Cellulite
Sara Hogan
Mara Weinstein Velez
Michael S. Kaminer
BACKGROUND
Cellulite (also known as gynoid lipodystrophy, adiposis edematosa, panniculosis deformans, nodular liposclerosis, edematous fibrosclerotic panniculopathy, local lipodystrophy, and status protrusus cutis) is a complex cellular dystrophy of dermal and subcutaneous tissue.1,2 Affected skin has a dimpled and/or nodular appearance, described as resembling an orange peel, cottage cheese, mattress or tufted cushion.3 It is most commonly found on the thighs, hips, buttocks, and, to a lesser extent, the stomach and posterior arms.
There are no epidemiological data on the incidence or prevalence of cellulite, although it is estimated to affect between 80% and 98% of postpubertal women.2,4 As such, it may be more appropriate to conceptualize cellulite as a female secondary sex characteristic.2,4,5 An estimated 2% men are affected.3 That cellulite occurs in nearly all women after puberty suggests an evolutionary significance. There is recent evidence that gluteofemoral subcutaneous adipose tissue has cardioprotective effects. Adipose located on the thighs and buttocks is inversely related to atrial stiffness and the ability of this tissue to entrap and store fatty acids and lipids, preventing their deposition into ectopic sites such as the liver, pancreas, and muscle.5,6,7 Cellulite may also play a role in energy homeostasis and insulin sensitivity via localized production of adipokines.8
PRESENTATION
Patients present with complaint of rippling, dimpling, or irregular contour of the skin most commonly on the hips, thighs, and buttocks.
DIAGNOSIS
Clinical Diagnosis
Initial assessment of a patient with cellulite should include a detailed history of previous cosmetic procedures and surgeries, trauma, peripheral vascular disease, and lymphatic disease. Information about recent weight loss or gain should be noted, and ideally weight should be stable for at least 3 months before treatment. Data such as weight, body mass index (BMI), and circumferential measurement of thighs, hips, and waist may be collected. Thigh circumference measurement can be used to assess response to treatment, but this has low reliability and decrease in circumference does not always correspond with reduction in cellulite severity.4
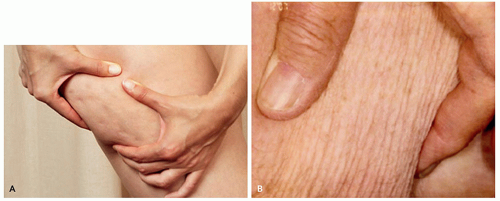
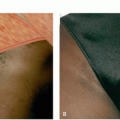
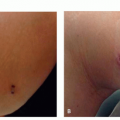
On physical examination, patients should be observed under tangential lighting both lying supine and standing and be evaluated for asymmetry and contour irregularities. Patient may be asked to contract muscles in affected areas to accentuate dimples.9 The pinch test forces adipose upward into the reticular and papillary dermis, making cellulite more evident to better assess severity (Figure 6.2.1).
A study of several objective measures (BMI, photonumeric analysis, cutometry, and high-frequency ultrasound) found poor correlation with clinical severity of cellulite.10 Stereoscopic systems, such as the Vectra 3D system (Canfield Scientific, Fairfield, NJ, USA), utilize multiple cameras to construct a 3-dimensional image of cellulite areas and can compute surface height and volume change within the target region. Stereoscopic measurement, however, is often too small to capture a cellulite pattern and does not directly measure cellulite itself.4 Magnetic resonance imaging (MRI) can identify dermal thickness and underlying septae of cellulite, and approximate cellulite severity, but requires combination with other modalities, such as confocal microscopy or histopathology, to observe treatment effect on connective tissue structure.11,12
Several different classification systems have been proposed to categorize cellulite by clinical and histopathological features. In their seminal 1978 paper, Nürnberger and Müller proposed the first classification scale, which graded cellulite severity from 0 to 313 (Table 6.2.1).
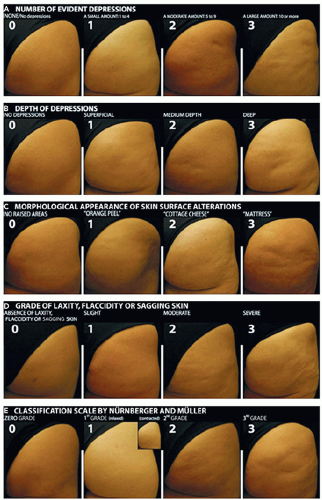
In 2009, Hexsel and colleagues developed the photonumeric cellulite severity scale to objectively assess the effects of treatment modalities. Considered the current standard for cellulite evaluation, 5 clinical features—(1) number of evident depressions; (2) depth of depressions; (3) morphological appearance of the skin surface alterations; (4) grade of laxity, flaccidity, or sagging skin; and (5) Nürnberger and Müller scale—are reviewed and each scored from 0 to 3. The final sum of scores, from 1 to 15, is calculated to classify cellulite as mild (1-5), moderate (6-10), or severe (11-15)14 (Figure 6.2.2).
TABLE 6.2.1 Nürnberger and Müller Scale for Grading Cellulite | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
Histopathology
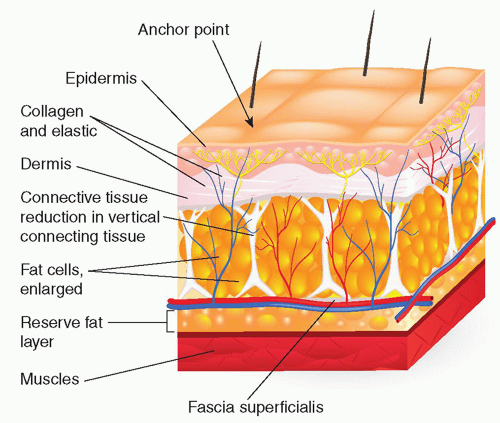
Subcutaneous adipose tissue is arranged in 2 layers of fat separated by 2 planes of connective tissue. The superficial, or areolar, layer is made of large, globular, vertically oriented adipocytes, whereas the deeper, or lamellar, layer contains smaller, horizontally oriented adipocytes and numerous blood vessels.2 The areolar layer tends to be more prominent in women and thickest in the femoral area.
In cellulite, thick fibrous septae extending from the deep connective tissue layer to muscle pull down on adipocytes, causing sequestration of fat into discrete pockets and the topographic depressions that are perceived clinically.1 Septae differ in thickness owing to varying density of myofibroblasts. These septae have 3 main orientations: perpendicular, parallel, and oblique (45° angle), and the difference in arrangement of these septae differs by gender.13,15,16 Anatomic studies show that women with cellulite have a higher a percentage of perpendicular septae than unaffected women or men, whereas men are more likely to have septae arranged in a “crisscross” or oblique pattern11,13,15 (Figure 6.2.3).
Magnetic resonance microimaging of cellulite demonstrates a diffuse pattern of extrusion of underlying adipose tissue into the dermis, which correlates with cellulite grading.11 Another study of thigh adipose biopsies found that this diffuse extrusion contains papillae adiposae, or standing fat lobules, which undulate along the dermal-hypodermal interface and are responsible for the “mattress-like” appearance of cellulite.16 Much less is known about raised, nodular components of cellulite. Because these areas can be painful and associated with increased temperature, an underlying inflammatory reaction may be involved. MRI of raised cellulite lesions failed to identify differences in adipose lobule size or septa compared with depressed lesions, with researchers positing that raised lesions may be a consequence of neighboring depressed lesions.17
Differential Diagnosis
Deep morphea
Eosinophilic fasciitis
Skin laxity
PATHOGENESIS
When first described by Albert and Paviot in 1920, cellulite was proposed to be a noninflammatory dystrophy of mesenchymal tissue, wherein defects in water metabolism led to saturation of adjacent tissues by interstitial fluid.2 It is now thought that cellulite is caused by a complex interplay of endocrine-mediated dermal and subcutaneous inflammatory events, microvascular dysfunction, and connective tissue fibrosclerosis.2,5
Recent studies show a correlation between cellulite and the insertion/deletion polymorphism in the HIF1A gene encoding for angiotensin 1-converting enzyme. Women with the D allele of the gene produce increased amounts of angiotensin II in their subcutaneous adipose tissue, causing decreased local blood flow and reduced expression of adiponectin.5 Women with the rare mutant T allele of the gene are protected against the development of cellulite owing to their blunted physiological fibroinflammatory response.18,19 From a genetic standpoint, a 2-hit hypothesis is then proposed for the development of cellulite: the first hit, a carriage of the D allele and localized adipose tissue hypoperfusion, and the second hit, a microenvironment of proinflammatory cytokines, decreased adiponectin expression and formation of collagen strands.5
Microvascular dysfunction in the subcutaneous tissue results in hypoxia, inflammation, and adipose tissue degeneration, followed by septal capillary neoformation and increased collagen synthesis and eventual fibrotic response around adipocytes with formation
of thick septae.2,5,20 Biopsies of cellulite show a mild inflammatory infiltrate of macrophages and lymphocytes and microhemorrhages in fibrous septae, which are thought to result in dermal atrophy. Herniated adipocyte lobules cause sustained mechanical tensions and prevent venous return, further contributing to hypoperfusion.21,22 This partially explains the observation that a sedentary lifestyle with prolonged periods of sitting or standing, and thus impediment of normal blood flow, is correlated with higher severity of cellulite.2
of thick septae.2,5,20 Biopsies of cellulite show a mild inflammatory infiltrate of macrophages and lymphocytes and microhemorrhages in fibrous septae, which are thought to result in dermal atrophy. Herniated adipocyte lobules cause sustained mechanical tensions and prevent venous return, further contributing to hypoperfusion.21,22 This partially explains the observation that a sedentary lifestyle with prolonged periods of sitting or standing, and thus impediment of normal blood flow, is correlated with higher severity of cellulite.2
The postpubertal onset of cellulite denotes the significant role of sex hormones in the pathogenesis of the condition. Menstruation requires endometrial secretion of the metalloproteases (MMPs), collagenase, and gelatinase to cleave triple helical domains of fibrillary collagen and water binding of glycosaminoglycans (GAGs), respectively.21 MMPs are also secreted in the dermis, and their cyclical secretion during monthly menstrual cycles allows for progressive dermal collagen destruction, adipose herniation, and worsening of cellulite with age.1 Estrogen activates fibroblasts and increases adipocyte response to anti-lipolytic alpha-2 adrenergic receptors and promotes GAG synthesis, which results in increased interstitial osmotic pressure and fluid retention.5 17-β-Estradiol, specifically, stimulates replication of adipocytes.2 During pregnancy, increases in prolactin, insulin, and overall fluid volume promote cellulite lipogenesis and fluid retention and cause the worsening appearance of cellulite.2 Testosterone, in contrast, stimulates connective tissue, resulting in decreased adipocyte protrusion through septae, and fewer dimples on the skin surface. This supports why so few men develop cellulite and explains the phenomenon whereby men with prostate cancer on estrogen therapy, develop cellulite.17
TREATMENT
The last decade has seen an exponential increase in the number of published studies on the efficacy of cellulite treatment, yet the body of literature is lacking.23 Research design is limited by small patient groups, lack of control groups, inadequate blinding of investigators, subjective efficacy end points, failure to test of statistical significance, and lack of extended follow-up.4,24,25 Additionally, because there is no biophysical measurement to quantify cellulite, evaluation of treatment efficacy is challenging. A systematic review of 67 cellulite treatment studies found no clear evidence of good efficacy for any of cellulite treatments evaluated.4 Another review of 73 studies noted significant heterogeneity in outcome measurements, as well as difficulty identifying effective and definitive therapy, and therefore concluded that no treatment is completely successful.20 The growing amount of evidence highlighting the importance of connective tissue fibrous septae in formation of cellulite, combined with the success of modalities targeting these structures (discussed later), suggests that this may be a major focus of both future research and therapy.
In the setting of lack of evidence-based treatment, it is therefore important to counsel patients that there may be variability regarding treatment response. Patients should be advised that noninvasive treatments may require multiple treatments before results are noted and that interval maintenance treatments may be required. Treatment modalities are reviewed in the following sections from least to most invasive.
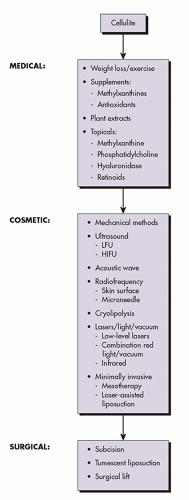
Algorithm 6.2.1 illustrates an overview of cellulite treatment.
Medical
Weight Loss
Higher BMI is positively correlated with increased severity of cellulite. This is primarily due to the finding that connective tissue structures in overweight and obese individuals are weaker and less dense, allowing for increased extrusion of adipose through the hypodermis.15 MRI of cellulite shows thinner dermis in people with high BMI and higher percentage of adipose inclusion in the dermis.11,17 Weight loss may reduce the dimpled appearance of cellulite and lessen cellulite severity.5,26 This effect, however, is not observed among all patients. In a study of 51 women recruited from weight loss programs, all participants were noted to have increased skin
compliance.26 Although the majority of participants had improvement of cellulite with weight loss, those who had increased skin laxity at baseline, appeared to have worse cellulite.26 This is consistent with the observation that cellulite worsens with aging and increased flaccidity of skin.14 Patients should be cautioned that, although weight loss often improves cellulite, it can exaggerate dimpling in a small subset of patients.
compliance.26 Although the majority of participants had improvement of cellulite with weight loss, those who had increased skin laxity at baseline, appeared to have worse cellulite.26 This is consistent with the observation that cellulite worsens with aging and increased flaccidity of skin.14 Patients should be cautioned that, although weight loss often improves cellulite, it can exaggerate dimpling in a small subset of patients.
Dietary Supplements
A diet that is high in carbohydrates promotes hyperinsulinemia, lipogenesis, and, subsequently, cellulite formation. When weight loss occurs in the setting of a low-calorie diet, β1 and β2 lipolytic adrenergic receptors have increased sensitivity, although more so in abdominal adipocytes than femoral adipocytes.27 Subcutaneous tissue in women has decreased sensitivity of α2 anti-lipolytic adrenergic receptors compared with men and has more α2 receptors located in the femoral area.27 Fasting causes decreased sensitivity of α2 receptors, which explains why adipocytes in the gluteofemoral area remain unchanged in size unless a person is fasting for an extended period of time and thus why people with a genetic predisposition for gluteofemoral adipose deposits have the greatest difficulty losing weight in these areas.1,27
Hundreds of over-the-counter dietary supplements to address cellulite are marketed to the general population with claims of improving metabolism, enhancing microcirculation, promoting collagen and elastin synthesis, and decreasing edema. Common ingredients in these supplements include herbal agents, methylxanthines such as caffeine, and antioxidants. In one study, Cellasene (Medestea Internazionale, Torino, Italy), a botanical complex of Vitis vinifera (common grape vine), ginkgo biloba, Centella asiatica (gotu kola), Melilotus officinalis (sweet clover), Fucus vesiculosus (bladder wrack seaweed), fish oil, and borage seed oil, was found to be superior when compared with control subjects in cellulite improvement and reduction of thigh circumference; another study of the same complex, however, found not only no improvement in cellulite but also an increase in body weight among subjects.28,29 Another study of patients who consumed 100 mL of organic chokeberry juice for 90 days found improvement in thickness of all skin layers at the end of the treatment period.30 A clinical trial of a bioactive collagen peptide supplement in women with moderate cellulite found on imaging that the treatment group had statistically higher dermal density after 6 months, with the effect more noticeable in normal-weight participants (compared with their overweight counterparts).31 The authors attributed these results to improved dermal structure from extracellular
matrix protein biosynthesis and rat model findings that the supplement increased expression of collagen I and IV and reduced MMP activity.31
matrix protein biosynthesis and rat model findings that the supplement increased expression of collagen I and IV and reduced MMP activity.31
Topical Treatments
The majority of cellulite treatments available to consumers are topical treatments, available for purchase over the counter at drug stores, department stores, mass merchandisers, boutiques, and spas.1 The diversity of active compounds in topical cellulite treatments poses difficulty in assessing the effectiveness of ingredients.5 One study identified 232 ingredients in 32 different cellulite creams, predominantly botanicals, emollients, and caffeine.32
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree