CHAPTER 9 Relationship Between Fat Tissue and Lymphangiogenesis
KEY POINTS
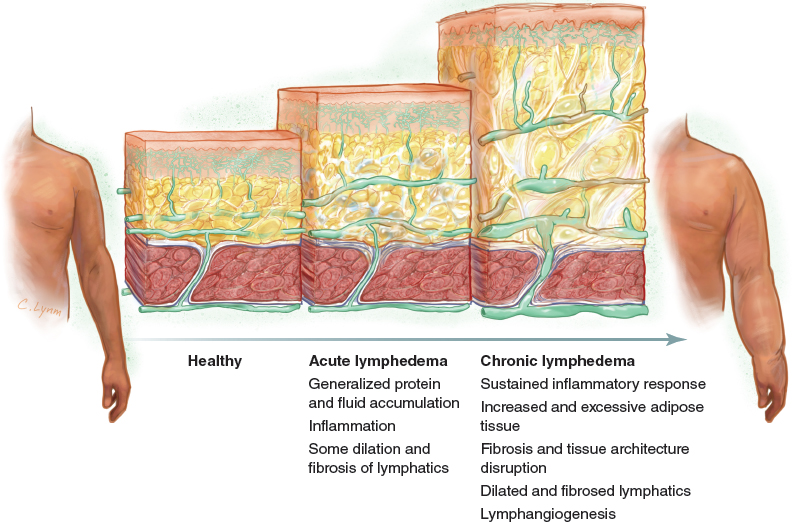
Adipose tissue overgrowth is a key clinical feature in chronic lymphedema.
Lymphangiogenesis may play a critical role in lymphedema-related adipogenesis.
It is still unclear how the intricate mechanisms of lymphangiogenesis, inflammation, and adipogenesis are interrelated.
Fat deposition has long been recognized as one of the clinical features of elephantiasis. In a very elegant clinical description, Moritz Kaposi, 1 in the nineteenth century, stated: “If an incision is made on a limb with advanced elephantiasis… the subcutaneous tissue is increased many fold…” In the twentieth century, debulking procedures to remove excess skin and fat became popular as the preferred surgical treatment for patients with lymphedema.
Until recently, much of our knowledge about the pathophysiology of lymph stasis focused on protein retention, altered immune cell trafficking, and inflammation. Little attention was directed to fat deposition, notwithstanding the fact that some authors considered it a subproduct of the inflammatory response. 2
In the late 1990s, a great discussion ensued after Brorson and Svensson’s publication of a series of patients treated with liposuction. 3 Soon after, advances in the comprehension of molecular mechanisms related to lymphangiogenesis and adipogenesis provided new insights into the relationship between lymph stasis and fat.
This chapter discusses how these findings have influenced our current knowledge of the relationship between fat tissue and lymphangiogenesis and how this understanding can lead to future therapeutic approaches in patients with lymphedema.
Adipogenesis
For a long time, the known functions of subcutaneous fat were limited to energy storage and regulation of body temperature. Perhaps because of its relatively simple structure, adipose tissue was a victim of several decades of scientific neglect. It was not until the more recent recognition of the burden of obesity in modern society that further investigation into the nature of adipose tissue unveiled a whole complex and dynamic physiologic organ.
Adipogenesis is the process of differentiation that turns preadipocytes or multipotent stem cells into mature adipocytes. This is regulated by several mechanisms (transcription factors, cofactors, and signaling) from as early as the embryonic period until adult life. The number of adipocytes does not vary after childhood and adolescence, even after weight changes. 4 Thus most weight gain or loss depends on the size variation of the intracellular storage of triglycerides and cholesteryl esters in fat droplets.
Because the total population of fat cells remains constant in adulthood, new adipocytes can be generated by two different pathways: preexisting undifferentiated progenitor cells or through dedifferentiation of mature adipocytes into preadipocytes, which can proliferate and differentiate into mature adipocytes. 5 The annual turnover rate of fat cells is estimated to be 10%. 4
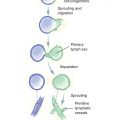
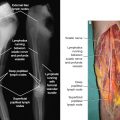
The differentiation from a stem cell precursor to a mature adipocyte involves a complex sequence of gene expression and signaling and is subject to the environment in which it occurs, either in culture or in vivo. Multipotent stem cells are capable of differentiating into mesodermal cell types (adipocytes, chondrocytes, osteoblasts, and myocytes). Under stimulation by hormones, cytokines, and growth factors (insulin, insulin-like growth factor 1, glucocorticoids, mineralocorticoids, and thyroid hormone), some multipotent stem cells become committed to the adipogenic lineage (preadipocytes). Preadipocytes then differentiate into adipocytes by the action of transcription factors that activate specific genes responsible for the adipocyte phenotype. The most important transcription factors at this phase are CCAAT/enhancer-binding protein alpha (C/EBP-alpha) and peroxisome proliferator-activated receptor gamma (PPAR-gamma). 6 Fig. 9-1 shows the normal relationship between the lymphatic system and fat in a limb.
Lymphangiogenesis
Although lymphangiogenesis is discussed in greater detail elsewhere in Chapter 7, some brief aspects of lymphatic system development will illustrate the relationship between the lymphatics and fat.
The lymphatic system appears during the sixth and seventh weeks of embryonic development, 7 4 weeks after the primary components of the blood circulation arise. Two different theories about the origin of the lymphatics were proposed at the beginning of the twentieth century. Sabin’s anatomic studies 8 suggested that sprouting from blood endothelial cells was the origin of the lymphatic system. This was in opposition to Huntington’s theory of the centripetal formation of the lymphatics from mesenchymal lymphangioblasts, which reached the venous system later in development. 9 Some controversies still remain, and evidence seems to support both theories. 10 , 11
The regulation and development of the lymphatic system depend on several signaling factors and cell receptors. The most important signaling factors are glycoproteins known as vascular endothelial growth factors (VEGFs). VEGFs are the primary regulators of endothelial proliferation, angiogenesis, vasculogenesis, and vascular permeability. 12 There are six VEGF subtypes (A, B, C, D, and E) and placental growth factor (PlGF) that bind to specific membrane tyrosine kinase receptors.
Three different VEGF tyrosine kinase receptors have been identified so far:
Vascular endothelial growth factor receptor 1 (VEGFR-1) (Flt-1)
VEGFR-2 (Flk-1 and KDR)
VEGFR-3 (Flt-4)
VEGF-B and PlGF bind to VEGFR-1, whereas VEGF-A interacts with both VEGFR-1 and VEGFR-2. VEGF-E binds to VEGFR-2, and both VEGF-C and VEGF-D bind to VEGFR-3. VEGFR-1 and VEGFR-2 mediate angiogenesis, whereas VEGFR-3 is involved mainly in lymphangiogenesis.
VEGFs and VEGFRs are essential for blood vessel development and angiogenesis. Unlike the other VEGFs, both VEGF-C and VEGF-D promote lymphangiogenesis through their specific lymphatic endothelial receptor VEGFR-3. Nevertheless, early experimental inactivation of Flt4, the gene responsible for encoding VEGFR-3, also results in defective blood vasculogenesis and angiogenesis, 13 demonstrating that VEGF-C not only has a critical role in the development of the lymphatic system but is also essential in the development of the embryonic cardiovascular system.
Lymphangiogenesis is a developmental process that occurs mainly in the embryonic phase and is uncommon in adult tissue. In adults new lymphatic tissue is formed in wound healing and is induced by some tumors. Thus the exact role of VEGF-C and VEGF-D in normal human physiology is unclear. Some VEGF actions have been described: stimulation of growth of lymphatic capillaries and recruitment of inflammatory cells, 14 regulation of endothelial fatty acid, and control of salt-dependent interstitial volume and blood pressure. 15
Interleukin-7 (IL-7) and interleukin-1 beta are cytokines involved in lymphoid tissue regulation and inflammation and can also participate in lymphangiogenesis. IL-7 increases the expression of lymphatic markers (lymphatic vessel endothelial hyaluronan receptor 1 [LYVE-1], podoplanin, and Prox1) in endothelial cells, and it induces the formation of lymphatic vessels in vivo. Interleukin-1 beta upregulates VEGF-C, and IL-7 upregulates VEGFR-3 and the expression of VEGF-D in endothelial cells. 16
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree