Chapter 30 STRUCTURAL FAT GRAFTING IN CRANIOFACIAL SURGERY
The new era in craniofacial surgery is defined by two key ideas: tridimensional restoration and regeneration. These are new concepts and new ways of thinking about the reconstruction of craniofacial defects. From that perspective, adipose tissue can serve as a multipotent tool. The current concept of structural fat grafting is to volumize the tissue while providing a source of adipose-derived stem cells (ADSCs) to optimize the benefits of these cells.
The concept of replacing volume with fat grafts was for the most part discarded in the 1970s, but some surgeons, such as Sydney Coleman, were persistently using it, mainly for cosmetic purposes. During the first decade of the twenty-first century, the revolutionary use of fat in reconstructive breast surgery by Gino Rigotti opened new avenues for research into the regenerative purposes of ADSCs.
Embryology
The extremely interesting embryology of the craniofacial tissues emphasizes the possibility of different strategies for regenerative research involving this area. Interaction between the neural crest and pharyngeal arches, composed of ectoderm, mesoderm, and endoderm, may be responsible for the uniqueness of this specific region. It is quite significant that only cranial neural crest cells are able to form cartilage and bones, compared with neural crest cells anywhere else in the body, which are able to form only neurons, ganglia, and pigment cells. The facial skeleton and most of the cranial vault structures originate from the neural crest and undergo intramembranous ossification; this differs from the process undergone by bones in other regions of the body, which are derived from mesoderm and which undergo endochondral ossification. 1
Cranial neural crest cells are also primary contributors to the fibroadipogenic progenitor cells of the face, whereas the fibroadipogenic cells in the trunk are of mesodermal origin. This may be an implication of the unique role of adipose-derived stem cells in the correction of craniofacial abnormalities. Some craniofacial abnormalities—for example, Treacher Collins syndrome, CHARGE syndrome, Waardenburg syndrome, and Mowat-Wilson syndrome—are directly associated with neural crest cell number, function, or ability to migrate. Thus the use of ADSCs may be a solution for repairing or reversing many craniofacial anomalies or defects. 2
Exploring the Expanding Ap plications of Fat Grafting
A few initial positive clinical observations about fat grafting were noted and inspired clinicians to use the beneficial adipose tissue properties not only for volume replacement but also for regeneration. After Rigotti’s published experience with treating the radionecrosis of irradiated breast tissue, 3 I started to graft fat to other areas of the body, including the craniofacial region.
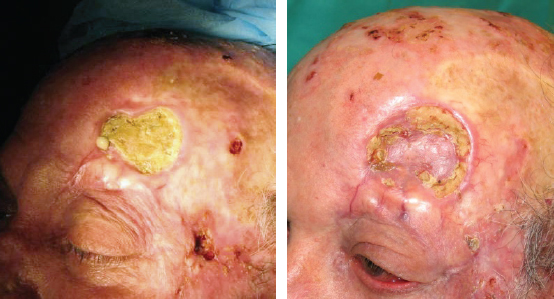
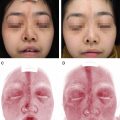
One of the first amazing results I achieved was in this 75-year-old patient with a deep radionecrotic ulcer after irradiation to the forehead for the treatment of basal cell carcinoma. Injecting processed fat to the indurated, purulent tissue with exposed necrotic bone did not garner much enthusiasm and support from my colleagues. I too was skeptical about the possible result in this patient. Surprisingly, at follow-up, the patient showed so much improvement that I was inspired to repeat the fat grafting after 3 months. The patient’s nearly healed ulceration is shown after debridement of the necrotic bone, two fat transfers, and a skin graft. Macroscopic neovascular regeneration was clearly visible in areas that were previously necrotic and deficient in blood vessels. The amazing results that could occur based on the regenerative properties of fat transfer were evident. The neovascularization potential of ADSCs has been thoroughly investigated and well described. 4
After this particular case, I started to use structural fat grafting in many difficult clinical cases, where the “traditional” approach did not produce results that were satisfactory aesthetically or functionally. Going forward, I employed fat grafting to reconstruct, regenerate, rejuvenate, and repair.
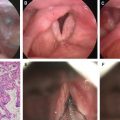
In oncologic patients, reconstruction with metal plates and free tissue transfers often produces unsatisfactory results. It exposes these individuals to multiple procedures, with further progressive scarring and the possibility of microsurgical compromise. A high rate of complications, including extrusion, is often very disappointing for both the patient and surgeon. Thus the ability of fat grafting to facilitate wound healing now plays an important role in these cases. 5 – 7
The neovasculogenic properties of fat can heal radionecrotic ulcerations and cover exposed metal implants, such as plates and screws. Conservative management with multiple fat grafting sessions can often minimize a soft tissue defect over an exposed plate and provide secondary closure.
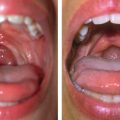
In malnourished, immunocompromised patients or in those undergoing oncologic treatment, fat grafting may be the only solution for nonhealing radionecrotic ulcerations, defects after failed flap surgery, or the treatment of persistent orocutaneous fistulas. This use of fat grafting is not very well described in the literature, so there may be doubt regarding the likelihood of a successful outcome.
However, on many occasions, structural fat grafting has helped me to salvage nonadhering flaps that had been transferred to previously irradiated wound beds, thus preventing further dehiscence. Perseverant debridement in combination with the injection of fat into the tissue surrounding a fistula has enabled healing without major surgical procedures and their potential complications.
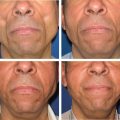
In general, fat grafts can be used to replace volume in contour defects, to restore symmetry, and to improve the quality of soft tissue, including scars and keloids. Small defects and disfiguring scars can be treated with a simple procedure instead of complicated soft tissue manipulation and flap surgery . Major posttraumatic soft tissue volume defects that would usually require microsurgical transfer can be very well reconstructed with structural fat grafting, which can be repeated to produce a satisfactory result. What can be frustrating and discouraging in such cases is the unpredictability of the wound with regard to the acceptance of injected fat.
In fact, surgeon patience and patient understanding are necessary to succeed in cases in which there are large-volume losses, extensive scarring, and the presence of allogenic implants. The results are not guaranteed because of the unpredictable take—or, rather, predictable fat graft necrosis—and the partial loss of original volume. Fat grafting is a minor surgery with a minimal complication rate that can be repeated until the optimal result is achieved. After the completion of a sometimes long process, these procedures actually carry a very high patient satisfaction rate.
Examples of current uses of fat grafting in craniofacial surgery are summarized in the table.
Material and Methods
Planning the reconstruction is the most crucial part of the preparation for surgery and should include the following:
Detailed analysis of the defect
Assessment of the types of tissue that need to be reconstructed (bone, soft tissue, muscle, skin)
Design of a one-stage or multistage reconstruction
Assessment of the completion of facial growth, when applicable
Evaluation of any comorbidities
Determination of patient expectations
My approach is to first correct bony abnormalities, which can be treated with a soft tissue cover in the same procedure or during a second stage. Volume replacement is essential for the final aesthetic result and should be dealt with during the final stage if not corrected at the time of the original reconstruction.
Despite ongoing debate about the optimal method for harvesting and processing fat and what instrumentation to use, there is still no consensus. The best described, most researched, and most commonly used method is the method described by Coleman (see Chapter 1). His technique includes the harvesting of fat through a handheld syringe, centrifugation of fat at 3000 rpm for 3 minutes, and then injection of the lipoaspirate through a blunt infiltration cannula with layered placement. 8 Eto and colleagues documented this method and illustrated how the fat transfer behaves in the recipient site 9 ; their research describes the pathophysiology of fat survival in great detail. With the use of the information provided by these researchers, it is easy to predict the fat take/absorption ratio. This makes planning much easier, and there is a level of comfort regarding the results that are likely to be achieved.
PLANNING
Planning for complex cases can be difficult. Designing a craniofacial procedure with fat grafting must be very precise. For patients with very severe posttraumatic or congenital deformities, it may be useful to stage the reconstruction. I recommend that bony defects be addressed first, delaying soft tissue reconstruction for at least 6 to 12 months to ensure settling of residual swelling and the completion of the healing process.
For patients with congenital abnormalities such as facial clefts, the main purpose of the fat graft is to provide cover for the defect to allow bone to grow, with possible interim distraction or bone grafting, if necessary, until facial growth is completed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree