2/0). The operation concludes with fixation of the base of the umbilicus and skin suture.
Fig. 3.31). The fascia is closed transversely with a continuous suture (e.g., polypropylene 0 or 2/0). The operation concludes with fixation of the base of the umbilicus and skin suture.
Lateral dissection of the peritoneum from the posterior layer of the rectus sheath may not be possible with very large umbilical defects. In these cases, the defect can be closed with a retromuscular graft, similar to midline incisional hernia repair. If rectus diastasis is also present, repair of the midline up to the xiphoid is recommended.
Laparoscopic umbilical hernia repair is similar to midline incisional hernia repair.
Prefabricated mesh is available for open intraperitoneal placement as an alternative to preperitoneal mesh implantation. The operation technique is relatively easy, as the grafts are placed in the free abdominal cavity and dissection of the peritoneum is not necessary. However, these implants come with the risks of direct mesh-bowel contact, represent a large foreign body burden, and are relatively expensive.
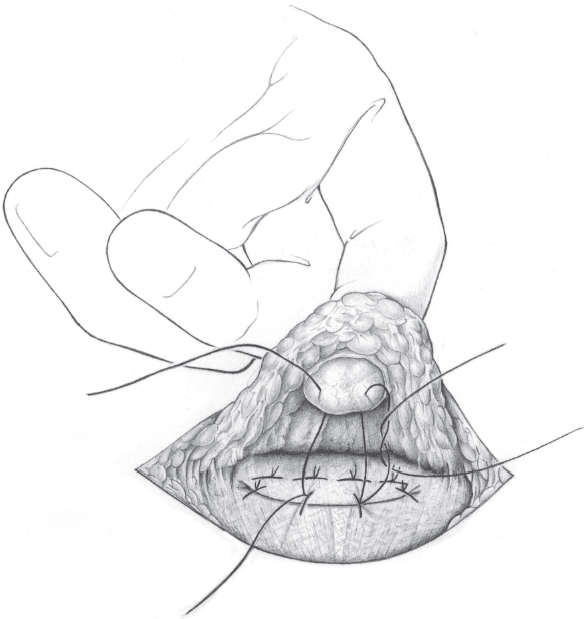
Fig. 3.27 The base of the umbilicus is pushed down with the index finger and fixed to the fascia with 1 to 2 interrupted 2/0 PGA sutures.
Fig. 3.29 Preparation of the mesh bed, dissecting the peritoneum from the linea alba and posterior layer of the rectus sheath for 3 cm around the fascial defect.
Fig. 3.30 Placement of PGA fixing sutures in the individually tailored mesh prior to implantation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree