Chapter 8 DETERMINATION OF FAT VIABILITY
One of the valid concerns about structural fat grafting can be the potentially high rate of poor graft take of the grafted tissue over time in the graft site, which may reach at least 50% of the filled volume. 1 , 2 The most acceptable explanation of poor fat graft take has been based on Peer’s cell survival theory that the number of viable adipocytes at the time of transplantation may correlate with the ultimate volume of surviving grafted fat. 3
To obtain long-term survival of transplanted autologous fatty tissue, the harvested and processed fatty tissue parcels must remain viable before implantation. Although several studies were performed in the past searching for improvement of fat transfer, 4 – 6 the viability of fat grafts harvested and processed by a number of techniques remains essentially less well known. Recently, several experimental laboratory tests including immunohistological staining have been used to determine the viability of fat grafts; the search for the best test to evaluate the viability of fat grafts continues.
In this chapter we will review several laboratory tests used to study the viability of fat grafts and provide some scientific explanation for each test used in the laboratory. Therefore surgeons who perform structural fat grafting will be able to familiarize themselves with these techniques and may be able to use one of these techniques to guide their clinical practice in fat grafting.
Laboratory Tests
Laboratory tests that have been used to determine the viability of fat grafts include viable adipocyte counts, colorimetric assays, adipocyte-specific enzyme assays, routine histologic examination, and immunohistochemical staining. Each test must be processed in vitro and may be time consuming; each has been selected by investigators as a single test or in combination to determine the viability of fat grafts experimentally.
VIABLE ADIPOCYTE COUNTS
Viable adipocyte counts through tryptan blue staining is a well-established test for detecting viable cells within a specific tissue. The test requires enzyme digestion to release adipocytes from a parcel of grafted fat. However, enzyme digestion can damage many adipocytes, which may lead to the underestimation of the viable adipocyte number. Each viable adipocyte with an intact membrane should be stained blue under microscopy. All blue-stained cells are counted as viable adipocytes. Clearly, more blue-stained adipocytes in a given volume should represent better viability of fat grafts.
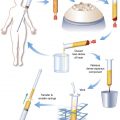
In general, 1 g of a fat graft specimen is collected. Each specimen is washed with phosphate-buffered solution (PBS) three times. It is then mixed with 1 mg/cc of type I collagenase (Sigma, St. Louis, MO) in PBS containing 5% bovine serum albumin (Sigma) for digestion and is incubated at 37° C in a CO2 incubator. After 1-hour incubation, the digestion is terminated with 10% (v/v) fetal calf serum (Sigma), and any remaining tissue fragments are removed by straining the digested fatty tissue through a piece of large-weave gauze. The digested fat grafts are fractionated into mature adipocytes (top layer) and stromal pellet (bottom) after centrifugation at 200 g for 10 minutes. The viable adipocytes are determined after 0.4% trypan blue vital stain (Sigma) from a 100-microliter sample with 1:1 dilution with trypan blue. The number of viable adipocytes is then counted with a hemocytometer under a microscope with 400× magnification. 7 , 8
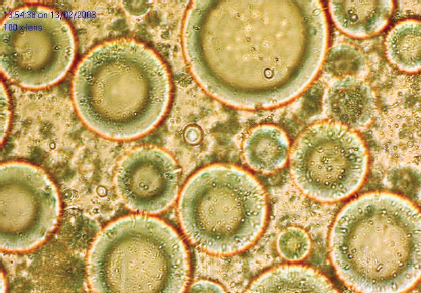
The normal appearance of intact viable adipocytes is shown. This cell counting technique can be difficult in adipocytes because of the scant cytoplasm in mature adipocytes. 9 In addition, since a cell isolation procedure is used in the test, it may not accurately reflect the viability of fat grafts in the intact tissue, because isolation of adipocytes eliminates the stromal component surrounding the adipocytes. 10 Nevertheless, this test can provide meaningful information about the viability of fat grafts and can serve as a baseline test in determining the viability of fat grafts.
COLORIMETRIC ASSAY
A colorimetric assay uses tetrazolium salt, 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenyl-amino-carbonyl]-2H-tetrazolium hydroxide (XTT), which can be reduced by active mitochondrial dehydrogenase in viable cells, yielding a highly colored formazan product. Even newly dead cells will not cleave XTT, so the assay is sensitive to the presence of living cells. Therefore a quantitative in vitro colorimetric assay of cell proliferation has been used to determine the viability of fat grafts. 4 , 6 , 9
Rohrich et al 4 described the colorimetric assay as follows:
The colorimetric assay using XTT [2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenyl-amino)-carbonyl]-2H-tetrazolium hydroxide; Roche Molecular Biochemicals, Branchburg, NJ] is designed for spectrophotometric quantification of cell growth and viability. The tetrazolium salt XTT is metabolized into a water-soluble formalin dye through mitochondrial dehydrogenase activity and is measured at 450 to 500 nm. The colorimetric assay system using XTT (Roche) is designed for spectrophotometric quantification of cell growth and viability. The Cell Proliferation Kit II XTT (Roche Diagnostics No. 1465015) can be used to assay the viability of adipocytes through the following protocol. First, the XTT labeling reagent and the electron-coupling reagent (from the kit) are prepared in a 37° C waterbath. Culture medium (Dulbecco’s modified eagle medium, pH 7.4) is transferred (1.0 ml) into each well (24-well plate). One well is designated as control (media + XTT + no cells). Duplicates of each sample are performed. A heat-treated fat sample (60° C, 10 minutes) serves as a negative control. Specimen aliquots (0.2 ml) are placed into each well, except for control wells. XTT labeling reagent (5.0 ml) and electron-coupling reagent (0.1 ml) are mixed, and 1.5 ml of the mixed XTT reagents is added to each well. Absorbance readings are performed at 0, 1, 2, and 3 hours at 492 nm and 690 nm. At each time point, aliquots (0.75 ml) are transferred from each well into a 96-well plate for readings (Spectramax Plus 384; Molecular Devices, Sunnyvale, CA). Between readings, the 24-well plate is incubated at 37° C.
The XTT assay provides a good correlation between the number of viable cells within fat grafts and the resulting formazan absorbance values. 4 , 6 , 9 , 11 Because the assay does not rely on a cell isolation procedure, it may accurately reflect viability of fat grafts with the intact stromal component surrounding the fat cells. However, the assay is not specific for adipocytes. Any viable cells within fat grafts—blood-derived cells, endothelial cells, adipose-derived stem cells, and fibroblasts—can influence the total absorbance values. 9
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree