Overview of the Thoracic Cage
The thoracic cage serves two principal functions: to protect the thorax and upper abdomen and their associated viscera, and to allow movement for ventilation. It consists of 12 pairs of ribs and their respective costal cartilages; the manubrium and sternum anteriorly, and the thoracic vertebrae posteriorly. The costal cartilages of the true ribs (1–7) articulate directly to the manubrium/sternum. The costal cartilages of the false ribs (8–10) unite as a single synchondrosis and join the costal cartilage of the 7th rib. The floating ribs (11 and 12) have no anterior articulation. Three layers of intercostal muscles exist between each rib (external, internal, and innermost) and a neurovascular bundle (artery, vein, nerve) runs in a groove below each rib between the internal and innermost layers of muscle. The thorax is defined inferiorly by the diaphragm and superiorly by the clavicles.
Congenital Defects
Nagarajan Muthialu and Mazyar Kanani
Congenital chest wall anomalies may present at any age up to adolescence and may occur in isolation, or represent one marker in a spectrum of defects.
Pectus Deformities
In this spectrum of malformations, there is an abnormal contour of the anterior chest wall. It is thought the underlying problem lies with the relative rate of growth of the costal cartilages in relation to the growth of the ribs and sternum. If the elongated cartilages cause the anterior chest wall to buckle inwards, pectus excavatum results. If there is buckling outwards, the result is pectus carinatum. However, some have proposed that the defects arise from abnormal growth of the diaphragm.
Both conditions may be symmetric or asymmetric in nature, with a varying composite of anomalies, such as sternal angulation, tilt, and rotation about the long axis – all of which need to be taken into account during cosmetic correction. Within the broad spectrum, some patients may even have a mixed defect.
Pectus Excavatum
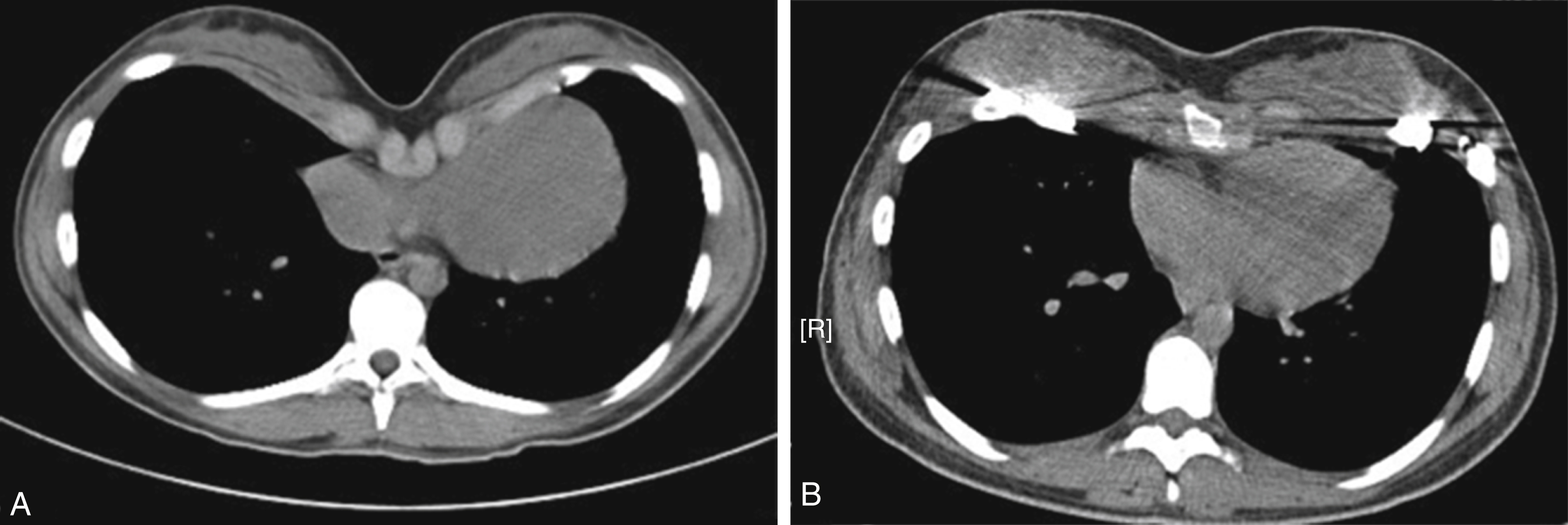
In this condition, there is a depression in the anterior of the chest wall of varying shape and severity ( Fig. 39.1 ).
Morphologic Varieties
In most cases, the depression is symmetrical on either side of the midline and limited to the lower half of the sternum. More severe forms are deeper and extend further upwards toward the manubrium. At the more severe end of the scale is the so-called “Grand Canyon” variety where there is a deep and long depression. It can be a challenge to treat successfully, particularly since many of these are asymmetrical. In about 5% of cases, there is a combined excavatum/carinatum deformity.
Epidemiology
The incidence of the defect is 1 in 150–1000 live births. Although the defect may be present anytime after birth, most patients present around the time of puberty during their phase of rapid body growth. There is also an association with connective tissue disorders such as Marfan’s and Ehlers–Danlos syndromes where scoliosis is also more frequent.
Clinical Evaluation
Prior to surgical correction, we undertake a number of investigations to determine the severity and extent of morphology, as well as impact on physiology.
Imaging
We routinely perform a plain chest radiograph that demonstrates the overall shape of the thoracic cavity. A plain chest radiograph is also a very useful screening tool to assess the spinal curvature initially.
This is followed by computerized tomography (CT scan) imaging, which can be used to construct the 3D anatomy of the thoracic cavity. The Haller index of severity can be calculated from this. This is the ratio of the transverse diameter and the anteroposterior diameter of the rib cage. A normal ratio is 2.5. Of special importance is the shape and axis of the body of the sternum. Sternal torsion and asymmetry have an impact on the type of surgery. CT scan can also show associated anomalies including spinal deformities and also the extent of cardiac compression.
We also routinely perform transthoracic echocardiography to assess overall ventricular function. There is an association with mitral valve prolapse, which may be seen with connective tissue disorders.
Functional Assessment
As part of our initial evaluation, we also perform exercise testing of the patient. This is following some reports of a modestly reduced functional capacity arising from the combination of right ventricular compression and reduced pulmonary capacity in some patients.
Lung function testing including spirometry becomes useful whenever a restrictive lung capacity is suspected. In combination with exercise testing, this offers a very useful physiological way to assess functional implication of the pectus deformities, and also serves as a prognostic index for postoperative recovery.
Surgery for Pectus Excavatum
Timing
There are advantages and disadvantages to both early and late repair. Each case is considered individually. Important factors to consider include the age at presentation, symptoms, severity of the deformity, and type of surgical approach. As a general rule, younger patients have a more malleable chest, which will make the minimally-invasive Nuss procedure an attractive option here. However, it has to be tempered with the fact that some of these children will outgrow their bar and need further bar insertion. Also, early bar removal in younger patients may result in recurrence as the chest continues to remodel with age. Nevertheless, the severity will reduce the age at intervention.
In the literature, surgery has been performed successfully from the ages of 1 year to 50. , In our experience, for the reasons noted above, the ideal is around the age of puberty.
Influence of Morphology
We reserve the minimally-invasive Nuss procedure for symmetric cases. If the symmetry extends to a so-called Grand Canyon-type deformity, we may either use two Nuss bars, or elect to perform an open sternochondroplasty. If it is likely that any second bar needs to be inserted high up, we favor an open approach due to the risk of placement over the great arteries in the mediastinum. In some cases with mild asymmetry, the Nuss bar can be molded appropriately.
Nuss Procedure
The patient is usually positioned supine on the operating table with the arms abducted following induction of anesthesia and epidural catheter placement.
Once positioned, necessary landmarks are drawn on the chest – the deepest part of the chest that indicates the level where the bar will lie, the point beneath the nipples at the level of where the bar will lie, and the two lateral points where the vertical skin incisions are made for the introduction of the bar and stabilizers.
The two lateral skin incisions are made, and the subcutaneous plane is developed to the marked point beneath the nipples at the level of where the bar will lie. These will be the points at which the bar will exit the chest wall on either side. Then, the thoracoscope is introduced into the right pleura through a port placed either just beneath or into the lateral skin incision. Carbon dioxide insufflation permits exposure. Next, the bar introducer creates a subcutaneous tunnel from the skin incision to the point of entry into the chest. Entry through the chest wall is observed via thoracoscopy. The introducer is passed to the pleural cavity on the other side, anterior to the pericardium under direct vision. It is guided through the chest wall. Once safely out of the skin, it is tied by umbilical tape to a molded Nuss bar of appropriate size. As the introducer is removed, the bar is carried with it into the chest. The bar is then finally flipped with a specially-designed flipper and secured to the chest wall by bilateral stabilizers secured to it by steel wire (see Fig. 39.3B ).
Following the procedure, the patient is extubated on table and a chest radiograph is taken immediately to check for pneumothoraces and bar position.
Once on the ward, the patient is observed in the high dependency unit for the duration of the epidural catheter – usually 4–5 days. Adequate analgesia is very important here once the epidural is removed. Mobilization aided by physiotherapy is the final step before discharge. A final posteroanterior and lateral chest radiograph check the bar position.
Bar Removal
We elect to remove the bar anywhere between 2 and 3 years. Although the bar can be left in for 4 years without problems, the longer it is left, the greater the degree of calcification around the bar, which can make removal more challenging. Removal is performed as a day-case procedure under general anesthesia. The greatest risk comes from pneumothorax, and there are also isolated reports of injury to great vessels.
Pectus Carinatum
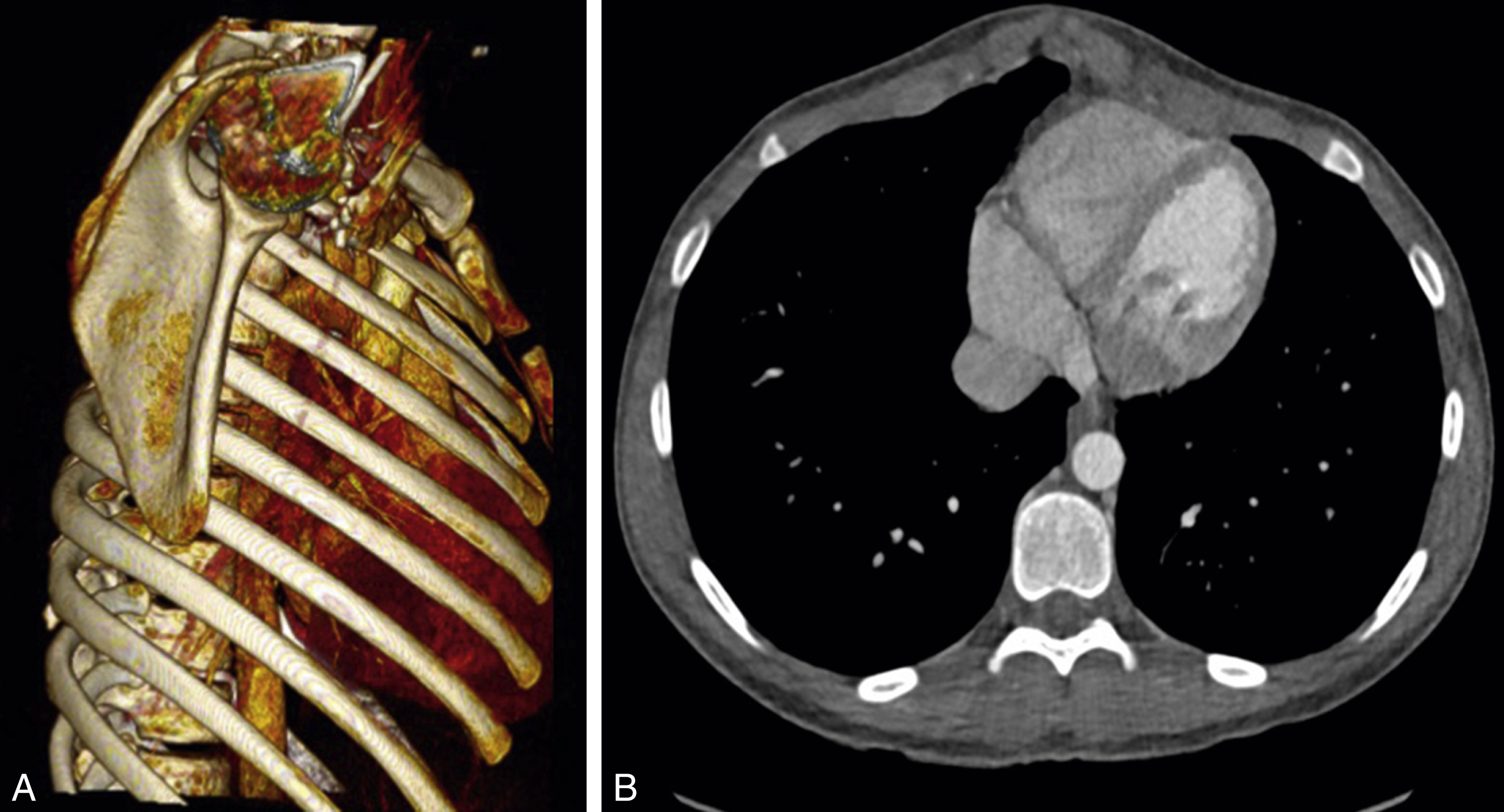
This is also known as “pigeon” or “keel” chest. In this condition, there is variable protrusion of the chest. It is less common than pectus excavatum, being seen in about 0.06 of all live births ( Fig. 39.2 ).
Morphologic Varieties
Depending on the exact extent of the protrusion, there are a number of variations.
- •
Symmetrical : The sternum is arched forwards. If there is some depression of the costal cartilages, the sternal prominence is exaggerated. In some cases, the body of the sternum fuses with the xiphoid at a 90-degree angle.
- •
Symmetrical deformity with a normal sternum : here, the anterior prominence comes from protrusion of the costal cartilages with a relatively normal sternal contour.
- •
Asymmetry with combined carinatum and excavatum : protrusion of costal cartilages on one side only causes rotation of the sternum about its long axis, causing a relative excavatum on the contralateral side.
- •
Pouter pigeon chest : there is prominence of a thickened manubrium so that the deformity is limited to the upper sternum only.
Surgery for Pectus Carinatum
Timing of Operations
This deformity becomes most apparent during adolescence when the infantile potbelly recedes to make the anterior chest more prominent during the period of rapid body growth. Although we do not have a distinct cut-off age, surgery is mainly reserved after this final adolescent growth-spurt has begun and chest wall natural chest wall modeling is complete.
Reconstructive Aims
The main aim of surgery is to remodel the anterior chest wall to the normal contour – through trimming the elongated costal cartilages and returning the sternum to its natural alignment. Surgery will provide an immediate result, but in order for the remodeling to be complete, we encourage physiotherapy and muscle-building exercises to enhance the muscular contribution to chest wall shape.
Technique of Repair
Our method of repair is based on that of Ravitch, but with the addition of any of the metal implants that we may have to consider, if needed for further stability. Chest wall stabilization using the Strasbourg Thorax Osteosyntheses System (STRATOS, MedXpertGmbH, Heitersheim, Germany) of costal bars ( Fig. 39.3A ) was considered very effective, till we recently reported on a high fracture rate of these bars.
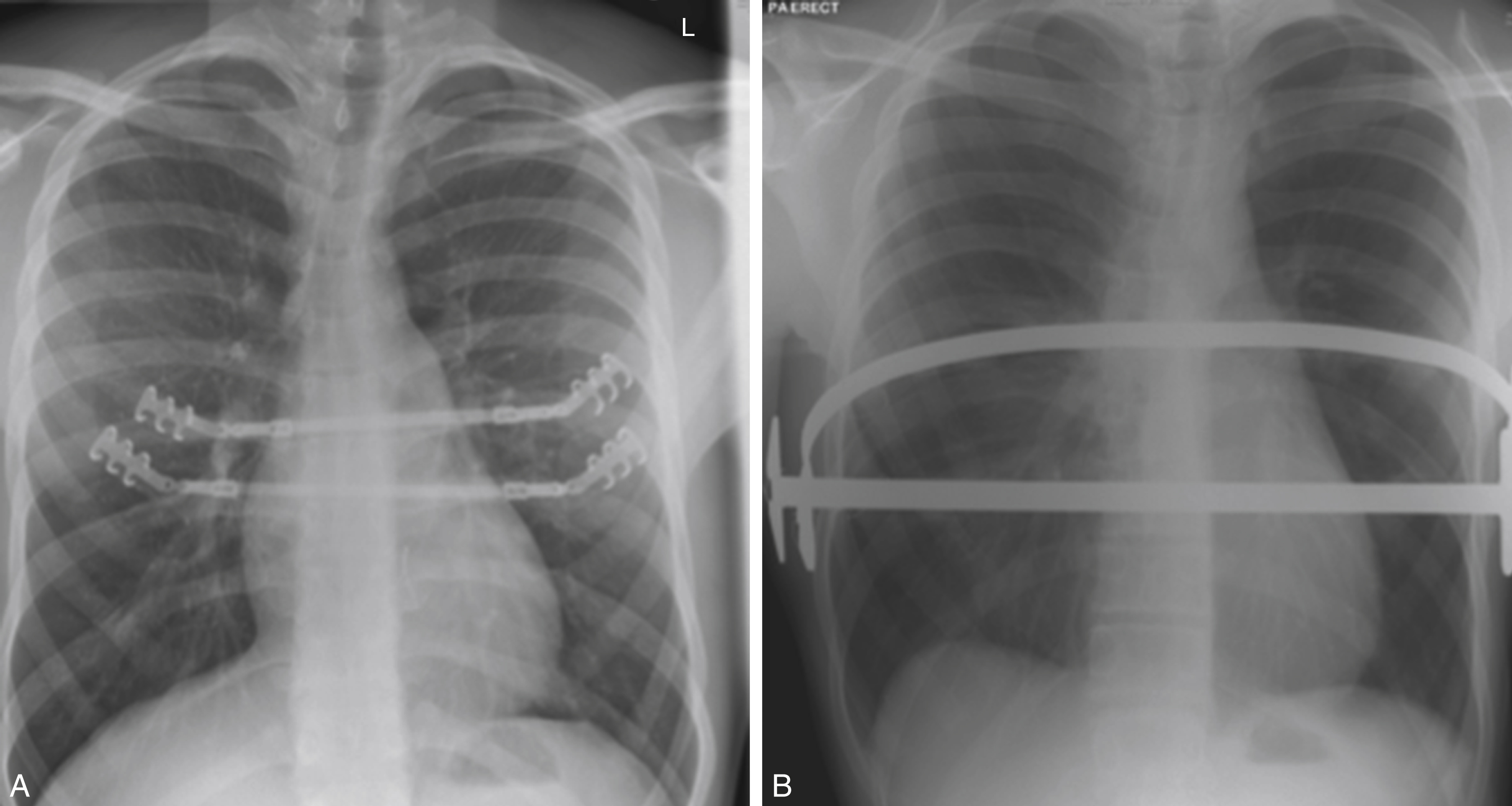
The patient is positioned supine, with the arms by the side. We prefer to mark the skin first – indicating both the line of incision, and the most prominent areas of cartilage/sternum. The incision is horizontal at the level of the xiphisternal junction, being some 10 cm with a gentle upwards curve. The subcutaneous plane is developed, at first, superiorly to the highest cartilage involved then inferiorly to the xiphisternum. This is seldom above the 3rd rib. Then the pectoralis major muscles are detached, sparing their clavicular origins. Care must be taken not to enter the intercostal spaces, especially in thin patients. Next, the plane inferior to the incision is developed. The uppermost origins of the rectus abdominis muscles may have to be detached if the underlying cartilages are involved. At this point, the xiphisternum is also detached from the body of the sternum with diathermy.
Once the underlying involved costal cartilages and ribs are exposed, resection can begin. The cartilages are initially detached at the sternocostal joints through a combination of diathermy and sharp dissection. Care must be taken not to damage the underlying internal thoracic vessels, or parietal pleura. In order to preserve growth potential in the juvenile sternum, we try to preserve the underlying perichondrium when shelling out the exuberant cartilage. Generally, the lower the cartilage level, the greater the lateral extent of resection. The lateral extent of resection will be determined by the final position of the sternum.
A sternal osteotomy is performed at a level determined by the angle and position of protrusion. This allows the sternum not only to be straightened, but also rotated appropriately to the normal anatomical plane. The sternum is fixed in the correct position using absorbable suture, or stainless steel wires depending on the size of the patient.
Next, the free ends of the ribs are fixed to the lateral edge of the sternum with a figure-of-eight absorbable sutures, making sure that they lie flat without protrusion. The xiphisternum is also reattached to the body of the sternum with absorbable sutures.
If there has been multilevel involvement with the risk of chest wall instability, we add STRATOS titanium rib bridges to provide final stability.
The muscles are reapproximated, ensuring that the two pectoralis major muscles on either side are brought together in the midline. This will help determine the final contour of the chest, and allow better healing of the bone beneath. Two drains are inserted and placed in the subcutaneous plane. If the pleural cavity has been entered, a suction catheter is placed at the end of the case to evacuate air. The rest of the tissues are closed in layers and dressing applied. Once the patient is in the recovery room, a chest radiograph is taken to ensure there is no pneumo- or hemothorax.
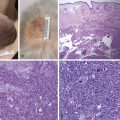
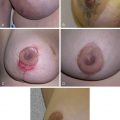
Poland Syndrome
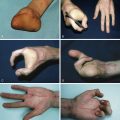
Poland syndrome is a rare congenital anomaly characterized by hypoplasia of the breast and nipple, absence of the costosternal portion of the pectoralis major muscle, absent pectoralis minor, aplasia or deformity of the costal cartilages and ribs 2–4 or 3–5, alopecia of the axillary and mammary region, and unilateral brachysyndactyly.
Epidemiology and Etiology
The incidence of Poland syndrome ranges from 1 in 7000 to 1 in 100,000 live births. Males are affected more frequently, with a 2:1 to 3:1 ratio. The right side of the body is involved in 60%–75% of patients.
There are a number of theories as to why it develops. One suggests that there is hypoplasia and interruption of the subclavian artery during the 6th week of development. Another theory suggests a disruption of the lateral plate mesoderm affecting the development of the upper limb girdle. There is an association with Klippel–Feil syndrome and Möbius syndrome, with bilateral congenital facial nerve palsy and paralysis of the eye abductors.
Clinical Manifestations
Although there is some variation upon the theme, the most consistent feature is the absence of the sternocostal head of the pectoralis major muscle. The underlying pectoralis minor may be absent altogether. Other chest wall muscles, such as the latissimus dorsi and serratus anterior, may also be absent – which is important when considering surgical soft tissue coverage. In up to a third of females, there is absence of breast development.
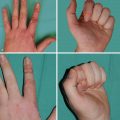
Absence of underlying ribs and costal cartilages causes the ipsilateral chest to be sunken. The most frequently absent or hypoplastic ribs are 2–4, or 3–5. As a result, upwards of 10% of patients may also have lung herniation and chest wall instability leading to paradoxical movements during respiration. Hand abnormalities in Poland syndrome include shortness of the middle phalanges with cutaneous webbing (or even full syndactyly), to complete absence of the hand (ectrodactyly). These tend to be ipsilateral to the chest wall anomalies.
Reconstructive Aims and Indications for Surgery
Surgical intervention may be indicated for the following reasons:
- •
Unilateral depression of the chest wall with cosmetic implications
- •
Lack of adequate protection of the heart and lung with lung herniation
- •
Chest wall instability with resulting paradoxical chest movement
- •
Hypoplasia or aplasia of the female breast.
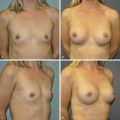
Surgical Management
This will be influenced by the age and sex of the patient, as well as pattern of involvement. Both the chest wall contiguity and muscular coverage need to be addressed. Many have advocated a two-stage approach in children – first repairing the chest wall defect, followed by a second procedure, which provides chest wall soft tissue coverage and sculpting of the absent anterior axillary fold. A single-stage approach may be reserved for older children and adolescents.
The approach is via a transverse incision – which will provide excellent access not only to the rib defects, but also to the sternum, which may be rotated as part of the constellation of defects. A subperichondral resection of the costal cartilages is performed of the affected rib ends. A sternal osteotomy is performed to allow it to be rotated to the corrected plane. Where there is rib aplasia, rib grafts may be used to bridge the defect. Synthetic mesh has also been used to provide a wider coverage. We have, however, in more recent cases been using artificial bone or STRATOS bars to bridge the defect and provide chest wall stability.
A musculocutaneous flap of the latissimus dorsi can be used for coverage and provide reconstruction of the absent axillary fold. In our center, such cases are performed as a joint procedure between the thoracic and plastic surgeons.
Sternal Cleft
This is a rare malformation characterized by failure of fusion of the sternum in the midline. It may be partial, where there is a U-shaped dip at the upper end of the manubrium, to a more extensive defect extending all the way to the xiphisternum, where there are two bars of bone to which attach the costal cartilages.
This defect in the sternum may occur in isolation, but it may also herald associated anomalies, such as cardiac malformations.
Surgical Approach and Timing
Our preference is to repair this defect in the neonatal period or at least as early as possible, as the more pliable tissues allow for a better chance for primary repair with autologous tissues.
The repair is traditionally carried out via a midline vertical incision and the subcutaneous plane developed over the sternum and adjoining costal cartilages. A wedge osteotomy is performed of the bony bridge between the two sternal bars. This will allow better mobilization when bringing the two sides together without tension.
If the two sides of bone can be brought together, they may be sutured together with either stainless steel wire, or else a heavy absorbable suture. In neonates or early infancy we use heavy absorbable suture. We have recently developed a primary repair with a periosteal flap and sliding osteochondroplasty, thereby avoiding the use of metal implants.
If there is tension laterally, the sternocostal joints may have to be released and resutured or bridged with STRATOS bars.
We close the soft tissues over the bone by suturing the pectoralis major, rectus abdominis, and sternocleidomastoids to their counterparts in the midline. The subcutaneous tissues and skin are then closed primarily to complete the operation.
Jeune Syndrome: Asphyxiating Thoracic Dystrophy
This autosomal recessive condition leads to characteristic skeletal abnormalities in the form of generalized chondrodysplasia, limb defects, mild dwarfism, and importantly, a constricted chest. This latter feature makes the resulting physiological problems particularly difficult to manage.
Epidemiology and Clinical Features
The incidence is 1:100,000 to 1:130,000 live births. Since 2006, this has been classified under the umbrella of short rib skeletal dysplasias with or without polydactyly. The natural history of the condition is that the majority of patients do not live beyond the age of 10, mainly from the complications of respiratory or renal failure, which are some of the extra-skeletal manifestations of the disease.
Chest Wall Morphology
Failure of normal costal cartilaginous growth leads to cessation of normal chest wall expansion and a persistently narrowed, bell-shaped rib cage. The result of the defect is that the ribs and costal cartilages are shortened, with a fixed horizontal orientation. This results in failure of normal lung development with resulting respiratory failure and pulmonary hypertension, recurrent chest infections and restrictive lung disease.
Chest Wall Surgery
There is no easy surgical solution to the chest wall defects seen in this condition. A number of approaches have been attempted, often through staged interventions.
Lateral thoracic expansion was first outlined in 1995 by Davis et al as a staged approach for patients who are older than 12 months. Dissection is carried down to the beds of the 4th to 9th ribs. Those ribs are divided along their shaft, and their adjoining intercostal muscle released. This will permit the rib to be connected to the rib below by a titanium plate in order to expand the chest. Additional chest wall support is obtained by uniting the supperiosteal tissue of rib bed to its counterpart below. In those where this technique has been used, new bone formation has been demonstrated.
While staged approach seems to contribute to better perioperative management in the past, we demonstrated that single stage bilateral repair offers overall improvement in respiratory dynamics in such a way that the ventilatory as well hemodynamic managements become simpler when bilateral surgery is performed at the same time. The operative time is shortened overall, as well as the length of intensive care time, thereby potentially limiting the risk of overall morbidity and burden of care.
Less frequently, there are reports of the modified Nuss technique being used to provide expansion. In the former technique, two bars are implanted bilaterally to provide expansion in the lateral plate. It has been reserved for older children and is limited by the need for a bar change.
More recently, thoracoplasty using the vertical expandable titanium rib technique, has been described. A vertical titanium device is anchored bilaterally to ribs 2 and 10. Ribs 3–8 are then transected and pulled laterally, and finally connected to the titanium device. Growth is allowed by progressively adjusting the device.
Acquired Defects
Oliver J. Smith, Robert Pearl, and Simon Withey
In adult patients the acquisition of chest wall defects almost always occurs due to one of four etiologies: midline sternal wound dehiscence following cardiothoracic surgery; trauma; neoplasms; tissue damage secondary to radiotherapy. Each of these pathologies presents different reconstructive challenges in relation to patient factors such as overall health and adjuvant therapy, and local factors such as tissue quality, infection, and excision margins.
Sternal Wound Dehiscence
The midline sternotomy incision was introduced into cardiac surgery in the 1950s. Early management of sternal wound dehiscence involved debridement and packing of the wound, leaving it to heal by secondary intention. This led to high mortality rates with surviving patients often requiring prolonged hospitalization. In the 1970s the technique of early debridement and introduction of fresh, well-vascularized omental flaps into the wound defect was pioneered by Lee et al. The use of muscle flaps to reconstruct the sternal dehiscence was developed from this, and has become the gold standard treatment leading to a significant reduction in mortality rates.
Etiology
Wound dehiscence is reported in 0.5%–5% of midline sternotomies and is a serious complication which can lead to potentially fatal mediastinitis. The primary underlying causative factor in dehiscence in most patients is partial or complete devascularization of the hemisternum after harvest of the internal mammary artery (IMA) for cardiac bypass. When both IMAs are harvested the risk of wound complications increases dramatically. Risk is increased in patients who are already suffering with underlying vascular disease and are therefore less able to perfuse the sternum through collateral circulation, such as those with poorly controlled diabetes or heavy smokers. Other generalized factors such as obesity, cardiac or respiratory failure, or immunosuppression also increase the risk, as well as local factors such as previous chest wall irradiation.
Infection is often not the causative factor of wound dehiscence and is usually a secondary complication. A wide range of Gram-positive and negative organisms can be detected from wound cultures, with the commonest being Staphylococcus aureus and Pseudomonas aeruginosa. However, organisms cultured may not be pathologically active and may be commensal to the wound surface. Antibiotic resistance in organisms that are active in causing infection is becoming a serious problem and treatment will be based on local guidelines.
Pairolero and Arnold proposed a classification system of sternal wound infections as outlined in Table 39.1 . However, this is limited by the fact that it fails to categorize the degree of vascularity in these wounds and is focused mainly on the secondary complication of infection. Starzynski categorizes sternal defects based on the degree of anatomical involvement following operative debridement. This can help to predict the degree of physiological compromise, however as all patients react differently, its use clinically can be limited.