Introduction
Compartment syndrome in either upper or lower extremity is often underestimated and may have detrimental consequences for functioning of the involved extremity. Muscle groups in the extremities are separated by fascial sheaths to ensure that muscles move in the right vector and achieve maximal efficiency during contraction. Fascial sheaths also provide additional surface area for muscle origins because bone alone does not offer enough surface area. Fascial sheaths protect neurovascular bundles.
Groups of muscles in the arms and legs, together with blood vessels and nerves, are contained in compartments surrounded by non-elastic fascia and/or bone; therefore, whenever bleeding and/or edema occur, increased pressure is inevitable. This increased pressure can harm the intra-compartmental soft tissues, leading to detrimental consequences. Focusing on the different compartments in both the upper and lower extremities a distinct architecture can be recognized.
In this chapter we address the anatomy of both upper and lower extremities to illustrate the rationale for the occurrence of compartment syndrome, its consequences, and management strategies.
Anatomy
Fascia
The thickness of the fascia is determined by the amount and direction of the applied force. Comparing the fascia of the lower leg (i.e., fascia cruris) to the fascia of the forearm (i.e., fascia anti-brachii), not only can a significant difference in thickness be noted, favoring the fascia cruris, but also a difference in the direction of the fascia fibers. The fascia cruris fibers run in a circular direction, whereas the fibers of the fascia antibrachii do not have a uniform shape or direction. This difference can be explained by the constant contraction and relaxation of the calf muscles, which is of much higher amplitude than that of their forearm counterparts.
Upper Extremity
Forearm
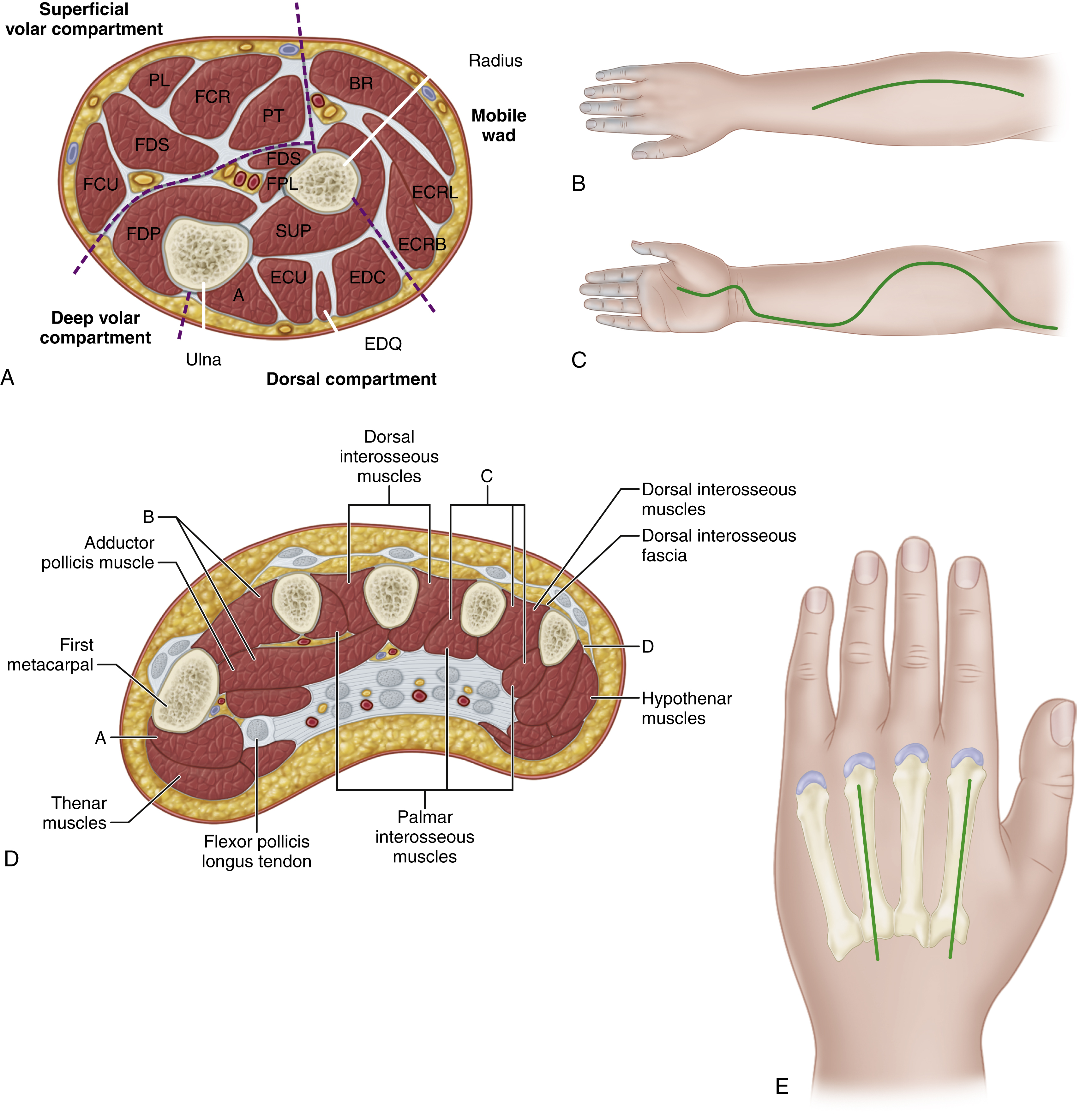
The forearm contains three compartments: dorsal, volar, and mobile wad compartments. The dorsal compartment contains the abductor pollicis, extensor pollicis longus, extensor carpi ulnaris, extensor digiti minimi, and extensor digiti communis muscles. Inferior to the muscles, the radius and ulna can be found, separated by the interosseous membrane. The volar compartment is separated into superficial and deep muscles. The deep muscles include the flexor pollicis longus, flexor digitorum profundus, and pronator quadratus. The superficial muscles are the palmaris longus, pronator teres, flexor digitorum superficialis, flexor carpi ulnaris and radialis. The ulnar and radial bone and the interosseous membrane border the compartment dorsally. Volar borders are the subcutaneous antebrachial fascia. This fascia also separates the volar compartment from the lateral compartment (i.e., the mobile wad ), which holds the brachioradialis, extensor carpi radialis brevis and longus muscles ( Fig. 51.1A–C ).
Hand
Looking at the different osteofascial compartments of the hand, 10 separate compartments can be distinguished: four dorsal interossei compartments, three palmar interossei compartments, the adductor pollicis compartment, and the thenar as well as hypothenar compartments ( Fig. 51.1D, E ) .
Lower Extremity
Lower Leg
Anatomically, the lower leg can be divided in four compartments: the deep and superficial posterior compartments and the lateral and anterior compartments.
The fascia cruris, tibia, fibula, and interosseus membrane surround the anterior compartment . The anterior tibial muscle, extensor hallucis longus, and extensor digitorum longus are the muscles in this compartment. The anterior tibial artery and veins run along these muscles distally and the deep peroneal nerve innervates them. The lateral compartment comprises the long peroneal and brevis muscles and the common peroneal nerve proximally. The crural fascia, intermuscular septum fascia, and fibula compartmentalize these structures. The superficial posterior compartment is separated from the deep compartment by the transverse intermuscular septum and is ensheathed by the fascia cruris. This compartment holds space for the plantar, soleus, and gastrocnemius muscles. The deep posterior compartment is encircled by the already mentioned transverse septum, the tibial and fibular bones, and the interosseus membrane; it also contains the tibial nerve and posterior tibial and peroneal arteries and veins. Muscles that are located in this compartment are the posterior tibial, flexor hallucis longus, and flexor digitorum longus muscles.
Foot
Three compartments cover the full length of the foot: medial, lateral, and superficial compartments. In addition, six intrinsic compartments are described (i.e., four interossei compartments and an adductor compartment) and a calcaneal compartment.
The medial compartment holds the flexor hallucis brevis and the abductor hallucis muscle. The lateral compartment contains the abductor digiti quinti and the flexor digiti minimi muscles. In the superficial compartment the flexor digitorum brevis, the flexor digitorum longus tendons, and the lumbrical muscles are located. Also, the medial plantar nerve is running through this compartment. The interossei and adductor compartments hold the interossei muscles and the adductor muscle, respectively. The calcaneal compartment not only comprises the quadratus plantae muscle, but also the posterior tibial, medial, and lateral plantar nerves. All vessels of the foot run through this compartment: the posterior tibial, medial, and lateral plantar arteries and veins.
Pathophysiology
Each compartment has an ideal pressure for optimal tissue perfusion, based on the balance between the hydrostatic and osmotic pressure. The intracapillary hydrostatic pressure normally varies between 20 mmHg and 30 mmHg and relates to the difference between venous and arterial pressures. The osmotic pressure or interstitial fluid pressure ensures prevention of outflow and ranges between −5 and +5 mmHg.
Disturbance of this pressure balance can occur as a result of an increase of intracompartmental pressure (ICP) or a decrease of compartmental volume caused by bleeding or swelling of the tissue or a reduction of external volume due to high-pressure compression or casting. It is generally accepted that an increase of the interstitial fluid pressure above the intraluminal pressure of the capillary will prohibit adequate perfusion of the intracompartmental structures.
Translated to a clinical decision tool, different authors choose to have a threshold within 10–30 mmHg of the diastolic blood pressure for surgical fasciotomy in adults. Others state that a fasciotomy is indicated with absolute compartment pressures of >30–45 mm Hg. ,
These threshold values may help decide the necessity for a decompression of a compartment in children. Tharakan et al published their work on compartment pressures in children with normal and fractured forearms. They describe a mean compartmental pressure of 10.22 mmHg (range 3–33 mmHg), which is similar to those reported in adults.
As soon as the perfusion is jeopardized, hypoxia will lead to edema and the initiation of anaerobic metabolism. This cascade leads to a further increase of ICP and thus an even greater decrease of capillary flow. Subsequently, only an intervention can halt this process. If intervention is not performed within 3 hours after the onset of ischemia, an impaired muscle response will occur. Six to eight hours after ischemia the muscle and soft tissue will have suffered irreversible damage.
Subjective
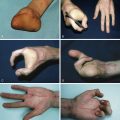
Compartment syndrome is a clinical challenge where the clinical history foretells the physician the problem. Classically, pain, pallor, pulselessness, paresthesia, and paralysis (also called “the five Ps”) are described as the clinical symptoms related to compartment syndrome. Although pain and loss of sensibility are not universal findings, in our survey only one of our patients was asymptomatic (see the section discussing ischemic contracture, below). In all other patients symptoms were present but unfortunately not adequately recognized or treated.
The review by Wall et al confirmed “the five Ps,” except their observation that pulses may be present. Moreover, the pain may be out of proportion to the injury and there is a palpable painful tenseness or swelling of the compartment. Also important is pain on passive stretch of wrist and fingers in the developing compartment syndrome of the upper extremity. Progressively, the increased pressure will lead to ischemia of the nerves, resulting in late-onset clinical signs as sensory and motor deficits.
In the upper extremity a supracondylar fracture of the humerus is the most frequent cause for the development of an ischemic contracture, followed by a fracture of the forearm. These fractures should be monitored carefully with regard to compartment syndrome, especially in children.
It is important to note that compartment syndrome is often missed or discovered later because of modern early treatment with drugs to diminish pain. In most modern hospitals it is supposed to be a medical mistake if patients have pain. At our unit and in our hospital compartment syndromes have been missed for this reason. To prevent this misdiagnosis Schmidt et al have initiated a multicenter prospective study to develop a prediction model for the likelihood of acute compartment syndrome. They have completed their inclusion and published their collected data and are currently developing their prediction model.
Objective
Intracompartmental Pressure
An objective measurement for the diagnosis of compartment syndrome is evaluation of ICP. Aside from the decision of evaluating pressure, one should perform a fasciotomy immediately if there is any doubt about compartment syndrome. In our medical practice, working in a trauma referral center dealing with substantial injuries to the extremities, it is common practice to perform preventive open fasciotomies, especially if vascularization is compromised more proximally.
All techniques described for the assessment of pressure monitoring are based on the same concept, which is, in fact, an intracompartmental placement of a specific measuring device (i.e., the subtle difference between the different techniques), a fluid column, and a transducer for measurement of ICP. As such, either a continuous or a repeated ICP measurement can be established. Ideally, the same clinician monitors the ICP with an interval range between every 30 minutes to 12 times a day.
A recent experimental study by Hammerberg et al compared the reliability of different devices, both digital and analog, using three different terminal devices. The devices investigated were the 18-gauge bevel-tipped needles, the side-ported bevel-tipped needles, and the slit catheters. They concluded that the differences between these three terminal devices are small and unlikely to have an impact on clinical decision-making for fasciotomy. However, when the electronic measurement approach is compared with the analog Whitesides’ technique, the electronic technique is favored because it does not require prior preparation. Nevertheless, Hammerberg et al recommended using the slit catheter for continuous monitoring of one compartment. In the event multiple compartments are at risk or multiple measurements in the same compartment are required, the clinician is advised to use either a standard 18G or side-port bevel-tipped 18G needle.
Near‐Infrared Spectroscopy
Near-infrared spectroscopy uses a differential light absorption to measure the level of hemoglobin, thus providing a measure of oxygenation. Recognizing decreased muscle oxygenation is correlated to the intracompartmental tissue perfusion gradient and thus could be helpful in the diagnosis of acute compartment syndrome. Furthermore, Reisman et al showed an inverse correlation between tissue perfusion and ICP. They noticed an 8% difference, with the perfusion dropping below 10 mmHg. As near-infrared spectroscopy can scan at different depths intracompartmentally, it seems to be useful for monitoring patients at risk of compartment syndrome. However, Shuler et al have recently published their innovative work on continual near-infrared spectroscopy and could not find a strong correlation between presence of compartment syndrome and deviated values. They advised additional interventional studies.
Treatment
If a compartment syndrome is not adequately released, necrosis with all its consequences will be unavoidable. In the presence of the aforementioned symptoms our advice is to act with aggressive decompression of compartments without delay. Surgery is the only treatment that can prevent the disastrous consequences of compartment syndrome by restoring the microcirculation and preventing further damage to the muscles and nerves. Measures such as opening of the plaster cast and elevation of the extremity are not sufficient in these cases. Sheridan and Matsen found that the frequency and severity of complications of decompression were directly related to the promptness with which it was performed. In cases of a suspected vascular injury, decompression has to be combined with vascular exploration and restoration of blood flow. At our unit most decompressions are performed preventively. This is especially the case in crush injuries of the extremities, and particularly if vascularization of the involved extremity is at risk. We do not have a wait-and-see policy and thus do not use this objective evaluation on a regular basis. Table 51.1 provides an overview of fasciotomies carried out in our unit between 2002 and 2012 in order to prevent compartment syndrome or further damage due to ischemia in cases of compartment syndrome. This table well illustrates our policy not to wait and see.
| Upper Extremity ( n = 231) | CTR | GR | IR | HHT | DF | VF |
|---|---|---|---|---|---|---|
| Primary ( n = 186) | ||||||
| Crush (76) | 76 | 57 | 54 | 33 | 40 | 41 |
| Multitrauma (44) | 41 | 24 | 26 | 17 | 16 | 17 |
| Deglovement injury (24) | 21 | 10 | 13 | 8 | 6 | 5 |
| Glass/stab injury (31) | 31 | 11 | 2 | 1 | 5 | 7 |
| Burn injury (6) | 6 | 5 | 6 | 5 | 4 | 4 |
| Circular saw injury (5) | 5 | 2 | 0 | 0 | 0 | 0 |
| Secondary ( n = 45) | ||||||
| Extravasation (12) | 12 | 9 | 6 | 5 | 8 | 10 |
| Postoperative bleeding (20) | 18 | 9 | 6 | 1 | 6 | 7 |
| Infection (13) | 11 | 6 | 6 | 6 | 6 | 6 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree