Introduction
Although combined burn and other traumatic injuries are relatively uncommon (<5% of injuries in civilian burn centers, 5–20% of injuries in conflict settings), they are often complex management conditions, have competing care and rehabilitation needs, and require alignment of frequently segregated resources and services. Their relative rarity means that few health care systems and clinical teams have cared for enough patients with combined injuries to be facile in balancing competing care priorities. However, given 11 million people with burn injuries seek care each year globally and 45,000 burn-related hospitalizations occur in the United States alone, there is a high probability that burn and trauma teams will care for people with combined injuries.
For decades, mortality after combined injuries was significantly higher than isolated burn or trauma injuries, purportedly due to the additional physiologic and immunoinflammatory insult of a burn injury on the multiply injured patient and the challenges associated with orchestrating care for people with multidimensional injuries. However, more contemporary reports suggest that well-planned and organized trauma and burn care systems have reduced the disparity in mortality rates between combined and isolated injuries. This improvement has been in no small part due to knowledge gained from military experience with blast injuries and multidimensional trauma and disseminated to civilian trauma centers. ,
This chapter reviews the epidemiology and general management principles for patients who have experienced combined burn trauma and/or blast injuries. Detailed guidelines for the care of these complex patients are beyond the scope of this chapter; however, by introducing and highlighting key concepts, the reader should be more equipped to plan, recognize, and organize care for the multiply injured patient.
Epidemiology
Incidence and risk factors
A US single-center review of 10 years of experience (1990–2000) caring for 24,903 patients with isolated trauma, burns, and combined injuries found that 92.5% of patients had only trauma injuries, 7.1% had only burn injuries, and 0.4% had combined injuries. Similarly, a 6-year retrospective analysis (1998–2004) of the American College of Surgeons Committee on Trauma National Trauma Data Bank (NTDB) reported on 423,959 patients, stating 97% had only trauma injuries, 1.8% had isolated burn injures, and 1.2% had combined injuries. The most recent published analysis of the NTDB that included patients cared for between 2007 and 2015 showed an annual incidence of 0.4% to 0.6% of combined injuries. Burn injuries are more common among casualties during conflict (5–20% of all civilian and military casualties) and after disasters, , and combined burn and other injuries and blast injuries are markedly more common in these scenarios. ,
The risk factors for combined injuries generally reflect those for trauma generally—that is, younger men are more susceptible. Compared to burn injuries alone, most combined injuries are the result of motor vehicle collisions, falls during escape from fire, interpersonal violence, and explosions. , ,
Injury characteristics and care utilization
Most patients with combined injuries have both small burn sizes (i.e., <20% total body surface area [TBSA]) and low injury severity scores (ISS). , The nonburn injuries associated with combined trauma are generally, in decreasing order of prevalence, musculoskeletal, multisystem, intracranial, intrathoracic, and intraabdominal.
Analysis of the NTDB reported that patients with combined injuries are more likely to have longer critical care stays (mean 20 days vs. 14 for burn and 7 for trauma alone), ventilator days (mean 17 days vs. 13 and 9), and lengths of stay (mean 19 days vs. 10 and 8) compared to either injury alone. Similarly, they experienced a greater rate of complications, such as acute kidney injury (AKI; 2.5% vs. 0.7% for burn and 0.6% for trauma alone), acute respiratory distress syndrome (ARDS; 3.3% vs. 0.6% and 0.9%), venothromboembolism (more than twofold higher), hospital-acquired (e.g., pneumonia, central line-associated bloodstream infection, catheter-associated urinary tract infection, severe sepsis) and wound infections (more than threefold higher), unplanned intubations (more than twofold higher), and unplanned intensive care unit admissions (more than threefold higher), compared to burn or other injuries alone.
In some cases, care utilization can be optimized by not performing a full trauma evaluation (e.g., trauma series, extended focused assessment with sonography in trauma [eFAST], axial imaging) in patients with burn injuries who are at very low or no risk of having other injuries. As an example, flash burns or propane tank explosions, although dramatic and can cause significant burn injuries, do not generate enough energy to cause blast injuries. Therefore, without other reasons to perform a full trauma evaluation (e.g., jump from height to escape structure fire, obvious penetrating injury, exposure to overpressurization wave), such patients can be cared for with focus on their burn injury.
Mortality and injury severity adjustment
In the 1980s to 2000s, mortality after combined burn and trauma injuries was uniformly higher than for isolated injuries of similar severity, suggesting a synergistic effect of combined injuries on rates of complications and mortality. , The two US single-center and NTDB reports aforementioned , and another from the National Trauma Registry for England and Wales reported mortality rates two to six times higher for patients with combined injuries compared to those with isolated injuries even when stratified by overall injury severity. In the early 2000s, even patients with combined trauma and minor burns (i.e., <20% TBSA) had nearly a fivefold increased risk of mortality compared to those with minor burns alone.
In spite of advances in both burn and trauma care over the last two decades, the hypothesis that mortality decreased significantly since these landmark analyses has been refuted by an updated analysis of the NTDB by Grigorian et al. in 2018 that included 6,539,211 trauma patients, 152,686 burn patients, and 33,813 combined injury patients. Identified trends included the observation that the impact of burn injuries on the mortality of patients with minor trauma (i.e., ISS <16) was lower in 2007 to 2015 compared to 1994 to 2002 (odds ratio [OR], 2.45; 95% confidence interval [CI], 2.26–2.66 compared to OR, 4.04; CI, 4.51–4.66). Meanwhile, the impact of burn injuries on mortality among patients with severe trauma increased (OR, 1.37; CI, 1.29–1.47 compared to OR, 1.26; CI, 1.05–1.51). The impact of the severity of trauma on mortality in combined burn-trauma patients with larger burns (i.e., ≥20% TBSA) was negligible. Conversely, an increase in burn size was associated with a near linear increase in mortality for combined injury patients. The largest impact was noted in patients with minor trauma (i.e., ISS <16), very severe trauma (i.e., ISS ≥26), and smaller burn sizes (i.e., TBSA <20%). However, for patients with large burn injuries (i.e., TBSA ≥20%), ISS had only a negligible contribution to mortality among people with combined injuries. Taken together, these data suggest that there has been little progress with regard to mortality rates of people with combined large burns and trauma regardless of injury severity. The results contrast significantly with literature demonstrating major improvements in survival for patients with increasingly larger burn sizes and more severe injury severities. These more contemporary results suggest that even small burns or other injuries result in increased mortality for people with combined injuries without more strategic approaches to care. Whether this is due to synergistic immunoinflammatory insults, additional challenges to already complex care, or both remains unknown.
Note that comparing and adjusting for injury severity among patients with combined burn and other injuries is challenging. ISS considers burn size but does not differentiate between second- and third-degree burns above 10% TBSA, uses categoric classifications for burn size, and includes inhalation injury in estimating overall anatomic injury severity for patients with burn injury. As a result, ISS has poor performance for predicting burn-related mortality that is improved when age and burn size are included as continuous variables (i.e., burn ISS [BISS]).
Models that adjust for ISS in addition to burn size and inhalation injury likely double-count the risk from those burn injury characteristics and potentially overstate the contribution of risk from each injury characteristic. This was confirmed by Janak et al. in 2018 when they compared ISS systems among US casualties during operations in Iraq and Afghanistan between 2003 and 2013. The authors found that use of traditional ISS (i.e., using abbreviated injury scores for all injuries, including burn and inhalation injury) and nonburn ISS (i.e., age, burn size, inhalation injury, and nonburn ISS) resulted in different interpretations for the effect of burn size on mortality. Specifically, they found that the association of burn size with mortality was obscured by the inclusion of traditional ISS. Therefore it is likely better practice to use the nonburn ISS method and/or BISS when modeling mortality for people with combined burn and other injuries. ,
Pathophysiology of shock and organ dysfunction after combined injury
Shock
Shock occurs when the delivery of oxygen and metabolic substrates to tissues and cells and removal of metabolites are insufficient to maintain normal aerobic metabolism. The concept of mal- or hypoperfusion highlights the imbalance between substrate delivery (i.e., supply) and substrate requirements (i.e., demand) that occurs at the cellular level during shock. Tissue hypoperfusion and cellular injury activate a cascade of cardiovascular, neuroendocrine, and immunoinflammatory responses designed to compensate for inadequate oxygen delivery and metabolite removal. The pathophysiologic sequelae of shock may be due to the direct effects of inadequate tissue perfusion on cellular and tissue function and/or one’s overzealous responses to the shock state. Shock in patients with combined injuries is common and can result from one or more of seven etiologies. In 1934, Blalock described four categories of shock: hypovolemic, vasogenic, neurogenic, and cardiogenic.
-
■
Hypovolemic shock, the most common type, results from loss of circulating blood or its components.
-
■
Distributive or vasogenic shock, as seen in sepsis or anaphylaxis, results from decreased resistance to blood flow within capacitance vessels of the circulatory system, causing an effective functional decrease in circulating volume.
-
■
Neurogenic shock is a form of vasogenic shock in which spinal cord injury (or spinal anesthesia) causes vasodilatation.
-
■
Cardiogenic shock results from failure of the heart’s pump function as might occur with dysrhythmia, heart failure, or severe blunt cardiac injury.
Two additional categories of shock have been added to those originally proposed by Blalock:
-
■
Obstructive shock occurs when circulatory flow is mechanically impeded (e.g., pericardial tamponade, tension pneumothorax, pulmonary embolism).
-
■
Traumatic shock is often seen in multisystem trauma with significant soft tissue and bony injuries with or without significant hemorrhage that occurs secondary to the release of toxins from injured tissues that are normally excluded molecules, termed damage-associated molecular patterns or danger signals, and secondary upregulation of proinflammatory mediators that can create a state of shock that is much more complex than simple hemorrhagic shock.
Burn shock is a seventh and unique shock state that manifests as a combination of distributive, hypovolemic, and (occasionally) cardiogenic shock caused by a cascade of systemic inflammatory activation, oxidative stress, and ischemia-reperfusion injury. Implicated pathways and mediators include complement, cytokines (interleukin 2 [IL-2], IL-4, IL-10, granulocyte-macrophage colony-stimulating factor, interferon γ, IL-5, IL-13, IL-17, IL-6, IL-8, tumor necrosis factor α, IL-1β, monocyte chemotactic protein 1), macrophage activation, nitric oxide synthetase upregulation, and mast cell and platelet degranulation. Additionally, microvascular thrombosis within and around the burn wound causes endothelial activation (e.g., glycocalyx shedding), consumptive coagulopathy, and fibrinolysis, which can exacerbate the coagulopathy of hemorrhagic shock.
Early identification and effective management of shock are imperative because cumulative oxygen debt (i.e., oxygen required to oxidize lactic acid after anaerobic cellular respiration) is associated with poor outcomes. As an example, in over 250 high-risk surgical patients, the calculated oxygen debt correlated directly with organ failure and mortality. The maximum oxygen debt in nonsurvivors (33.2 L/m 2 ) was greater than that of survivors with organ failure (21.6 L/m 2 ) and survivors without organ failure (9.2 L/m 2 ). In addition, the total duration of oxygen debt and the time required to restore perfusion correlated with outcome. Survivors were able to reverse the oxygen debt-induced injury, while nonsurvivors were unable to recover from the oxygen debt. Thus the magnitude of the oxygen debt, its rate of accumulation, and the time required to correct it all correlate with recovery from shock and survival.
It is difficult to directly measure the oxygen debt during resuscitation of injured patients. Easily obtainable clinical parameters, such as arterial blood pressure, heart and respiratory rate, shock index, urine output, central venous pressure (CVP), and end-tidal carbon dioxide, are poor proxies of tissue perfusion. Therefore surrogate parameters have been sought to estimate the oxygen debt, such as serum lactate and base deficit (BD). Experimental studies have shown that serum lactate and BD correlate with both oxygen debt and mortality. , BD is the amount of base in millimoles that is required to titrate 1 L of whole blood to a pH of 7.4 with the blood fully saturated with oxygen at 37°C (98.6°F) and a Paco 2 of 40 mmHg. It is usually measured by arterial blood gas analysis. Good correlation between the BD and survival has been shown in patients with shock. In bluntly injured patients with a BD of 0 mmol/L at presentation, mortality was 8% compared with 95% mortality among patients with a BD above 25 mmol/L. Studies among patients with burn injuries have shown similar positive correlations between BD and mortality. Differentiating types of shock in patients with combined injuries is difficult and requires a systematic approach to evaluation and treatment, which is described in subsequent sections.
Organ dysfunction
In addition to the specific pathophysiologic features of burns and other injuries alone, people with combined injuries, particularly severe injuries, are at higher risk of complications, such as AKI, ARDS, and multiple organ dysfunction (MOD), and are more likely to have long-term quality of life challenges. , Development of MOD is one of the major risks for mortality among those who survive the first hours and days after severe injury. One of the earliest pathophysiologic hypotheses of MOD revolved around the observation of a bimodal distribution of MOD after injury. The initial peak (i.e., first hit, traumatic injury, hemorrhage in people with combined injuries) was attributed to a massive insult that induced profound systemic inflammation that precipitated MOD. The subsequent peak (i.e., second hit) was attributed to a subsequent reactivating event (e.g., infection, transfusion, surgery, increasing systemic inflammation from burn injury among those with combined burn and other trauma) in an already primed or vulnerable host.
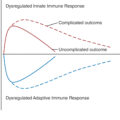
Since these early hypotheses, studies of the pathophysiology of posttraumatic MOD and burn shock , , suggest that injury triggers two simultaneous, opposite responses: proinflammation and immune suppression that result in synergistic immunoinflammatory dysregulation. Analyses of the Glue Grant database confirmed that the genomic expressions of pro- and antiinflammatory responses were simultaneous (i.e., not compensatory) and not necessarily equivalent. Proinflammation manifests as systemic inflammatory response syndrome that results from activation of the innate immune system and is associated with early MOD. Suppression or inhibition of the adaptive immune system and apoptosis limits proinflammation and manifests as a preconditioned homeostasis to protect against additional insults and promote healing. However, excessive and/or prolonged inhibition of the adaptive immune system can degenerate into severe systemic immunosuppression that is associated with immunoparalysis, impaired wound healing, sepsis, and late MOD (i.e., persistent inflammation-immunosuppression catabolism syndrome). , Ultimately, MOD is related to the intensity, duration, balance, and interplay between these basic immunoinflammatory responses ( Fig. 35.1 ). These responses do not simply cancel one another out but interact, disrupt, sustain, and even amplify one another after combined burn and trauma injuries. ,
Immunoinflammatory responses after combined burn and other injuries. The “1st Hit” represents initial injury; the “2nd Hit” represents subsequent insult from progressive inflammation from combined burn injury, major surgery, massive transfusion, and/or infection. MOD , Multiple organ disfunction; PICS , persistent inflammation-immunosuppression catabolism syndrome; SIRS , systemic inflammatory response syndrome.
Initial assessment and management
The adage, “Burn-injured patients are trauma patients first,” stems from shared etiologies and the risks associated with delayed and/or missed injury-related diagnoses. Given that motor vehicle crashes, falls during escapes from structure fires, and explosions can all exert sufficient forces to cause a multitude of injuries, a high index of suspicion must be maintained, and a systematic approach to the injured patient should be used. This begins in the prehospital setting where first responders and emergency medical service providers should use standardized evaluation, management, and destination triage algorithms to ensure that patients with burns and potential combined injuries are cared for in the appropriate first center. ,
Once at a hospital, the initial goal is to identify and alleviate any potential life-threatening or life-altering injury (e.g., impending airway loss, pneumothorax, uncontrolled bleeding, major burn) without delaying definitive care. Providers and trauma teams should use an ABCDE (airway, breathing, circulation, disability, exposure) approach to the primary survey of injured patients like that espoused by the American College of Surgeons Committee on Trauma Advanced Trauma Life Support, American Burn Association (ABA) Advanced Burn Life Support, British Burn Association Emergency Management of the Severe Burn Course, and others (e.g., World Health Organization Basic Emergency Care Primary Trauma Care). Special ABCDE-related considerations regarding patients with combined injuries are reviewed in this section.
Airway
Inhalation injury is covered comprehensively elsewhere in this text; however, because this chapter may be useful to emergency and trauma care providers as well, salient points in the airway management of patients with combined injuries are briefly reviewed. In addition to usual considerations for basic and advanced airway management (e.g., airway obstruction, depressed level of consciousness, hypoxemia/hypercarbia, refractory acute pain), patients exposed to superheated gases, steam, and smoke require additional attention due to potential consequences of inhalation injury. There are three types of inhalation injury: upper and conducting airway injury from heat; alveolar injury from smoke irritants; and systemic toxicity from carbon monoxide, cyanide, and other poisonous inhalants.
Upper airway injury can result in airway obstruction. This is most common in patients with deep head and neck burns, intraoral and hypopharyngeal burns, large burn size, and need for resuscitation due to their burn injury and/or other injuries. Other signs and symptoms, such as carbonaceous sputum and singed facial hair, are relatively poor predictors of requiring intubation. Without high-risk features, airway loss is rare; if it does occur, it takes time (and often resuscitation volume) for edema to develop. Frequent serial examinations in a continuously monitored setting for voice quality and subjective sensation of changes in work of breathing are important in such patients. Patients with burn injuries greater than 50% TBSA are rarely able to be managed throughout resuscitation without a definitive airway and should be intubated when/where it is most safe. Young children, who have a conical periglottic airway and small cricoid ring, are at greater risk of airway loss compared to adults as described by Poiseuille’s law (i.e., airway resistance is proportional to the length of the airway and viscosity of the inhaled gases, and inversely proportional to the radius to the fourth power).
Managing the airway of a patient with combined burn and trauma and inhalation injury is complex and should be done by the most experienced airway provider. Skin loss or eschar around the face and neck, need for inline cervical stabilization, edematous upper airway, and significant oxygen consumption/carbon dioxide production make it difficult to ventilate with bag valve masking even with an airway adjunct (e.g., nasal or oral airway) without rapid desaturation and airway loss. Use of advanced airway adjuncts may be required to prevent rapid desaturation should rapid sequence intubation fail (e.g., use of laryngeal mask airway). To reduce the risk of first attempt failure, intubation with video laryngoscopy, use of a bougie, and/or flexible fiberoptic laryngoscopy should be considered.
Smoke inhalation manifests clinically hours to days after injury. It is rarely a cause of severe hypoxemia during the initial evaluation, and other causes should be sought (e.g., pneumothorax, hemothorax, shock, aspiration, pulmonary contusion). However, patients with significant smoke inhalation have more profound immunoinflammatory activation, will require greater resuscitation than expected from commonly used fluid prediction formulae, and are at higher risk of pulmonary complications and death. Supportive care and inhaled heparin, bronchodilators, and N -acetylcysteine can be administered once the patient is stabilized and in an intensive care unit to mitigate the effects of inhalation injury.
Systemic toxicity from carbon monoxide should be presumed if the patient was burned in an enclosed space or presents with signs and/or symptoms of smoke inhalation injury. Initial care with fraction of inspired oxygen (FiO 2 ) of 1.0 should be maintained until a percent carboxyhemoglobin level has returned as normal. Recall that most pulse oximeters are unable to differentiate between hemoglobin bound with carbon monoxide and hemoglobin bound with oxygen. Therefore significant hypoxemia can be present with a normal oxygen saturation reading. An arterial blood gas analysis with carboxyhemoglobin level is required. There is no place for hyperbaric oxygenation in the multiply or critically injured patient given challenges with adequate monitoring and rapid intervention should it be needed. Cyanide toxicity presents as hypotension and lactic acidemia despite earnest resuscitation. As there is no clinically useful test for cyanide, and delayed treatment can be fatal, hydroxocobalmin, the hydroxylated active form of vitamin B12 should be administered promptly when the history and syndrome are consistent with cyanide toxicity. Hydroxycobalamin chelates cyanide and forms renally excreted cyanocobalamin. Unfortunately, hydroxycobalamin administration in both healthy volunteers and people with inhalation injury is associated with oxaluria, oxalate nephropathy, and AKI. Therefore patients without hypotension and persistent lactic acidemia despite immediate resuscitation should not be given the anecdote.
Breathing
In addition to aspiration, severe inhalation injury, and usual traumatic causes for problems with oxygenation and ventilation, patients with deep and fully or near-circumferential burn injuries of the chest and trunk are at risk of hypoventilation often manifested by increased P peak and P plat . This can be addressed by incising the eschar (and only the eschar) along the infraclavicular, anterior axillary, and subchondral lines. Additional incisions can be made to connect these and across the abdomen as indicated if the release is insufficient.
Circulation
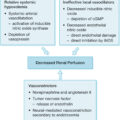
Hypotension in the multiply injured patient is bleeding until proven otherwise. The clinical consequences of burn-related inflammation and shock (e.g., capillary leak, vasodilation, myocardial dysfunction) often take hours to climax. Additionally, hypoperfusion to partial-thickness burns can result in sufficient cellular and ischemia-reperfusion injury to cause wound progression. Wound progression results in less potential for skin healing, greater surgical needs, and greater metabolic demands.
Sufficient peripheral and/or central venous access should be attained immediately, and the typical causes of shock among injured patients should be systematically evaluated and addressed. In cases when venous access cannot be immediately obtained, intraosseous access should be achieved without delay. Note that use of ultrasound and puncture through burned skin may be required. Suturing cannulas in place can prevent dislodgment when dressings and tape will not stick. The additional circulation step required for patients with combined injuries includes recognition of a major burn injury (i.e., one that requires resuscitation) or not. Major injuries are those that are 20% TBSA or greater or 10% to 15% TBSA or greater if very deep or on patients at the extremes of age. Numerous fluid prediction formulae are available to guide resuscitation. However, during the primary survey, less attention should be paid toward accurately estimating burn size, and more should be paid toward identifying and managing other injuries, starting grossly appropriate burn resuscitation, and planning next steps. Once recognized as having a major burn injury, the ABA advocates for administering the following initial rates of Ringer’s lactate solution:
-
1.
Adults: 500 mL/hour
-
2.
Larger children (age 6–13 years): 250 mL/hour
-
3.
Smaller children (age ≤5 years): 125 mL/hour
Several additional steps are important. Warmed fluid should be delivered via a pump (i.e., not with gravity or a pressure bag) to ensure consistent and accurate fluid administration. A urinary catheter should be placed after the primary survey to facilitate goal-directed resuscitation using urine output as a marker of end-organ perfusion. Assessment of urine output character (e.g., presence of heme pigments, concentrated urine) and volume per hour should be documented and assessed to guide changes in the hourly rate of fluid administration. Additional resuscitation calculations are typically not necessary when an hourly, goal-directed approach is used. In such complex patients, use of a decision-support tool (e.g., documentation flowsheet, Burn Navigator software) can reduce the cognitive burden on taxed and/or inexperienced providers and ensure appropriate changes to the hourly fluid rates are made. ,
Note that the edema that results from both the burn shock and resuscitation volumes causes an increasing distance from the peripherally accessed veins to the skin level. Therefore peripheral venous access sites should be regularly assessed, and central venous access should be considered for patients with burn injuries of 30% TBSA or more. Adjuncts to resuscitation (e.g., albumin, plasma, vasopressors, vitamin C, plasmapheresis, ultraearly excision) are typically not required in the first hours after injury while initial trauma evaluation is being performed.
Patients with hemorrhagic shock should be using damage control resuscitation (DCR) principles. The goal of DCR is to prevent the consequences of the lethal triad of progressive metabolic acidosis, coagulopathy, and hypothermia—all of which are also common in patients with burn injury. The key principles of DCR include:
-
■
Permissive hypotension
-
■
Restriction of crystalloid resuscitation
-
■
Earlier blood transfusion with balanced plasma and platelet to red blood cell transfusion ratios
-
■
Goal-directed correction of coagulopathy
Although there is little evidence to guide how concomitant hemorrhage-related and burn resuscitation should proceed, the neglect of one for the other is reliably unsuccessful. Theoretically, it would be ideal for some baseline burn resuscitation to continue while DCR is being performed. Practically, patients with hemorrhagic shock require all available access lines for whole blood or 1:1:1 transfusion and medications (e.g., tranexamic acid, calcium, muscle relaxant, vasopressor). Therefore, we recommend prioritizing DCR until anatomic hemorrhage control plasma-based with burn resuscitation occurring in the background to mitigate the risk of coagulopathy while addressing both hemorrhagic and burn shock. Frequent assessment of arterial blood gases and coagulopathy should be performed to optimize volume, blood component therapy, and crystalloid infusion rate. Once hemorrhage control is obtained, burn resuscitation is then prioritized. This can be operationalized by tasking team members (e.g., burn provider, nurse) with managing the burn resuscitation while DCR is also being performed.
Exceptions to this resuscitative strategy (i.e., permissive hypotension) include three main groups: people with combined burn injury, patients who are elderly, and those with traumatic brain injury (TBI). Persistent hypotension in patients with burn injuries can lead to significant oxygen debt, wound progression, and risk of sudden cardiovascular collapse. Studies of permissive hypotension have excluded burn injuries for these reasons. For patients who are elderly, many of whom are on antihypertensive medications at baseline, even a systolic blood pressure of 110 to 120 mmHg may be indicative of relative hypotension and cerebral hypoperfusion. Therefore resuscitation in these individuals must consider their expected baseline perfusion pressures. In civilian trauma centers, patients with multiple blunt injuries commonly encountered and the most critical of their injuries is often a TBI. In the setting of potential TBI, hypotension remains the best-defined risk factor known to produce a secondary insult to the injured brain tissue and significant worsening of both survival and functional outcomes.
In patients at risk of high spinal cord injury, hypovolemia from hemorrhage should be sought and excluded before the diagnosis of neurogenic shock is considered. To assume that the cause of hypotension in a multiply injured patient is due to neurogenic shock without first evaluating and treating potential hemorrhage is often a costly mistake. In patients who have neurogenic shock, the severity of the spinal cord injury correlates with the magnitude of the cardiovascular dysfunction. Patients with complete motor and sensory deficits from spinal cord injury are over five times more likely to require vasopressors for neurogenic shock compared to those with incomplete lesions. Similarly, patients with high cervical spine injuries (i.e., C1–C5) are more likely to require cardiovascular intervention compared to those with lower cervical or high thoracic spine injuries. Most patients with neurogenic shock will respond to volume resuscitation alone, with adequate improvement in perfusion and resolution of hypotension. Administration of vasopressors (e.g., norepinephrine, phenylephrine) can improve peripheral vascular tone, decrease vascular capacitance, and increase venous return. Appropriate and rapid restoration of blood pressure may also improve perfusion to the spinal cord, prevent progressive ischemia of the spinal cord, and minimize secondary injury to the spinal cord. Restoration of normal hemodynamics should precede any operative attempts to stabilize the vertebral fracture.
Disability
During the disability assessment, examination of extremities that are at risk of single or multiple compartment(s) syndrome from deep (near) circumferential burn injuries and/or fractures, particularly in the setting of a major burn injury and impending large resuscitation, is required. Patients with combined burns and long-bone fractures are at particularly high risk of compartment syndrome and may require fasciotomy as both inflammation/capillary leak and resuscitation ensue. Serial examinations with or without measurements of compartment pressures are required for all injured extremities, particularly those with collocated burns and fractures. Erring on the side of decompression of the tense limb(s)/compartment(s) is prudent when patients are not able to participate in examinations.
Escharotomy to prevent or decompress polycompartment syndrome in extremities with circumferential deep burn injuries is rarely required immediately. The procedure can typically be performed in a more controlled environment after initial assessment and management have been completed. The goal is to have the escharotomy performed by the most experienced provider, in the cleanest environment practical, and before the edema and increases in compartment pressures exceed venous outflow and capillary perfusion pressures to nerves and muscles. Escharotomy incisions can bleed significantly. Therefore they should generally not be performed in the field or in an emergency unit within a burn center.
Exposure
Patients should be entirely disrobed with care to prevent hypothermia. Loss of the skin thermoregulation capacity after burn injury dramatically increases the risk of hypothermia in patients with combined injuries. Hypothermia during resuscitation has consistently been found to be associated with increased rates of complications and mortality in burn-injured patients. Additionally, hypothermia is one of the three pillars of the lethal triad and should be avoided in all trauma patients. Serial assessment of core temperature should be performed to ensure maintenance or improvement in body temperature. Techniques to improve body temperature are initially preemptive, such as maintaining a warm trauma bay, removing wet clothing, using warmed fluids/blood products and/or fluid warmer, keeping patients covered with warmed blankets, using mylar head coverings, and using warming lights when able. When patients are below 35°C and/or not rapidly warming with passive interventions, use of intravascular or intraesophageal warming catheters or probes, respectively, may be necessary. Forced air warming devices may exacerbate hypothermia, particularly when patients are wet (including from wound transudate) or in shock.
During the exposure portion of the primary survey, estimation of burn size and depth should occur with expert burn providers present when feasible to guide clinical decision making (e.g., need for escharotomy or escharectomy, resuscitation strategy, survivability). Exposure should not be performed multiple times by multiple teams or providers. Photos for the medical record and/or Lund and Browder-type diagram completion should be performed during this exposure. If resuscitation has not already begun, the estimated burn size can be used to calculate the initial hourly fluid rate using an accepted fluid resuscitation formula as discussed elsewhere in this text (e.g., modified Brooke formula, Parkland formula, rule of 10s).
Resuscitation end points and technique for patients with combined injuries
Resuscitation end points
Physiologically, shock begins when oxygen delivery (DO 2 ) falls below the tissue oxygen consumption (VO 2 ) requirements. A persistent mismatch between the DO 2 and VO 2 is associated with progressive organ dysfunction and death. Unfortunately, there are major limitations in our ability to quantify this mismatch and assess perfusion status. For patients with isolated burn injuries, urine output has been the enduring measure of end-organ perfusion that guides resuscitation. However, urine output is not an ideal resuscitation end point in patients with combined injuries for several reasons, including its limited utility for instantaneous monitoring. Further, heart rate, systemic arterial blood pressure, skin temperature, and urine output provide relatively little information about the adequacy of oxygen delivery to tissues. Due to these limitations, it is necessary to use other surrogates of tissue hypoxia.
During anaerobic metabolism, pyruvate is converted to lactate rather than being recycled by entering the tricarboxylic acid cycle. Therefore increases in blood lactate concentration are evidence of increases in the rate of anaerobic metabolism. Numerous studies have documented that high blood lactate levels (≥2 mmol/L) portend an unfavorable outcome in patients with shock. Serum lactate levels are also affected by hepatic and renal clearance, both of which can be depressed due to preexisting comorbidities or prolonged tissue dysoxia. BD has been shown to have prognostic value in patients with shock. Note that lactate and BD can be persistently elevated in patients with burn injury related to the tissue hypoxia in the zone of stasis, cellular dysoxia from the inflammatory state, and peripheral vasoconstriction in the setting of relative hypovolemia.
Numerous measures have been used as surrogates for intravascular volume and adequate resuscitation, including CVP. Similarly, the inferior vena cava (IVC) diameter measured by ultrasound correlates with the CVP and can be used to predict fluid responsiveness in critically injured patients. One study found that patients with IVC diameter less than 2 cm were almost uniformly volume responsive compared to those with IVC diameter greater than or equal to 2 cm. However, titration of resuscitation to CVP or IVC diameter has not been shown to improve survival, and adequate sonographic windows may not be available in patients with chest wall emphysema related to a pneumothorax or those with deep truncal burns.
Cardiopulmonary interactions are responsible for pulse pressure variation (PPV), which is a surrogate for stroke volume variation (SVV). Pulse pressure is the difference between systolic and diastolic blood pressures at specific points during the cardiac cycle. PPV is the difference in pulse pressure that occurs over a respiratory cycle and can be monitored by examining the differing morphologies of the arterial waveform in patients who are mechanically ventilated, not spontaneous breathing, and are in sinus rhythm. In mechanically ventilated patients with controlled respiratory effort (i.e., not overbreathing the ventilator, synchronous respirations), the positive pressure delivered with each breath forces blood from the lungs into the left heart, effectively increasing preload and stroke volume. Simultaneously, the positive intrathoracic pressure decreases venous return to the right heart, lowering right heart preload and stroke volume. The pulmonary transit time of the smaller right heart stroke volume accounts for the short delay in decreased left heart stroke volume manifested during exhalation. PPV increases in states of hypovolemia where decreased intrathoracic blood volume and ventricular filling pressures are more sensitive to changes in intrathoracic pressure that occur during a respiratory cycle with PPV. A PPV of 13% or greater has been shown to discriminate between patients in whom additional volume will increase their cardiac index (responders) and those that will not (nonresponders) with a sensitivity and specificity of 94% and 96%, respectively. A recent meta-analysis of PPV in septic shock reported a pooled sensitivity of 0.72 (CI, 0.61–0.81) and a specificity of 0.91 (CI, 0.83–0.95) in predicting fluid responsiveness. However, resuscitation using an end point PPV less than 13% has not been shown to improve meaningful outcomes in critical injury and has been only moderately useful in patients with major burn injury given the degree and persistence of relative hypovolemia during the first 72 hours of injury.
Arterial waveform analysis is a noninvasive way to measure cardiac output (CO) and further characterize PPV and SVV. CO, PPV, SVV, and other hemodynamic parameters can be measured continuously with a minimally invasive lithium dilution CO (e.g., LiDCO) or pulse contour analysis CO (e.g., PiCCO or FloTrac). These systems utilize a pulse contour CO algorithm to estimate CO and systemic vascular resistance in the absence of invasive techniques. There is some evidence that the information garnered from such devices is most useful at a single point in time and compared to another point in time; when used for continuous assessment, the serial values show limited reliability. In small clinical trials, use of devices that utilize arterial waveform analysis has shown some promise, but large, multicenter trials looking at patient outcomes have yet to be reported.
Other resuscitation monitoring techniques, including esophageal Doppler, transesophageal echocardiography, and near-infrared spectroscopy (NIRS), have been reported. NIRS offers continuous, noninvasive bedside monitoring of tissue oxygenation. It measures oxygenation in the tissue’s microvasculature and, thus, not only examines the adequacy of tissue perfusion but also provides a window to noninvasively study tissue metabolism. In the clinical setting, NIRS has been used for continuous monitoring of metabolic variables, including tissue O 2 availability, tissue O 2 consumption, tissue O 2 saturation (StO 2 ), and changes in StO 2 in diverse populations of patients (e.g., injury, sepsis, heart failure). , However, the exquisite sensitivity of the technique and the rapid and labile nature of peripheral perfusion in critically injured patients result in poor reproducibility, thus the clinical utility of the technology has not been realized.
Until more practical and quantitative methods are introduced to measure the cumulative effect of the oxygen deficit and subsequent host response, it is likely that we will continue to use a combination of surrogates of tissue perfusion to diagnose shock and guide resuscitation.
Resuscitation strategies
There is debate over the ideal technique for resuscitation of burn-injured patients, particularly for those with combined injuries and multiple shock states. Given that both burn and traumatic shock require resuscitation and prevention of coagulopathy in the multiply injured patient, standard burn resuscitation techniques may be insufficient. Although goal-directed resuscitation is certainly standard of care, the timing and use of specific end points and thresholds for changes in resuscitation are debated. Evidence from randomized trials suggests that goal-directed therapy that includes use of additional measures (e.g., CO with thermodilution, PPV) likely optimizes preload, reduces tissue hypoxia, and results in lower MOD scores, but may not have an impact on mortality. A systematic review and meta-analysis that included 20 studies evaluating real-time end points for resuscitation of burn-injured patients reported a decrease in mortality with the use of certain hemodynamic alternative end points (e.g., CO, surrogates of end-diastolic left ventricular volume) rather than hourly urine output alone (risk ratio, 0.77; CI, 0.42–0.85; P = 0.004). Given this evidence and the increased risk of mortality among patients with combined injuries, use of several complementary end points may be advantageous.
There have also been studies that reported significant increases in fluid administration compared to fluid prediction formulae when alternatives to urine output were used as resuscitation end points. As an example, a randomized trial performed by Aboelatta and Abdelsalam reported greater 72-hour fluid administration when CO by thermodilution (e.g., PiCCO) was used compared to Parkland formula alone. Studies of the computerized resuscitation clinical decision support tool, Burn Navigator, which analyzes fluid inputs, vasopressor use, usual hemodynamic parameters, and hourly urine output to recommend changes in resuscitation rates for burn-injured patients using a proprietary algorithm, resulted in significant reductions in burn shock without increases in total fluid volume administered within 24 hours of injury and well under the Ivy index threshold (i.e., >250 mL/kg within the first 24 hours of injury, which is a major risk factor for the development of abdominal compartment syndrome). ,
In summary, given the complexity of resuscitation of patients with combined injuries and potentially multiple shock states and the lack of clear evidence regarding optimal resuscitation end points, we recommend addressing causes of obstructive and hemorrhagic shock immediately and then using a combination of end points in association with a computerized resuscitation clinical decision support tool that includes urine output as an end point to manage burn and traumatic shock. An example algorithm that considers these end points and strategies, as well as resuscitation adjuncts and special considerations for patients with combined injuries, is provided in Fig. 35.2 .
Example algorithm to guide resuscitation for patients with combined burn and other injuries. CVP , Central venous pressure; Echo , echocardiography; IV , intravenous; MAP , mean arterial pressure; PAC , pulmonary artery or Swan-Ganz catheter; PLR , passive leg raise; POCUS , point-of-care ultrasound examination(s); PPV , pulse pressure variation; ScvO 2 , central venous oxygen saturation; SVV , stroke volume variation; UO , urine output.
Special care considerations for combined injuries
The most common nonburn injuries among patients with burns include skeletal, intracranial, chest, and intrathoracic injuries as well as multisystem injuries. Guidelines for each of these injuries were created to optimize their specific management strategies and do not necessarily address the needs of other injuries. Each of these injuries poses unique competing care requirements and deserves special consideration.
Skeletal injuries
There is little evidence about the coincidence and management of collocated burn and skeletal injuries. Bradshaw et al. examined 22 patients with collocated burns and fractures. Their cohort included 17 operative patients (11 with internal fixation, 2 with external fixation, and 4 with temporary external fixation converted to internal fixation after several days) and 5 nonoperative patients. The authors reported nine complications of which five were infections, highlighting the potential for greater infectious complications when stable wound closure cannot be rapidly achieved due to a collocated burn injury. Bui et al. described 40 patients with combined burns and fractures. Twenty-one patients had collocated burns and fractures. Patients with collocated injuries had greater injury severity and escharotomy, both of which were associated with greater rates of complications (e.g., wound infections).
Given the lack of clear guidance on the appropriate way to manage patients with combined burns and fractures, we recommend adherence to damage control orthopedic principles when appropriate ( Table 35.1 ). , Decisions around the timing and staging of surgical burn care and fracture management should be based on several considerations:
-
1.
Physiologic condition of the patient
-
2.
Stability of the fracture and need for its rapid reduction and immobilization (e.g., associated with pulseless extremity, open fractures, highly comminuted fractures)
-
3.
Access needed for burn wound evaluation and management
-
4.
Burn wound depth and need for excision and reconstruction in single or multiple stages
-
5.
Risk of functional recovery deficits with delays in care, immobilization, and weightbearing restrictions
Table 35.1
Indications and Damage Control Orthopedic Interventions for Musculoskeletal Injuries for Patients With Combined Burn and Trauma
| Location | Indication | Intervention |
|---|---|---|
| Spine | Unstable thoracic and lumbar spine fractures | Percutaneous dorsal instrumentation |
| Pelvis | Complex pelvic ring injuries with nerve or vascular injuries | External pelvic fixation |
| Open pelvic injuries | External pelvic fixation | |
| Stabilization of the pelvis for pelvic packing | C-clamp | |
| Posterior pelvic ring injuries | Percutaneous screw fixation | |
| Hemodynamic instability with unstable pelvic fracture | Pelvic packing, embolization, or ligation | |
| Extremities | Open fractures with soft tissue contamination | External fixation of long bones |
| Open fractures with large soft tissue defects | External fixation of long bones | |
| Large bone defects | External fixation of long bones | |
| Complex intraarticular fractures | External fixation of long bones | |
| Fractures with concomitant vascular injuries | External fixation of long bones | |
| Soft tissues | Morell-Lavallee lesion | Debridement and VAC therapy |
| Soft tissue contamination | Debridement and wound care | |
| Large soft tissue defects | Debridement and VAC therapy | |
| Compartment syndrome | Fasciotomy | |
| Mangled extremity with uncontrollable hemorrhage | Open amputation |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree