16 Common Versatile Free Flaps for the Lower Extremity
Summary
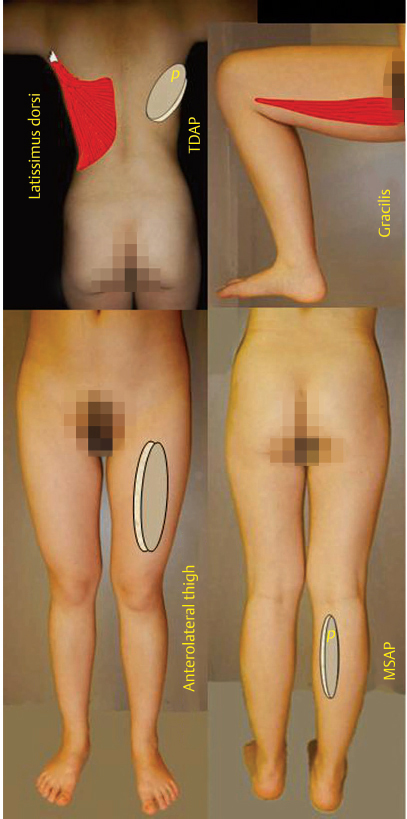
Few would argue that the lower extremity might be the most difficult body region to resurface not just due to the paucity of skin redundancy but also due to not uncommon vascular deficiencies as it is so distant from the heart itself. A surge in interest lately for the use of local flaps as the solution when a vascularized flap is indicated for lower extremity reconstruction, presumably to avoid the risks of microanastomotic failure or technical challenges, should not obscure the fact that free flaps will still always sometimes be needed. Local tissue damage or vascular embarrassment will instead require the selection of a free flap. Some have called the anterolateral thigh flap the “ideal” soft-tissue flap for all purposes in this regard. Other “workhorse” donor sites are the relatively thin medial sural artery perforator flap, the thoracodorsal artery perforator flap that can capture the large dorsal thoracic fascial territory, or the more malleable and versatile gracilis and latissimus dorsi muscle flaps. Many other options do exist such as the superficial circumflex iliac perforator flap, but competency at least in using the aforementioned flaps should be expected, as they each have almost universal applications.
Keywords: anterolateral thigh flap, gracilis muscle, latissimus dorsi muscle, medial sural artery perforator flap, thoracodorsal artery perforator flap, free flap, microsurgical tissue transfer
16.1 Introduction (▶ Fig. 16.1)
What exactly is reconstructive surgery? An exact answer easily eludes us, but Tagliacozzi was on the right tract when he stated that it is rebuilding missing parts so as to restore the function that has been lost.1,2 So how do reconstructive surgeons stand out from the wound center crowds who also claim this capability? Can it be because of our ability to solve more complex problems that require vascularized tissues in the form of what we call flaps? If simpler local options were inadequate, how could then a flap be moved from point A to point B? Hamilton (1854) answered that question by attaching one leg to another as a cross-leg flap.3 Even a staged toe-to-hand transfer was similarly possible using the Nicoladoni method.4 Filatov5 (1917) and Gillies6 (1920) independently developed the tubed pedicle as another means for distant tissue movement. Yet evolution was imperative as the immobilization necessary for success of these now-considered primitive maneuvers was lengthy, could lead to joint stiffness, and required multiple surgical interventions, while risking pressure sores, incompletely surviving flaps, or thromboembolic events. And most important, the outcome was suboptimal compared to today’s high expectations.
The beginning of this new beginning relied on animal experiments in the early 1900s by Carrel7 and Guthrie8 who perfected anastomoses of blood vessels permitting replantations and organ transfers in the laboratory. Many others followed, developing fundamental reliable and consistent operative techniques, the discovery of anticoagulation essential to decrease the risk of thrombosis, and obtained intraoperative magnification for better visualization.8 Jacobson and Tamai,8 then latter Acland,9,10,11 fabricated new designs in instrumentation and suture material that would facilitate what now is called the microvascular anastomoses. O’Brien set up a microsurgical research unit specifically to find answers to previously unrecognized physiological and clinical challenges at the microvascular level.12
Then a tsunami began. McLean and Bunke13 used the omentum as an autotransplant to the scalp. Harii et al,14 in 1972, performed the first successful free skin transfer using the temporal scalp for hair transplantation. Credit for the first composite tissue transfer, however, usually is given to Daniel and Taylor15 for their iliofemoral island flap placed in a single stage on a distal lower limb defect. The “Introduction” of that paper is preceded by some provocative and prophetic words by Harry J. Buncke, Jr., himself that cannot be overlooked: “The successful transplantation of a block of composite tissue by reanastomosing the microvascular pedicle has untold experimental and clinical possibilities.”15
And so it would be, and it was good. Soon muscle,16 bone,17 nerve,18 viscera,19 and every imaginable microsurgical tissue transfer as a “free” flap would be used anywhere in the body. Innovators willing to risk criticism and resistance instead flourished. Godina showed superior results for lower extremity trauma by the immediate use of free flaps.20,21 Hong honed technical expertise with supermicrosurgery.22 Millennials even proved a true value of the cell phone by obtaining thermographic images for perforator localization equal to that possible with CT angiography.23 The Chang Gung group obtained better outcomes using an intensive care “free flap” unit with well-trained nurses for monitoring that could detect earlier microanastomotic catastrophes that could then be more easily rectified, as the sooner the better.24
In this day of perforator flaps where a paradigm shift is being seen prioritizing local flaps for lower extremity soft-tissue coverage,25 is there still a role for free flaps? The answer even for the most casual observer should be “yes.” There can be no argument if local tissues have been too compromised or even absent as in a limb amputation where a joint or limb length must nevertheless be preserved.26 Almost all large wounds and even some small to moderate-sized wounds might still be better served by a free flap. Today, donor site morbidity also must be considered, as oftentimes just the aesthetics would be superior with a small free flap as opposed to a local flap where donor site closure had required a skin graft. Perusal of the process of flap selection in Chapter 13 recites all reasonably available free flap options, but those chosen here have been carefully selected for emphasis. The anterolateral thigh flap, some say, is the “ideal” soft-tissue flap.27 The thinness of the medial sural flap for many has supplanted use of the radial forearm flap as it avoids sacrifice of the radial artery.28 The thoracodorsal artery perforator flap can capture the same dorsal thoracic fascial territory as does the scapular/parascapular flap,29,30 but it has a potentially much longer vascular pedicle that could reach beyond a zone of injury. If a perforator flap is not an option or has failed, more malleable muscle flaps such as the gracilis and latissimus dorsi are also available, which are not only important for the lower extremity, but also are truly “workhorse” flaps for throughout the body.31 Finally, the groin flap has been reborn as the superficial circumflex iliac perforator flap and is better explained in Chapter 22.
References
[1] Tagliacozzi G. De Curtorum Chirurgica per Insitionem Libri Duo. Venice: G Bindonus; 1597
[5] Barsky AJ. Filatov and the tubed pedicle. Plast Reconstr Surg Transplant Bull. 1959; 24:456–462
[6] Bingham HG, Moore CE. Farewell to Queen’s Hospital, Sidcup. Br J Plast Surg. 1976; 29(4):297–301
[8] Tamai S. History of microsurgery. Plast Reconstr Surg. 2009; 124(6) Suppl: e282–e294
[10] Acland R. A new needle for microvascular surgery. Surgery. 1972; 71(1): 130–131
[12] Morrison WA. Bernard McCarthy O’Brien. Br J Plast Surg. 1994; 47:204–205
16.2 Chapter 16A: The Anterolateral Thigh Flap for Lower Extremity
16.2.1 Introduction to the Anterolateral Thigh Flap for Lower Extremity
Following the initial description by Song et al,1 the utility of the anterolateral thigh (ALT) flap (▶ Fig. 16.2) by reconstructive surgeons has increased tremendously. It is now considered the workhorse soft-tissue flap for reconstructive surgeries in various regions of the body.2,3,4,5,6,7,8 The relatively consistent anatomy, minimum donor site morbidity, ability to reconstruct simple and composite soft-tissue defects, versatility, long pedicle, and satisfactory caliber of vessels are among the attributes that have led to the popularity of this flap and expanded its utility for both locoregional and free tissue transfer in different body regions including lower extremity reconstruction.
16.2.2 Attributes and Detriments
Attributes
• Relatively constant anatomy with slight anatomic variations.
• Ease of harvest when anatomic variation is understood.
• Ability to reconstruct complex soft-tissue defects.
• Minimum donor site morbidity when used as a perforator flap.
• Ability to harvest the flap in the supine and lateral positions.
• Allows a two-team approach when used for contralateral lower extremity reconstruction.
• Adequate pedicle length that maximizes reach when used for locoregional reconstruction.
• Adequate caliber of vessels suitable for free tissue transfer.
• Pliable flap that can be thinned without compromising flap vascularity.
• Ability to be used as a sensate flap especially for weightbearing areas such as heel reconstruction.
Detriments
• Skin graft may be required for donor site coverage when the flap width exceeds 8 or 9 cm at the mid-thigh.
• Even if direct donor site closure is possible, the mid-thigh scar may be considered nonaesthetic.
• Rare inconsistency of the perforator anatomy.
16.2.3 Flap Components
The ALT flap can be harvested as a cutaneous, fasciocutaneous, or musculocutaneous flap. It can also be harvested as a chimeric flap to include other tissues such as the rectus femoris, tensor fascia lata, or vastus lateralis muscles or even femoral bone, where the ALT flap itself is supplied by a separate perforator or even independent source vessel. The missing tissues at the recipient site will dictate the necessary size, thickness, and components of the ALT flap.
16.2.4 Anatomic Consideration
Vascular Anatomy
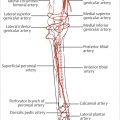
The blood supply of the ALT flap is primarily derived from the descending branch of the lateral circumflex femoral artery (LCFA). Clinical and anatomic studies have demonstrated that about 86% of the perforators follow a musculocutaneous course, while 14% follow a septocutaneous course.
Following its origin from the lateral aspect of the deep femoral artery, the LCFA courses posterior to the sartorius and rectus femoris muscles and divides into the ascending, transverse, and descending branches. The transverse branch passes laterally to pierce the vastus lateralis muscle, while the descending branch passes down in the septum between the rectus femoris and vastus lateralis muscles, giving off numerous muscular branches before anastomosing with the lateral superior genicular artery about the knee joint.
The flap pedicle length can be up to 10 to 13 cm with an arterial diameter of 2.0 to 2.5 mm at its origin, and a pair of accompanying veins of diameter from 1.8 to 3.0 mm.
Sensory Innervation
The skin of the ALT is supplied by branches of the lateral cutaneous nerve of the thigh and branches of the anterior cutaneous nerve of the thigh. The predominant nerve supplying the ALT skin territory is the anterior division of the lateral cutaneous nerve of the thigh. The lateral cutaneous nerve of the thigh (L2–L3) passes about 1 cm medial to the anterosuperior iliac spine (ASIS) and behind the inguinal ligament. It passes anterior to the sartorius muscle into the thigh. There its larger anterior branch then pierces the fascial lata about 10 cm below the ASIS to supply the skin of the ALT, while the posterior branch pierces the fascia higher to supply the skin of the lateral thigh.
Motor Innervation
The vastus lateralis muscle is supplied by a motor branch from the posterior division of the femoral nerve. This branch runs with the descending branch of the LCFA and can be included with the flap for coaptation with a motor branch at the recipient site when the vastus lateralis muscle is used for functional muscle transfer.
16.2.5 Anatomic Variations
The pattern and distribution of the vessels supplying the ALT cutaneous territory is variable and failure to understand these anatomic variations can lead to flap vascular compromise. The descending branch of the LCFA commonly arises from the LCFA but can originate from the deep femoral or common femoral arteries. A large-scale systematic review revealed that the majority of perforators to the ALT skin originate from the descending branch of the LCFA in 75 to 100% of cases. Origin from the transverse, ascending, and oblique branches has also been described with wide range of reported frequency. The same study revealed that septocutaneous perforators were present in about 20% of cases and ALT perforators were absent about 2% of the time.9
16.2.6 Patient Positioning
The flap is usually harvested with the patient in a supine position, but can also be harvested with the patient in a semilateral or true lateral position. The lower extremity is circumferentially prepped. Harvesting the flap from the contralateral thigh allows for a two-team approach.
16.2.7 Flap Design
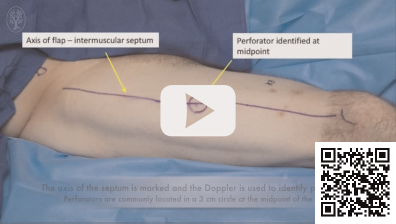
Routinely examine the patient in the preoperative area or in the operating room while awake to determine the location of the septum between the rectus femoris and vastus lateralis muscles. With a straight leg raise test, this septum may be palpated or even potentially visualized in patients with a thin body habitus. Otherwise, a line joining the ASIS and the superolateral border of the patella is marked and will represent the location of the septum, and potential location of septocutaneous perforators. Note that the septum is neither a straight line nor a fixed point, and its location could shift medially or laterally depending on the patient position on the table or if the lower extremity has undergone medial or lateral rotation. All markings will be more reliable with the patient supine and the lower extremity kept in neutral rotation.
A handheld audible Doppler is commonly used next to identify perforator signals, which are concentrated within a 3-cm-diameter circle centered over the midpoint of the septal line. The perforators themselves are most frequently found within the inferolateral quadrant of this circle. Musculocutaneous perforators, of course, will pierce the vastus lateralis muscle posterior to this line. When designing the chosen skin island, it is advisable to encompass more than one perforator, and to preferably shift the flap boundaries slightly posterior to the line joining the ASIS and superolateral patella so as to be able to capture both septocutaneous and musculocutaneous perforators, if present.
In patients with thicker thighs, identification of perforators with the audible Doppler examination can be misleading. Yu’s group proposed a very reliable, simple, and accurate model (ABC model) to guide perforator location.10 That is, there are usually one to three perforators in the ALT flap territory in predictable locations (A, B, or C). In an average person, perforator B is located near the midpoint, whereas perforators A and C are approximately 5 cm proximal or distal to perforator B. All three perforators are located approximately 1.4 cm lateral to the line joining the ASIS and superolateral patella, but again this can be highly variable.
A longitudinally oriented skin island is designed to incorporate the marked perforators. A skin island up to 35-cm long and 25-cm wide can be harvested on a single dominant perforator, but inclusion of more than one perforator is advisable. This will provide the surgeon the option of using more than one skin island, each based on a separate perforator, or just as a safety factor to prevent inadvertent flap twisting or devascularization.
Variations of the skin island design can place the perforator at centric or eccentric locations. Designing the skin island distally places the chosen perforator at an eccentric position proximally, and thereby increases the effective pedicle length when the flap is based on a proximal pedicle. When using the ALT as a distal-based pedicled flap for coverage of soft-tissue defects about the knee, the perfusion of the flap will be retrograde through communications with the superior lateral genicular system. In these circumstances, designing the skin island proximally on the ALT increases the effective distal pedicle length and enhances the arc of rotation and reach.
16.2.8 Flap Harvest
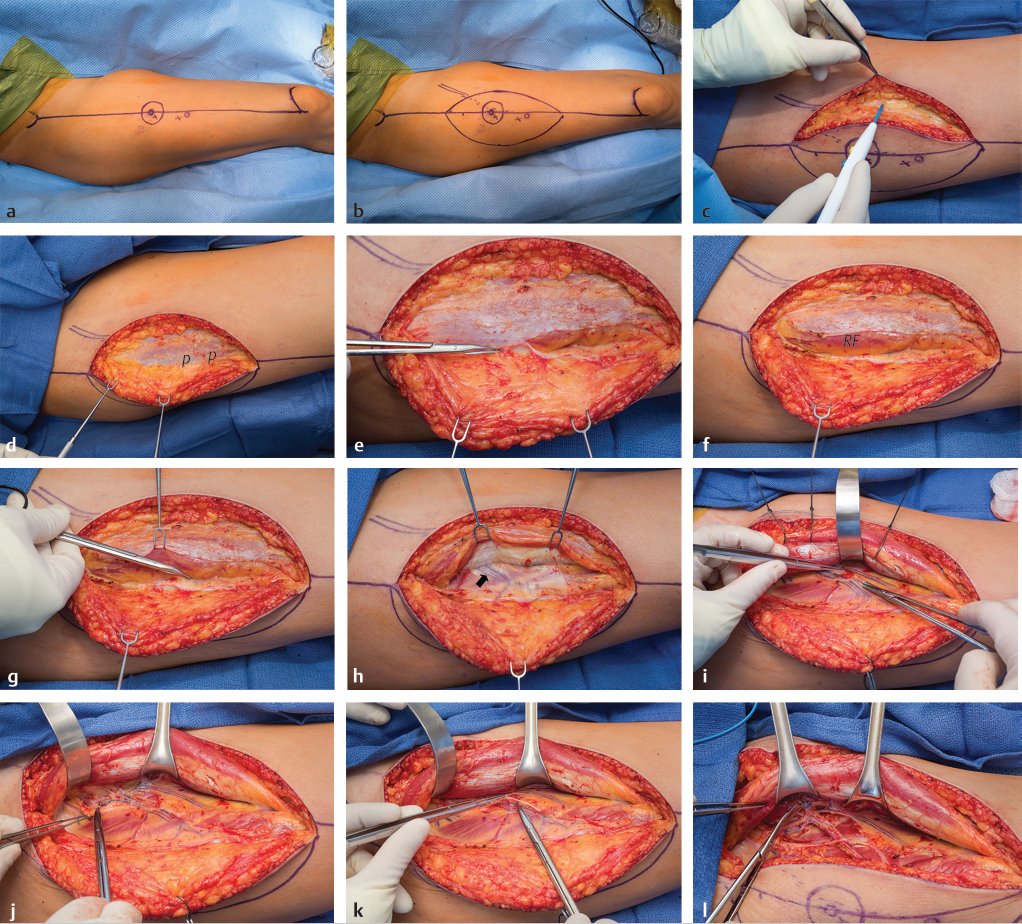
Suprafascial Approach
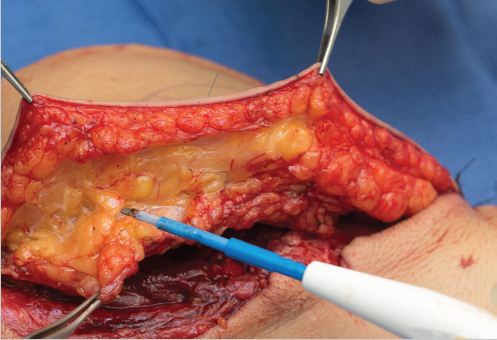
The medial flap boundary incision is made first and dissection is carried down to the level of the deep fascia (▶ Fig. 16.3 and ▶ Video 16.1). If additional soft-tissue bulk is necessary, the dissection could be beveled away from the flap to include more subcutaneous fat. The flap is then raised from medial to lateral in a suprafascial plane directly above the fascia lata, using cautery or tenotomy scissor until a skin perforator is identified piercing the fascia. Once perforators of adequate caliber are identified, the fascia is incised and opened medial to the perforators, and the remainder of the septum is opened to expose the descending branch of the LCFA. Dissection is then continued proximally toward the latter’s origin itself from the LCFA. While doing this, multiple motor branches will need to be carefully dissected away from the vascular pedicle and protected to avoid iatrogenic denervation of the thigh musculature, especially when the flap is used as a perforator flap without including part of the vastus lateralis muscle.
Video 16.1 ALT Flap for Lower Extremity Reconstruction. https://www.thieme.de/de/q.htm?p=opn/cs/20/7/12265275-1a6625fc
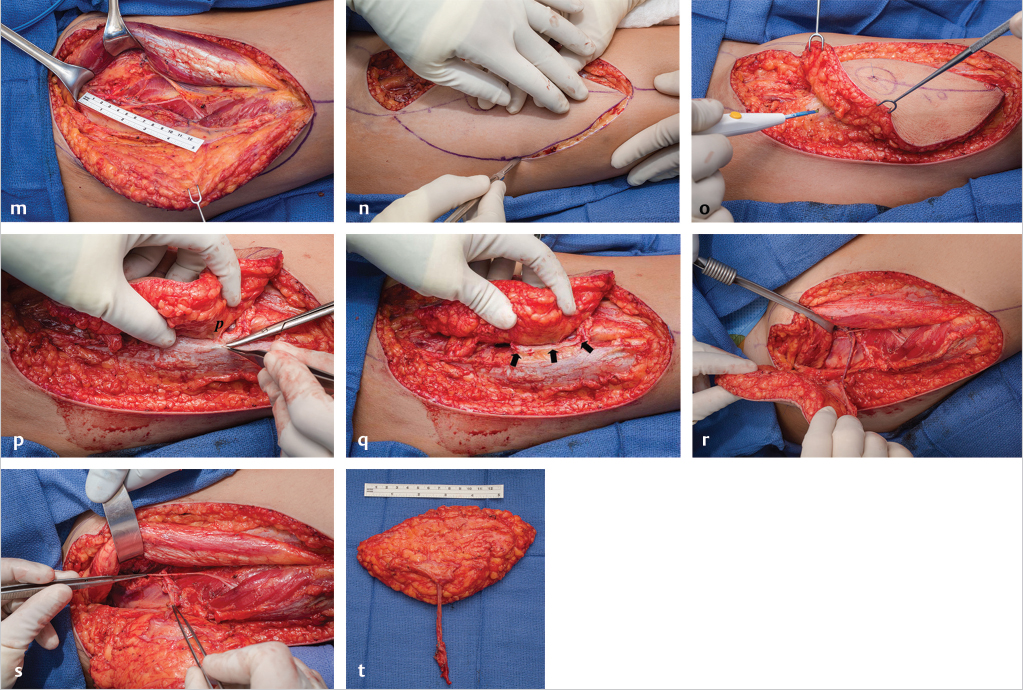
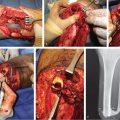
Fig. 16.3 Suprafascial anterolateral thigh (ALT) flap harvest. (a) A line is drawn from the anterosuperior iliac spine (ASIS) to the superolateral border of the patella. This represents the location of the septum between the vastus lateralis and rectus femoris muscles. At the midpoint of this line, a circle of 3-cm. diameter is drawn. Usually in the inferolateral quadrant of this circle a perforator can be found, here marked “x.” In addition, in this case a second perforator “x” was identified 5 cm more inferiorly. (b) Design of the skin island is best made to include at least two major perforators, and if placed eccentric to the perforators distally, it ensures not only a longer pedicle length, but also often a thinner flap at least in its distal portion. (c) The medial flap border incision is made first, and dissection carried down to the level of the deep fascia. Additional subcutaneous fat/fascia can be obtained while doing this by beveling the dissection in a more medial direction. (d) Next, dissection is performed in a medial to lateral direction directly above the deep fascia until the perforators (p) are visualized. (e) The fascia medial to the perforators is sharply incised and the septum between the rectus femoris and vastus lateralis muscles is visualized. (f) The remainder of the fascia is opened to completely expose the septum and the lateral border of the rectus femoris (RF) muscle. 8(g) One of the perforators (tip scissors) is seen emanating along the septum before entering the flap. (h) The rectus femoris muscle is retracted medially to expose the descending branch of the lateral circumflex femoral vessel (LCFV) pedicle (arrow) to the perforators. (i) Retrograde dissection of the pedicle commences from distal to proximal toward the descending branch of the LCFV proper. (j) Proximal pedicle dissection continues until the required length is obtained. (k) The motor branch to the vastus lateralis muscle (forceps) must be carefully separated from the vascular pedicle and preserved. (l) Here the vascular branch to the rectus femoris muscle has been clipped and is about to be divided, as will be any other distal branches if additional pedicle length is needed. (m) By following these steps, a 13-cm-long pedicle has been obtained. (n) Now the lateral flap border incision can be made. (o) Dissection will proceed from lateral to medial in the suprafascial plane. (p) This dissection stops when the previously selected perforators (p) are identified on their lateral side. The fascia is incised just lateral to them, preserving a small cuff of fascia with the flap. (q) Final narrow fascial cuff (arrows) seen around and protecting the two perforators as their intramuscular dissection is completed back to the source pedicle. (r) All dissection has been completed, making this an island flap connected to the thigh only by the source pedicle. (s) For a free flap, the pedicle is divided as far proximally as required, then as seen here passed underneath the motor nerve while being removed from the thigh. (t) The ALT free flap ready for transfer to the recipient site.
The extent of proximal pedicle dissection will be determined by the length and caliber of vessels required. When the ALT is used for locoregional reconstruction, a long pedicle is frequently needed to maximize reach. In these circumstances, the branch to the rectus femoris muscle can be safely divided to obtain an additional effective pedicle length.11,12 When the rectus femoris branch is divided, care should be taken to avoid extensive dissection around the rectus femoris muscle distally so the blood supply from minor distal pedicles can be maintained. The rectus femoris branch can also be divided if additional pedicle length is needed when using the ALT for free tissue transfer.
The lateral boundary incision is then made, and suprafascial dissection is continued from lateral to medial till the same perforators identified earlier are visualized. If the course of the perforator(s) is purely septocutaneous, they can easily be dissected down to the source vessel. If the perforators follow an intramuscular course, the usual tedious intramuscular dissection is performed with all muscular branches carefully ligated (muscle branches usually arise from the lateral and posterior surfaces of the perforators). A small cuff of fascia left around the perforator helps prevent pedicle twisting or avulsion, while also being something that can be held safely and maneuvered with forceps. Intramuscular dissection is usually carried out in a retrograde fashion back to the source vessel, but antegrade dissection is also possible.
When the flap is used as a sensate flap, the lateral cutaneous nerve of the thigh is included with the flap. The nerve is identified proximally below the fascial lata and additional length can be obtained by extending the dissection proximally toward the ASIS.
Video 16.2 Subfascial Harvest of the ALT Free Flap. https://www.thieme.de/de/q.htm?p=opn/cs/20/7/12265276-d0a33d54
Suprafascial dissection is preferred when a thin flap is required. This allows also for preservation of sensory nerves in the thigh that traverse over the fascia. In addition, preserving the fascia may minimize donor site morbidity such as by preventing muscle herniation.
Subfascial Approach
The medial flap boundary incision is made first and dissection is carried down to the level of the deep fascia (See ▶ Video 16.2). When bulk or deeper fascia is needed, dissection is beveled away from the flap to incorporate more subcutaneous fat or an extension of the fascia. The fascia is sharply incised directly exposing the underlying rectus femoris muscle. Subfascial dissection is then carried from medial to lateral, until the septum between the rectus femoris and vastus lateralis muscles is identified. If septocutaneous perforators are identified, the remainder of the dissection is straightforward. If no septocutaneous perforators are present (as in the majority of cases), careful dissection is then continued over the vastus lateralis muscle to identify musculocutaneous perforators. Once perforators of adequate caliber are identified, intramuscular perforator dissection back to the descending branch, further pedicle dissection, and nerve preservation continues in a fashion similar to the suprafascial approach. After completion of perforator and pedicle dissection, the lateral boundary incision is made, and subfascial dissection is carried from lateral to medial, back to the first deep fascial incision.
16.2.9 Pitfalls, Flap Modification, and Tips to Optimize Outcomes
Perforator Dissection and Flap Handing
Adequate hemostasis is of paramount importance as in all perforator flap surgery to allow unimpeded visualization of the perforator at all times. All side branches must be carefully clipped, then divided, or gently cauterized with bipolar cautery. Care should be taken to avoid placing excessive traction on the perforators, as an iatrogenic traction injury to the perforators can occur even with slight traction.
Absent or Inadequate Lateral Thigh Skin Perforators
The surgeon should be prepared for the possibility of absent or inadequate caliber lateral thigh skin perforators, as this has been reported to occur in about 2 to 4.3% of cases.9,13,14,15,16,17,18 The presence of adequate skin perforators should be confirmed first before making the lateral incision. If the vessels supplying the skin island are too small to perform safe intramuscular dissection, the flap can be raised as a musculocutaneous flap to include the vastus lateralis muscle with the skin island.
In the rare situation that a perforator is absent or injured, the surgeon has the following options:
• Perform audible Doppler mapping of the medial thigh skin. Dissection is then carried in a medial direction to identify those skin perforators. This is based on the knowledge that there is an inverse relationship between the number and caliber of vessels in the lateral and medial thigh regions. Almost always a perforator can be found in the medial thigh, and a flap harvested through the same medial incision without creating another donor site defect.13,19
• A freestyle approach is followed to look for vessels supplying the region of the thigh.15
• The lateral circumflex femoral vessel is exposed and the vessel branch supplying the tensor facial lata muscle is identified. A tensor fascia lata perforator flap is then harvested based on this vessel.
• Closure of the initial donor site and selection of an alternative flap.
Use of the Anterolateral Thigh as a Pedicled Flap
The ALT flap can be used as a pedicled or island flap for groin, medial thigh, perineum, trochanter, posterior thigh, contralateral inguinal, and knee coverage (see Chapter 15A: The Island Perforator Flap). Multiple techniques have been described to maximize reach when used as a locoregional flap, such as passage through a subcutaneous tunnel, placement below the rectus femoris muscle, and transmuscular transfer to the posterior thigh.11 The vascular branch to the rectus femoris muscle can be safely divided to maximize effective pedicle length.12
When the ALT flap is used for knee coverage, it must be based distally on retrograde blood flow through the communication between the superior lateral genicular system and the descending branch of the LCFA or, less commonly, the profunda femoris artery. The pivot point is usually 3 to 10 cm above the patella. A higher incidence of venous congestion has been reported when the flap is based distally. To mitigate against this unfavorable outcome, the pedicle should routinely be dissected proximally to obtain extra length so that supercharging the flap will be facilitated if venous congestion occurs.20
The adequacy of distal retrograde blood inflow must also be confirmed before dividing the proximal pedicle. A vascular clamp applied temporarily to occlude the proximal pedicle will allow assessment of flap perfusion via the retrograde flow. If inadequate, the ALT flap can still be transferred as a free flap.
Use as a Flow-Through Flap
The ALT flap can be used as a flow-through flap in various situations. For example, when a vascular segment in the lower extremity is missing due to trauma or surgical resection, the ALT pedicle can be used to bridge the missing vascular gap. In addition, the distal end of the pedicle can be used to provide inflow/outflow to another free flap, serving as the recipient site for the latter.
Fig. 16.4 Primary anterolateral thigh (ALT) flap thinning. Note the difference in shape and size of fat lobules between the superficial and deep subcutaneous layers. The larger deep layer fat lobules are being removed along the thin, white fascial layer that is retained, which serves to separate them from the superficial layer.
Flap Thinning
When the ALT flap is used for pretibial, dorsum of the foot, ankle, or heel coverage, primary thinning of the flap can be considered especially in patients with excess subcutaneous fat. Thin flaps may have the ability to regain sensation quicker even when transferred as insensate flaps.
Flap thinning is performed while the flap is still being perfused before division of the pedicle to enable continuous monitoring of flap perfusion as well as obtaining adequate hemostasis. Preservation of a 2- to 3-cm soft-tissue cuff around the pedicle entrance into the flap is advised to maintain perfusion. To best accomplish this, the plane between the superficial and deep fat layers is first identified (▶ Fig. 16.4). The deep fat layer has larger fat lobules, while the superficial layer has smaller and more round lobules, the two usually separated by a thin fascial layer. Flap thinning requires removing the deep fat layer lobules until the superficial layer is reached. Further thinning can actually be performed up to the level of the subdermal plexus; however, flap survival after transfer may be more likely affected by how the flap is inset. Caution must then be observed if tension is excessive, or if a complex defect requires folding of the flap, as viability will be less reliable. Secondary flap thinning is a much easier and safer procedure, and should be a consideration until experience with primary flap thinning is reasonable.
Harvest as an Adipofascial Flap
This is particularly helpful when a thin flap is required (such as coverage of exposed tendons on the foot dorsum) in patients with excess subcutaneous fat. Raising an adipofascial flap follows the same technique as for subfascial ALT flap harvest. The dissection should be performed with a subfascial approach to preserve the vascular network that runs on the surface of the fascia. A minimum of 3 mm of fat should be preserved over the fascia to avoid damage to this fine vascular network.21
Adding Bulk to the Flap
Flap bulk can be increased by inclusion of the vastus lateralis, rectus femoris, or tensor fascia lata muscles, or by including more subcutaneous fat as by beveling away from the incisions of the flap boundaries themselves as needed.
Use for Tendon Reconstruction
The fascia lata component of the ALT flap has been described for primary tendon reconstruction.2 This is best done by keeping the more lateral and thicker fascia lata with the cutaneous paddle as a composite flap, then rolling it up as a tube to serve much like a vascularized tendon graft.
16.2.10 Donor Site Management
Primary closure of the donor site is preferable, but obtaining a flap of adequate size always takes precedence. A skin island with width exceeding 8 to 9 cm at the mid-thigh level may preclude direct donor site closure, which otherwise could result in a thigh compartment syndrome. The donor site may still then be closed primarily at the superior and inferior aspects, but the middle portion may require split-thickness skin grafting. Using a dermal regeneration template prior to skin grafting or tissue stretching devices will allow time for swelling to subside, which could permit even further or complete closure of the middle portion of the donor site. This may then avoid the risk of skin graft failure, particularly common after a suprafascial flap harvest, and definitely provide a better aesthetic outcome.
Alternatively, synchronous local advancement flaps based on other perforators (e.g., keystone or V-Y flaps) may facilitate complete primary donor site closure, but at the price of additional scarring of the thigh. Preoperative and postoperative thigh tissue expansion has also been described to avoid the need for a skin graft.22,23
Another option, if a wider skin island is needed, is to select a long but narrow longitudinal design. This will require the preoperative identification of at least two major perforators within that design based on that same source vessel, so that the skin paddle can be split into two islands, each based on an individual perforator. The skin islands can then be inset side by side at the recipient site using the “kiss” principle to provide the necessary coverage width, while still allowing primary closure of the donor site.24
16.2.11 Case Examples
Case 1: Locoregional ALT flap
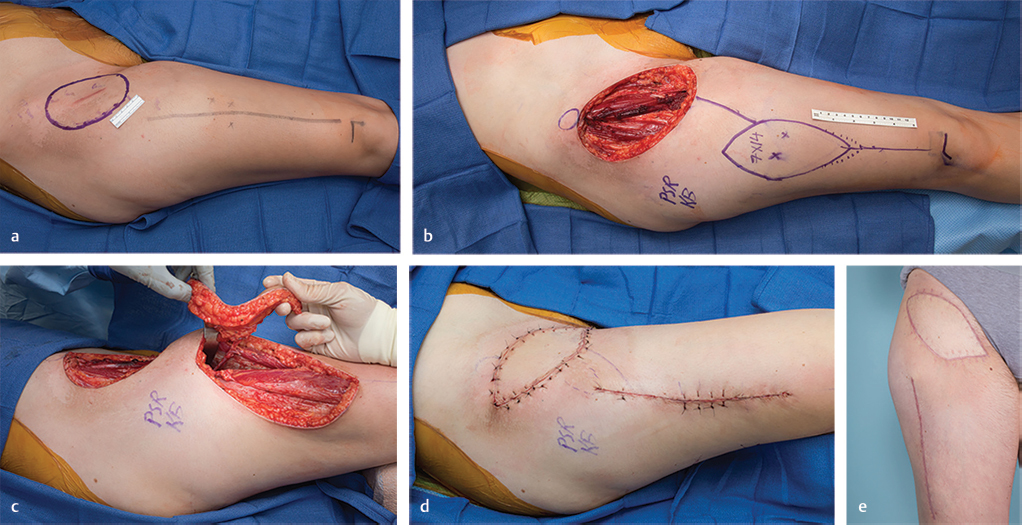
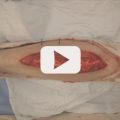
A 19-year-old woman had a right groin mass excised that proved to be a synovial sarcoma (▶ Fig. 16.5). Following neoadjuvant radiation, radical sarcoma excision resulted in a right groin defect requiring soft-tissue coverage. This was achieved by transposition of an ipsilateral island ALT flap.
Case 2: ALT Free Tissue Transfer
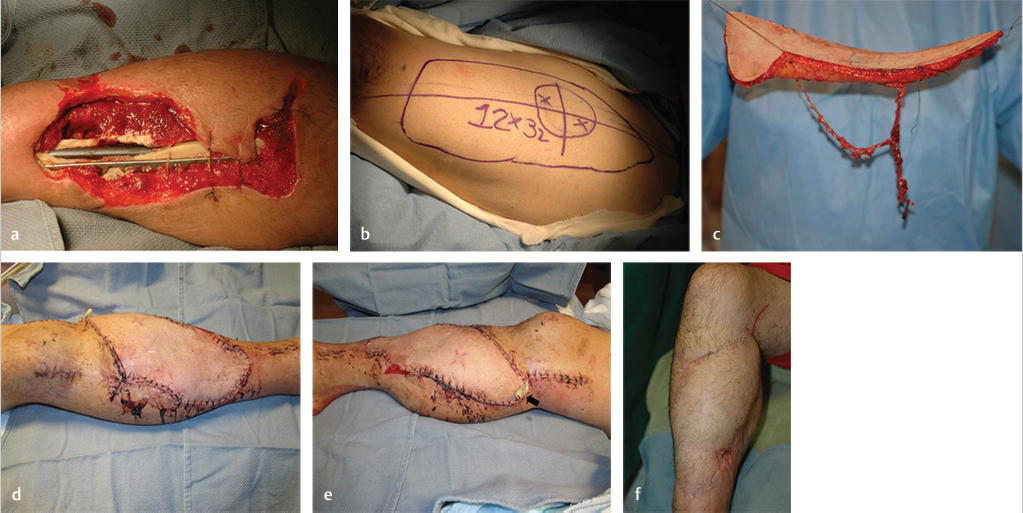
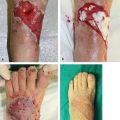
Following a motorcycle accident, this young man sustained a comminuted right tibia/fibula fracture with extensive soft-tissue degloving. Single vessel runoff remained, making this a Gustilo grade IIIC tibial fracture. Following plate and intramedullary rod fixation, the large defect was covered elsewhere with a latissimus dorsi free muscle flap that was unsuccessful. As a backup option for this now chronic wound, a contralateral ALT free flap was planned (▶ Fig. 16.6), not only to obtain another huge soft-tissue flap that was needed, but also to have a long pedicle with large caliber vessels that would reach the popliteal vessels outside the zone of injury and permit facile end-to-side microanastomoses. That proved to be successful, allowing limb salvage.
Fig. 16.5 Locoregional anterolateral thigh (ALT) flap transfer. (a) Planned radical excision of right groin synovial sarcoma, with preoperative location (x) of ALT perforators. (b) Resultant soft-tissue defect in irradiated wound. An ipsilateral ALT flap was designed about the previously marked perforators (x). (c) Following a suprafascial dissection, an island ALT flap was raised based on a single perforator. The retractor permitted visualization for enough pedicle lengthening that allowed sufficient reach to the defect and also creation of a subcutaneous tunnel through which the flap would be passed. (d) Island ALT flap inset with simultaneous primary donor site closure. (e) Excellent contour at 4-month follow-up. Vertical linear scar typical of mid-thigh donor site residue.
16.2.12 Postoperative Care Protocols
Extreme vigilance and attention to details are required to identify early flap compromise. Studies have shown that the most important factor in salvage of the failing free flap is early identification of any vascular compromise. Even pedicled flaps should undergo serial clinical examination by the surgical/nursing team (every 2–4 hours) starting in the recovery area for the first 24 hours. After the first 24 hours, the frequency of clinical examination varies based on individual cases.
Free flap monitoring typically follows a specified protocol of hourly clinical and, if nothing else is available, audible Doppler examination by a nursing staff specifically trained to care for free flap patients. Familiarity and experience of the nursing staff with the management of free flap patients has enabled care for this subset of patients on a general plastic surgery hospital floor. Admission to an intensive care unit is then rarely needed following lower extremity reconstructions with free flaps unless the general health of the patient and associated medical comorbidities require a higher level of care. Patients are maintained on strict bed rest for 5 days, followed by gradual resumption of lower extremity dangling.
16.2.13 Conclusion
The ALT perforator flap is a versatile flap often used in lower extremity reconstruction. The relatively constant anatomy, adequate pedicle length and caliber of vessels, ability to modify flap components based on the recipient site needs, minimal donor site morbidity, and excellent reported outcomes are among the reasons that have led to widespread adoption and utilization as both a pedicled and as a free flap, for challenges that so commonly present in the lower extremity.
Fig. 16.6 Anterolateral thigh (ALT) free flap. (a) Motorcycle accident with chronic open right tibia fracture after failed latissimus dorsi free muscle flap done elsewhere. (b) Huge contralateral ALT free flap designed about two perforators (x) identified using audible Doppler. (c) ALT free flap after subfascial harvest, supplied by two perforators connected to the descending branch of lateral circumflex femoral vessel (LCFV) pedicle. (d) Anterior view of inset ALT free flap and (e) medial view demonstrating the near circumferential extent of coverage following wound preparation. In addition, a corner of the flap was inset into the incision needed to expose the popliteal vessels that were used as the new recipient site (arrow), so as to minimize the risk of compression of its long pedicle that was needed to reach the latter. (f) Early successful coverage of this extensive right leg defect.
References
[22] Hallock GG. The preexpanded anterolateral thigh free flap. Ann Plast Surg. 2004; 53(2):170–173
16.3 Chapter 16B: The Gracilis Muscle Free Flap
16.3.1 Introduction to the Gracilis Muscle Free Flap
The advent of the realization of the clinical value of microvascular tissue transfers soon followed McLean and Buncke’s1 use of the omentum to cover a scalp defect, and Daniel and Taylor’s2 first successful composite tissue transfer. But imperfections of the latter’s groin flap such as anatomical inconsistencies rapidly led to the investigation for more pragmatic donor sites. The rebirth of muscle flaps around the same time frame as an important entity can be traced to Orticochea,3 who raised large peninsular flaps from the thigh safely without delay maneuvers, with reliability ensured by inclusion of the underlying gracilis muscle. However, without microsurgical capabilities, this had to be a multistaged regional flap for coverage of an exposed ankle bone.3 Quickly thereafter, the gracilis muscle (▶ Fig. 16.7) alone proved to be a reliable free flap donor site when Harii et al4 showed its utility for functional restoration of facial paralysis. Musculocutaneous free flap versions that followed were not always completely viable if the skin paddle had a vertical axis, but Yousif et al5 in cadaver studies proved this was because the “true” perforasome of the perforators from the dominant pedicle of the gracilis muscle had a transverse orientation in the upper thigh.
The gracilis muscle indeed deserves the appellation as a “workhorse flap,” as it has been found to solve all conceivable problems within the “skin and its contents,” the realm of the “true” reconstructive surgeon. Often preferable about the upper portion of the lower extremity as a local flap (see Chapter 14B: The Gracilis Local Muscle Flap), the gracilis muscle is extremely versatile also as a free flap. As a musculocutaneous version, the transverse myocutaneous gracilis (TMG)6 or transverse upper gracilis (TUG)5,7,8 free flap in the appropriate candidate is a secondary option for breast reconstruction. The gracilis muscle can be a dynamic transfer apropos for facial reanimation4,8,9 or restoration of a myriad of upper extremity functions.10 Although its small cross-sectional area limits potential power, ankle dorsiflexion has been achieved.11 More conventional roles in the lower extremity have been dictated when malleable fill is essential or coverage of small to moderate-sized defects needed.12 In the ubiquitous obese patient, this may be the best available thin flap that will not interfere with shoe wear and ambulation.
The gracilis muscle functions as a thigh adductor, knee and hip flexor, and medial hip rotator,8 but only a minimal deficit is noted if absent, so it is considered expendable.13 Potential donor site morbidity is minimal, and the resultant scarring on the medial thigh usually can be well hidden.13,14,15 It should be reiterated that all these attributes and unlimited roles demand that a complete knowledge of this donor site be mastered by all who use it!
16.3.2 Attributes and Detriments
Attributes
• Versatile “workhorse flap.”
• Expendable.
• Consistent anatomy.
• Expeditious harvest.
• Harvest possible in supine, lithotomy, or prone positions.
• Coverage of small and medium-sized defects.
• Thin contour.
• Autologous tendon reconstruction.
• Dynamic restoration.
• Virtually nonexistent donor site morbidity.
• Hidden donor site scar.
• Reliable to approach even in the morbidly obese.
Detriments
• Often found far more posterior in the medial thigh than believed possible.
• Not suited for large defects.
• Hyperesthesia of the medial distal thigh if the cutaneous branch of the obturator nerve is injured.
• Inconsistent reliability of vertical cutaneous paddle if composite flap.
• Significant arterial orifice atherosclerosis may make this unusable in end-stage peripheral vascular disease patient.
16.3.3 Anatomical Considerations
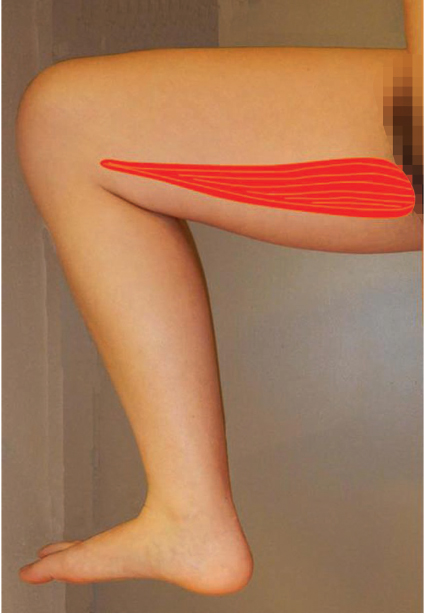
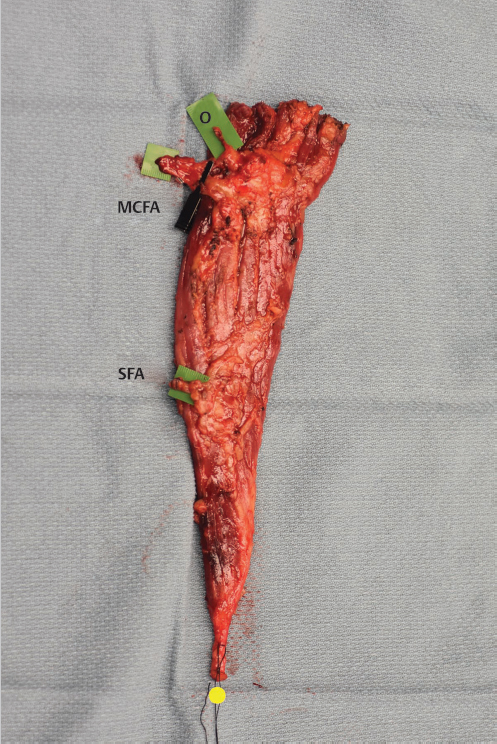
The gracilis muscle is found in the more posterior aspect of the medial thigh. It is the most superficial of the muscles of the adductor compartment, extending longitudinally for some 30 cm from its aponeurotic origin at the symphysis pubis and pubic arch to where its tendon inserts as part of the pes anserinus on the medial tibial condyle, where found inferior to the tendon of sartorius and above that of semitendinosus.7,8 The width of the muscle may be up to about 7 cm at the level of the major vascular hilum,16 then tapers distally toward its long tendon, thereby overall forming a somewhat triangular shape (▶ Fig. 16.8).
Fig. 16.8 The gracilis muscle has a triangular shape extending from a wide base at its origin until narrowing around its tendon distally (yellow dot). Here the medial circumflex femoral artery (MCFA) was the source of the dominant vascular pedicle, entering on the undersurface of the anterior border of the muscle. The anterior branch of the obturator nerve (O) entered obliquely just superior to the vascular hilum. A single distal secondary or minor pedicle originated from the superficial femoral artery (SFA).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree