CHAPTER 7 Lymphangiogenesis
KEY POINTS
Lymphangiogenesis is the formation of new lymphatic vessels.
Lymphangiogenesis is regulated by both prolymphangiogenic and antilymphangiogenic mechanisms.
B cells and macrophages are key sources of prolymphangiogenic growth factors, such as vascular endothelial growth factors.
T cells are a key source of antilymphangiogenic cytokines, such as interferon gamma.
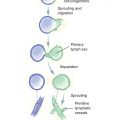
Lymphangiogenesis is the formation of new lymphatic vessels. Specifically, lymphangiogenesis refers to the sprouting of lymphatic vessels from preexisting vessels with migration and differentiation of lymphatic endothelial cells (LECs). In contrast, lymphovasculogenesis refers to the de novo formation of lymphatic vessels from mesenchymal-derived sources. These processes are not mutually exclusive and contribute to varying degrees to lymphatic vessel development and proliferation. This process is regulated by a complex interaction between prolymphangiogenic and antilymphangiogenic forces, the balance of which modulates a wide variety of effects in LECs, including migration, differentiation, proliferation, and survival.
Prolymphangiogenic Mechanisms
The discovery of lymphatic-specific markers such as Prox1, LYVE1, and podoplanin led to a significant increase in our understanding of the mechanisms that regulate lymphatic development and regeneration. These studies have identified several growth factors and cytokines that either directly or indirectly regulate LEC differentiation, proliferation, migration, and function. These molecules are expressed in various tissues and have overlapping functions. In general, the expression of lymphangiogenic factors leads to lymphatic vessel growth and regeneration, thereby increasing lymphatic function, although there are some tissue and temporal differences in the mechanisms of action. Although initial studies in this field focused on vascular endothelial growth factors, more recent studies have identified several crucial lymphatic regulators, which suggests that lymphatic homeostasis is a complex process in vivo.
VASCULAR ENDOTHELIAL GROWTH FACTORS
A crucial factor for LEC development is vascular endothelial growth factor C (VEGF-C). VEGF-C interacts with vascular endothelial growth factor receptor 2 (VEGFR-2) and vascular endothelial growth factor receptor 3 (VEGFR-3) and regulates a wide range of effects in LECs, including differentiation, survival, and migration. Vascular endothelial growth factor D (VEGF-D) is a closely related homolog that also activates VEGFR-2/VEGFR-3. VEGFR-3 expression is primarily limited to LECs in postnatal mice and is used as a specific marker to identify lymphatic vasculature. 1 VEGF-C, and to a lesser extent VEGF-D, are highly expressed in the regions of the body in which the lymphatics develop in abundance. 2 Gradients of VEGF-C regulate the migration of LECs during lymphatic repair after wound healing, and VEGF-C overexpression in the skin of transgenic mice results in lymphatic proliferation and vessel enlargement. 3 Similarly, gene therapeutic or recombinant protein delivery of VEGF-C results in lymphangiogenesis caused by increased budding, migration, proliferation, and differentiation of LECs. 4
The pioneering work of Karkkainen et al 2 showed that VEGF-C is essential for lymphatic development, because VEGF-C–deficient mice do not develop lymphatic vasculature and die in utero. Mice heterozygous for VEGF-C have severely hypoplastic lymphatics with significant lymphatic defects, including lymphedema. Although VEGF-C and VEGF-D appear to have overlapping effects, the independent roles of these growth factors are not completely understood, because congenital VEGF-D deficiency is not lethal. However, exogenous delivery of VEGF-D can rescue VEGF-C knockout mice and restore lymphatic differentiation and development. The key roles for VEGF-C in the regulation of lymphatic development and function are also reflected in VEGFR-3–deficient mice and humans. Although homozygous deficiency of VEGFR-3 results in intrauterine death, mice with a heterozygous inactivating mutation of VEGFR-3 develop chylous ascites and primary lymphedema with severely hypoplastic cutaneous lymphatic vessels. 4 , 5 Heterozygous inactivating mutations of VEGFR-3 have also been clinically identified and are the cause of Milroy disease, an autosomal dominant form of primary lymphedema.
The exogenous administration of VEGF-C improves lymphatic function in many models of lymphatic insufficiency and dysfunction. For instance, local delivery of VEGF-C in various animal models of primary or secondary lymphedema, including the rabbit ear, rat hindlimb, and mouse tail models, significantly decreases edema, improves lymphangiogenesis, and decreases the pathologic changes associated with lymphedema. 6 – 8 Similarly, the local delivery of VEGF-C in sheep, pig, and rat lymphadenectomy models enhances lymphatic transport and decreases lymphedema. 9 Adenoviral VEGF-C delivery induced growth, remodeling, differentiation, and maturation of lymphatic capillaries after lymph node dissection in mouse and pig models. 10 – 13 However, although these results are exciting, the clinical translation of these findings to patients with iatrogenic lymphedema resulting from cancer treatment has been hampered because VEGF-C is a major regulator of tumor growth and metastasis. 14 – 16 In addition, in some preclinical models, the exogenous administration of even massive doses of VEGF-C has not led to an improvement in lymphedema, which suggests that additional mechanisms are involved. 17 Therefore understanding these other pathways (see Antilymphangiogenic Mechanisms in this chapter) is critical if lymphangiogenesis and the restoration of lymphatic flow are the ultimate goals.
VEGF-A (often simply referred to as VEGF) is a well-known regulator of angiogenesis and activates VEGFR-2. However, recent work indicates that VEGF-A, which is also a regulator of lymphangiogenesis, plays a central role in this process in some circumstances. For example, inflammatory lymphangiogenesis in the lymph nodes is considered primarily regulated by increased B-cell expression of VEGF-A. These effects are the result of activation of VEGFR-2 on LECs, which subsequently activate a diverse set of intracellular pathways with significant overlap with VEGF-C/VEGFR-3. Similarly, gene therapeutic approaches with VEGF-A promote lymphatic vessel enlargement and proliferation. 18 , 19 However, in contrast to VEGF-C, VEGF-A administration is also associated with the proliferation of immature, leaky blood vessels.
Macrophages are a key cellular source of VEGF-C and VEGF-A during wound repair. Indeed, these cells play a key role in the regulation of lymphangiogenesis in several physiologic settings, including inflammation, tumor-associated lymphangiogenesis, and during wound repair. Bone marrow–derived monocytes are recruited by various inflammatory stimuli and differentiate into macrophages that then secrete inflammatory cytokines and chemokines. In addition, macrophages produce large amounts of VEGF-C, thereby promoting LEC migration and differentiation. A few studies have also suggested that macrophages can transdifferentiate into LECs, thereby directly contributing to and incorporating into newly formed lymphatic vessels. Abnormalities in macrophage function and VEGF-C production have been linked to impaired lymphangiogenesis in diabetic mouse models, suggesting that this pathway plays an important physiologic role. Other sources of VEGF-C include B cells and mast cells. However, the relative contribution of these cell types to lymphangiogenesis during wound repair remains less well understood. 20 , 21
FIBROBLAST GROWTH FACTORS
Fibroblast growth factors (FGFs) are a family of growth factors that are involved in lymphangiogenesis, angiogenesis, endocrine signaling pathways, and wound healing. 22 Although there are many isoforms of FGFs, the lymphangiogenic roles of FGF-1 and FGF-2 have been the most widely studied. These growth factors regulate lymphangiogenesis by direct (for example, direct effects on LECs) and indirect (by activation of VEGFs) mechanisms. However, the preponderance of evidence suggests that indirect mechanisms are more important in vivo. Several lines of evidence propose that these indirect mechanisms are related to increased expression of VEGF-C, VEGF-C independent activation of VEGFR-3, and upregulation of VEGFR-3 expression. 23 Thus, although the exact mechanisms by which FGFs regulate lymphangiogenesis remain unknown, it is likely that these growth factors have key roles in vivo and that the effects of these pathways are dose, site, and situation dependent. 24
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree