Introduction
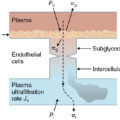
Fluid resuscitation is an early and essential component of the care of the burn patient. Thermal injury, particularly that exceeding 20% of the total body surface area (TBSA), causes burn shock. This problem lasts 24 to 48 hours and requires active fluid resuscitation. Burn shock features not only hypovolemia secondary to loss of fluid similar to plasma across the microvasculature but also varying degrees of decreased intrinsic myocardial contractility and increased systemic vascular resistance (SVR)—all of which decrease cardiac output and impair end-organ perfusion. In particular, loss of fluid across the microvasculature is the result of direct tissue damage in burned areas and, in patients with larger burns, of the systemic effects of massive inflammation. Recently, the occurrence of damage to the endothelial glycocalyx (termed endotheliopathy) has been recognized as an additional feature of burn shock. Without adequate replacement of ongoing fluid losses from the microvasculature, the patient will develop tissue and organ dysfunction, leading possibly to death. However, excessive fluid resuscitation, by exacerbating edema formation in the interstitium, is likewise hazardous. Thus timely and judiciously titrated fluid resuscitation following burn injury is vital.
Many methodologic areas of uncertainty and even contention remain to be better defined. There is no one best method of burn resuscitation accepted by all burn care professionals. Indeed, this variability is a consistent theme in various forums, such as the American Burn Association (ABA). Regardless of the approach to burn resuscitation, there are several universal aims: to (1) replenish the loss of circulating fluid, (2) accurately monitor the patient’s response to resuscitation, (3) modify intervention based on this response, and (4) prevent the complications of overresuscitation and underresuscitation. Resuscitation should restore intravascular volume at the lowest possible physiologic cost, supporting the patient while awaiting gradual resolution of increased microvascular permeability and other initial derangements.
Development of resuscitation formulas
Recognition of the need for adequate fluid resuscitation following burn injury and the development of effective protocols were important advances in burn care during the last century. Scientific recognition and study of the benefits of fluid resuscitation began with the cholera epidemics of the 19th century. O’Shaughnessy recommended intravenous (IV) fluid administration as a treatment modality for cholera. He studied the treatment in dogs, but not in humans. Latta subsequently treated human cholera patients with fluid resuscitation via the oral, rectal, and IV routes. Lewins described the use of an IV salt solution, recognizing that the volume of resuscitation required was correlated with the amount of fluid lost. Hartmann modified a physiologic salt solution developed by Ringer, in 1885, for rehydration of pediatric patients with infectious gastroenteritis. Starling, in 1896, described the action of capillaries and postcapillary venules as semipermeable membranes for the movement of fluid between the intravascular and interstitial spaces. This led to the identification of hydrostatic and oncotic pressures as the driving forces for fluid exchange and the now widely used Starling equation.
Fluid resuscitation for burn injury, however, was not utilized in the 19th century. Several authors suggested that burns should be treated with diuretics, bloodletting, venesection, and/or leeching. Sneve, in 1905, described the use of saline solutions, including enemas, for the treatment of burn injuries. Initially, his recommendations were met with skepticism because of the prevailing view that the cause of death following an extensive burn was absorption of toxic substances from the burned skin, and not hypovolemia.
In 1921, following the Rialto Theatre fire in Connecticut, Underhill reported that burn severity was correlated with the degree of hemoconcentration, which was caused by fluid depletion. He noted that fluid replacement following a burn injury must be rapid and was essential to survival. Additionally, he showed that the blister fluid was similar in composition to plasma and that the fluid lost following burn injury could be replaced intravenously with a physiologic salt solution.
Blalock, in 1931, studied burn shock in a canine model. Animals received large burns to one side of the body and were not resuscitated. Subsequent analysis revealed a large amount of fluid loss across the burn wounds accompanied by an increase in the hemoglobin level. Blalock concluded that this process was the major culprit in postburn hypovolemia and hypotension.
World War II brought an increase in combat burn injuries and greater emphasis on better treatment for these injuries, including initial resuscitation. Meanwhile, human blood plasma became readily available for IV use. These two events led to several new proposals for burn resuscitation utilizing IV plasma. A National Research Council conference in January 1942 proposed that burn patients receive an initial bolus of 500 mL plasma followed by 100 mL plasma per percent burn over the first 24 hours after injury. Harkins subsequently proposed a similar formula with a bolus of 1000 mL normal saline (NS) followed by 50 mL plasma per percent burn over the first 24 hours. He also recommended administration of large amounts of dextrose by mouth. Harkins also discussed the use of a formula in which 100 mL plasma was administered for every hematocrit point above 45%.
Several other burn resuscitation formulas were described during this time. Rhoads et al. recommended a continuous plasma infusion adjusted based on maintenance of peripheral circulation, as assessed by blood flow following a skin needle prick. Berkow recommended 50 mL plasma per percent burn, with one-third of the volume given in the first 2 hours, one-third in the next 4 hours, and the final third in the subsequent 6 hours. Black described a formula of plasma administration based on hemoglobin level. Elkinton published a complicated formula incorporating patient body weight, along with plasma protein and hematocrit levels.
The 1942 Cocoanut Grove nightclub fire in Boston was a watershed event in burn resuscitation. The fire led to 492 deaths and hundreds of injuries. Cope, at the Massachusetts General Hospital, resuscitated survivors with plasma and saline in a 1:1 ratio, 100 mL per percent burn. Lund, at Boston City Hospital, used a similar mixture of plasma and saline, but used clinical signs such as heart rate, blood pressure, and hematocrit as guides to resuscitation. Cope and Moore later described a formula that recommended 75 mL plasma and 75 mL isotonic crystalloid per percent burn, administered over the first 24 hours. They recommended that one-half of the calculated 24-hour fluid volume be administered in the first 8 hours and the second half in the subsequent 16 hours. This was the first description of the concept of giving one-half of the 24-hour total in the first 8 hours. This concept is retained in most formulas in current common use, to include the Parkland and Brooke formulas described later. Moore subsequently reported a more complex formula based on body weight.
An important component in the development of fluid resuscitation protocols was the introduction of methods to estimate the extent of burn injury. Berkow described a method in 1924 of estimating the extent of burn injury based on BSA proportions. Lund and Browder developed a now widely used chart to estimate burn size. The rule of nines, a rapid method to estimate the extent of burn injury, was published by Wallace in 1951. These and other burn-size estimation tools enabled wider use of fluid replacement protocols based on the extent of burn.
Evans et al., in 1952, incorporated burn size and patient weight in a formula, which provided 2 mL/kg/TBSA of a solution containing a 1:1 mixture of colloid and saline, plus 2000 mL 5% dextrose in water (D5W), in the first 24 hours. During the second 24 hours, 0.5 mL/kg of a 1:1 colloid to saline mixture, plus 2000 mL D5W, was given. Reiss et al., in 1953, replaced NS with lactated Ringer’s solution (LR) and reduced the proportion of colloid. The formula was 0.5 mL/kg/TBSA colloid and 1.5 mL/kg/TBSA crystalloid given over the first 24 hours. This was the original Brooke formula.
During the ensuing 2 decades there was a further move away from colloid use during the first 24 hours postburn. Baxter and Shires wrote in 1968 that administration of LR at a faster rate and higher volume than predicted by existing burn resuscitation formulas more rapidly restored functional extracellular fluid volume, hemodynamics, and acid-base status. The resulting Parkland or Baxter formula recommended LR at 4 mL/kg/TBSA during the first 24 hours. The formula then utilized plasma administration in the second 24 hours after injury to assist in restoring plasma volume to normal. For many years, this was the most widely used resuscitation formula.
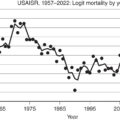
Pruitt et al., at the US Army Institute of Surgical Research (USAISR), reported in 1971 that varying doses of colloid used during the first 24 hours for resuscitation were no more effective than crystalloid alone with respect to the rate of plasma volume loss. This observation resulted in a modification of the original Brooke formula to 2 mL/kg/TBSA of LR over the first 24 hours, without the administration of colloid. They found that colloid became effective in the second 24-hour period and included that in their modified formula. The current formula for the second 24 hours uses a sliding scale for 5% albumin dosing based on burn size, ranging between 0.3 and 0.5 mL/kg/TBSA over 24 hours ( Table 8.1 ). Goodwin et al., at the same center, performed a randomized controlled trial of colloid- vs. crystalloid-based resuscitation. Patients in the colloid arm received 2.5% albumin in LR, and those in the crystalloid arm received LR alone. The colloid group experienced a more rapid restoration of cardiac output and received less fluid during the first 24 hours. However, there was an associated increase in extravascular lung water and an increase in all-cause in-hospital mortality. This reinforced the preference for avoiding colloids in the first 24 hours.
Table 8.1
USAISR Sliding Scale for Dosing 5% Albumin during Burn Resuscitation
| Burn Size (%TBSA) | Albumin Dose |
|---|---|
| 0–29% | 0 |
| 30–49% | 0.3 mL/kg/TBSA |
| 50–69% | 0.4 mL/kg/TBSA |
| 70–100% | 0.5 mL/kg/TBSA |
%TBSA , Percentage of the total body surface area burned; USAISR , US Army Institute of Surgical Research.
During the recent conflicts in Iraq and Afghanistan, and to simplify calculations for use on the battlefield, Chung et al., at the USAISR, proposed a rule of tens formula for adult burn resuscitation. Initial fluid rate in milliliters per hour is TBSA × 10, with an additional 100 mL/hour for each 10 kg of body weight greater than 80 kg. This generally results in a 24-hour volume of resuscitation that falls somewhere between the Brooke (2 mL/kg/TBSA) and the Parkland (4 mL/kg/TBSA) calculations.
The 2008 ABA Practice Guidelines for Burn Shock Resuscitation and two ABA State of the Science meetings (2006, 2016) recognized that an evidence-based standard of care is lacking. ABA guidelines recommend IV administration of fluid for burns greater than 20% TBSA. The Advanced Burn Life Support (ABLS) course recommends use of the modified Brooke formula for adults.
Currently, most North American burn centers use either the Brooke or the Parkland formula for initial fluid resuscitation of adults. Generally, the fluid rate is calculated based on administering one-half of the recommended fluid during the first 8 hours. For example, an 80-kg patient with a 40% TBSA burn resuscitated with the modified Brooke formula would be programmed for 2 × 80 × 40 = 6400 mL LR during the first 24 hours postburn. One-half of this, or 3200 mL, would be given over the first 8 hours. Thus the starting LR infusion rate would be 3200 mL/8 hours = 400 mL/hour.
The formulas only provide a starting point for fluid resuscitation, and no abrupt change is made at 8 hours. Subsequent fluid rates are adjusted based on the physiologic response, primarily urine output (UO). Burn centers vary in whether and how colloid is administered. It may be given immediately, not at all, at the 24-hour point, or earlier than that based on patient response. These issues are discussed in further detail later.
Burn resuscitation formulas have been developed specifically for thermally injured children. These formulas tend to differ in two ways from those developed for adults. First, pediatric formulas often are designed to account for the larger surface area to weight ratio seen in children. This usually means a higher volume of resuscitation fluid. The second difference is the addition of maintenance fluid, typically based on, for example, the 4-2-1 rule (4 mL/kg/hour for the first 10 kg, 2 mL/kg/hour for the second 10 kg, 1 mL/kg/hour for the third 10 kg). There are several purposes for this additional maintenance fluid. First, it addresses the fact that smaller children with smaller burns might otherwise receive less fluid for resuscitation than they require for maintenance. Additionally, dextrose serves as a carbohydrate energy source that is useful because of the relatively low levels of hepatic glycogen in small children. Finally, the saline serves as an additional source of sodium that helps prevent hyponatremia in children. For children, the ABLS course recommends the use of 3 mL/kg/TBSA, plus maintenance fluid as D5W LR.
Oral rehydration therapy for burn resuscitation
As described extensively in this chapter, IV administration is the recognized standard for burn resuscitation. In a resource-limited setting, however, IV resuscitation may not be possible. Deficiencies may include a limited number of trained health care personnel, lack of appropriate IV catheters and/or tubing, and a shortage of sterile IV fluids of appropriate electrolyte composition. Additionally, patients may live great geographic distances from a health care facility, and presentation may be delayed. In such circumstances, oral rehydration therapy (ORT) should be considered. ORT is a well-established technique for fluid resuscitation. , It has been shown to be effective in rehydrating patients with diarrhea in austere environments. The method is not technically challenging, can be performed by minimally trained, non–health care personnel, does not require any special equipment, and is remarkably inexpensive. Although prospective randomized clinical trials of ORT are still needed, several researchers have demonstrated the effectiveness of ORT in burn resuscitation.
The human gastrointestinal tract has the capacity to absorb large amounts of delivered fluid, up to 20 L/day. This absorptive capacity is retained even in the presence of thermal injury up to 40% TBSA. However, the composition of ORT fluid is important. Ingestion of large amounts of electrolyte-free water can lead to profound hyponatremia and the ravages thereof, including cerebral edema and even death. Sodium is an absolute requirement in ORT-based burn resuscitation. Sodium uptake through the small bowel is facilitated by the sodium-glucose cotransporter. Intestinal transmembrane transport of one glucose molecule is coupled to that of two sodium ions, resulting in an osmotically driven influx of more than 200 water molecules. Thus inclusion of a glucose source in ORT formulas enhances sodium and water intake by the patient. Other molecules that support water absorption include amylase-resistant starch, l -arginine, and several amino acids, but these are not widely available.
Cancio et al. listed 12 reports from 1944 to 1991 with a total of 709 patients in which ORT was used for burn resuscitation. Several important findings were noted. The use of NaCl alone led to metabolic acidosis. This is the reason for the recommended use of buffered salt solutions. Patients with burns greater than 50% TBSA were not successfully resuscitated with ORT alone. Vomiting was a common adverse effect associated with ORT resuscitation; however, it was usually successfully treated with antiemetics and continuation of ORT resuscitation. One author noted that tolerance of oral resuscitation was related to starting early, ideally within the first hour after injury. Children seemed to be relatively more tolerant of oral resuscitation than adults.
There are numerous commercially available and homemade ORT solutions. Since 1975, the United Nations Children’s Fund and the World Health Organization (WHO) have provided packets of oral resuscitation salts (ORS) to be used in the treatment of infectious diarrhea. The WHO ORS contain sodium, chloride, glucose, potassium, and citrate. Other pharmacologic options have been described in detail by Peck et al. Greenleaf et al. varied the sodium content and overall solution osmolarity and found that a nearly isotonic electrolyte solution was the most effective at expanding plasma volume during resuscitation. Tables 8.2 and 8.3 provide recipes for homemade ORT solutions. Note that salt can be substituted for baking soda if the latter is unavailable.
Table 8.2
Recipes for Homemade Oral Rehydration Solutions
From Cancio LC, Kramer GC, Hoskins SL. Gastrointestinal fluid resuscitation of thermally injured patients. J Burn Care Res . 2006;27(5):561-569.
| Liquid | Sugar | Salt | Baking Soda |
|---|---|---|---|
| Clean water (1 L) | 8 tsp | ½ tsp | ½ tsp |
| Gatorade (1 qt) | 0 | ¼ tsp | ¼ tsp |
| Lactated Ringer’s (1 L) | 8 tsp | 0 | 0 |
Table 8.3
Composition of Hypertonic Saline Solutions for Burn Resuscitation
| Author | Year | Na Concentration | Other Constituents |
|---|---|---|---|
| Monafo (first formula) | 1970 | 300 mEq/L | Lactate 200 mEq/L, chloride 100 mEq/L |
| Monafo (second formula) | 1973 | 250 mEq/L | Lactate 100 mEq/L, chloride 150 mEq/L |
| Warden | 1992 | 180 mEq/L | NaHCO 3 50 mEq/L lactated Ringer (LR) |
| Huang | 1995 | 290 mEq/L | Na acetate 160 mEq/L LR |
| Oda | 2006 | Decreased over postburn day 1, from 300 to 150 mEq/L, in 50-mEq/L steps | Variable |
Rectal infusion therapy, also known as proctoclysis, is another technique that may be helpful in austere environments. Proctoclysis was documented as an effective treatment following battlefield injuries in both world wars. Rectal infusions of either saline or tap water have been tolerated at rates up to 400 mL/hour. Therapeutic proctoclysis typically uses NS solution, and the total volume given may be as great as 6 L/day. Proctoclysis is a viable option for resuscitation when IV resuscitation is unavailable. It may be used alone or in conjunction with ORT.
Resuscitation fluid choice
Crystalloids
The earlier sections delineate the rate and route of fluid administration during burn resuscitation. This section will discuss the type of fluid(s) used in resuscitation. Current ABLS and ABA practice guidelines for burn shock resuscitation recommend crystalloid-based resuscitation. There are no definitive evidence-based studies to guide the choice of crystalloid solutions. The most commonly used fluid at this time is LR, which has been used successfully for burn shock treatment for years. NS was used previously as a resuscitation fluid; however, relatively large volumes of NS may be associated with hyperchloremic metabolic acidosis. Additionally, NS may decrease renal blood flow and glomerular filtration rate, predisposing to acute kidney injury. Plasma-Lyte has an electrolyte composition (sodium 140 mmol/L; gluconate and acetate instead of lactate), osmolarity (295 mOsm/L), and pH (7.4) that is closer to that of plasma than is LR, suggesting possible utility in burn resuscitation. However, there are no studies comparing the two solutions in burn resuscitation.
Hypertonic saline
Another approach to fluid management during resuscitation is the use of hypertonic saline (HS). There are a variety of preparations available with differing concentrations of sodium and anions. Monafo proposed that a lactated HS solution could achieve successful resuscitation with smaller volumes of fluid. A 1995 retrospective study by Huang et al. compared patients treated with HS to two cohorts treated with LR, one pre-HS and one post-HS. The HS group demonstrated a fourfold greater increase in acute kidney injury and twice the mortality. This report led to diminished use of HS during burn resuscitation. More recent trials have demonstrated safety and efficacy for HS during acute resuscitation. HS may be useful in patients who are particularly sensitive to volume overload (e.g., patients with congestive heart failure or advanced cirrhosis) or in patients with complicated resuscitations.
HS expands plasma volume and shifts water into the intravascular space, primarily at the expense of intracellular water depletion. It is unclear if this fluid shift is beneficial or harmful. Proposed benefits of this HS effect are much like those for colloid: decreased volume administration for a given UO, less risk of abdominal and extremity compartment syndromes, possibly improved pulmonary function, and better perfusion to injured tissue. The serum sodium must be monitored closely with HS use because of the risk of hypernatremia. Sodium levels greater than 160 mEq/L have associated with adverse cerebral and renal effects. Most studies of HS in burn patients enrolled small numbers. Although the majority of these studies demonstrate reduced volume of resuscitation fluid, it is not clear that this actually confers any meaningful clinical benefit.
Consisting of 7.5% saline and 6% dextran-70, hypertonic saline dextran (HSD) was originally developed for prehospital or battlefield use and is much more concentrated than the preparations of HS described earlier. In animal studies, HSD demonstrated an early fluid-sparing effect and a positive effect on cardiac function. , Interest in HSD has waned in recent years.
Colloids
There are several controversies associated with colloid use in burn resuscitation. The first is whether colloid should be used at all. The second is when colloid should be administered. The third is what type of colloid should be used.
Consistent with the study by Goodwin et al. described earlier, Du et al. demonstrated that colloid administered as fresh frozen plasma (FFP) with a fixed 2-L volume of LR reduced the total volume of fluid administered during the first 24 hours of resuscitation. Other studies with colloid have yielded similar results. Thus if the goal is reduction of total fluid administered during resuscitation, the addition of colloid achieves that goal in most circumstances. The mechanism for this effect is likely multifactorial. One of these factors is gradual restoration of microvascular integrity over time (see later). Another factor is correction of hypoproteinemia. Administration of large volumes of crystalloid during burn resuscitation dilutes and decreases plasma protein concentration and thus may accelerate the movement of fluid from intravascular to extravascular spaces, causing increased edema formation. Administration of a colloid solution could theoretically mitigate this effect.
The timing of colloid administration varies across burn centers. There are three general approaches: immediate use, early use beginning about 8 to 12 hours postburn, and delayed use beginning about 24 hours postburn. Some use colloid as a rescue fluid when crystalloid use becomes excessive or when complications of fluid overload become manifest. For example, Cochran et al. used albumin when resuscitation fluid volume exceeded Parkland formula recommendations. There are limited data on timing in humans. In an ovine model, Demling et al. demonstrated that in early postburn there is an increased rate of edema formation (measured as the lymph flow rate in a chronic lymph fistula) and an increased lymph to plasma protein ratio. By these measures, the ability of the microvasculature in unburned tissue to sieve proteins begins to recover between 8 and 12 hours postburn. This suggests that colloids may become more effective than crystalloids alone for volume expansion beginning about 8 to 12 hours postburn.
With respect to the type of colloid, 5% albumin is the most common colloid currently in use in burn centers. Although early formulas for resuscitation utilized plasma, viral transmission made it less than ideal, and thus 5% albumin replaced it. The availability of plasma safe from viral transmission now warrants its reconsideration. Limited data suggest that FFP may be superior to albumin in protecting the endothelial glycocalyx and that FFP therefore may be more effective in reducing the volume of fluid necessary for resuscitation. , , A prospective randomized controlled trial of plasma- vs. crystalloid-based resuscitation is underway.
Hetastarch solutions have been suggested for burn resuscitation. Outcomes regarding total volume ultimately administered are equivocal, and there may not be an advantage in other outcomes, such as renal and pulmonary dysfunction, length of stay, and mortality. , A recent Cochrane review showed an increase in acute kidney injury with these solutions. Thus hetastarch solutions are not recommended for burn resuscitation at this time.
A Cochrane review from 1998 analyzing the use of albumin in critically ill patients found an increased absolute risk of death of 6%. However, these studies included a variety of critically ill patients, and only three studies had burn patients. Each of these three studies was problematic from an analysis standpoint. Thus the conclusions drawn by the review cannot be confidently applied to burn patients. More recent randomized controlled trials have demonstrated the safety and efficacy of albumin use in critically ill patients, including burn patients. A multicenter trial of LR vs. 5% albumin plus LR showed no difference in the primary endpoint of multiple organ dysfunction score (MODS) to day 14. A meta-analysis of burn trials failed to demonstrate an effect of albumin use on mortality, but did show an 81% risk reduction for extremity compartment syndrome.
ABRUPT, a recent observational study by Greenhalgh et al., reported the resuscitation practices of 21 North American burn centers, of which 379 patients with burns greater than 20% TBSA were included. One-third of patients were resuscitated with crystalloid alone, and two-thirds were resuscitated with albumin and crystalloid. Patients in the albumin group received more total fluid than the crystalloid-only group (5.2 ± 2.3 vs. 3.7 ± 1.7 mL/kg/TBSA) and had worse outcomes (e.g., length of stay, ventilator days, acute kidney injury, and time to completion of resuscitation). However, patients in the albumin group were older, had larger burns, had greater incidence of inhalation injury, and had higher admission Sequential Organ Failure Assessment scores. The authors concluded that crystalloid alone was more likely to be used in patients with smaller burns, and albumin in older patients with larger burns. Finally, they stated that their results justified a prospective, randomized, multicenter trial comparing crystalloid alone to crystalloid with albumin; that study is in progress.
Monitoring resuscitation
Monitoring adequacy and effectiveness is key to providing successful resuscitation. UO is the most common resuscitation indicator. UO is physiologically sensible because it presumes adequate cardiac output, oxygen delivery, and renal perfusion. There is a paucity of evidence demonstrating the ideal hourly UO during resuscitation. The ABA guidelines recommend a UO of 0.5 mL/kg/hour in adults, 1 mL/kg/hour in children, and 2 mL/kg/hour in infants. ABLS includes the same recommendations, excluding 2 mL/kg/hour in infants. However, many authors target a lower UO, namely 30 to 50 mL/hour, in adults. Maintaining a UO at or above these recommendations signals effective resuscitation, while UO that falls below these levels signals inadequate resuscitation and is an indication for more aggressive fluid administration.
UO is typically measured hourly via a Foley catheter, and changes in the IV infusion rate are made based on whether this UO is greater or less than the target. It is unclear whether hourly measurement is ideal or whether it should be done more or less frequently. In the absence of protocolized resuscitation (see later), it is reasonable to titrate the fluid infusion rate up or down by about 20% every 1 to 2 hours, aiming to keep the UO within the target range mentioned earlier. To avoid overresuscitation, it is as important to titrate fluids down when the UO is too high as it is to titrate them up when the UO is too low.
The disadvantage of UO as an indicator is that in some patients it may be inherently abnormal and therefore not reliable. This may be seen in patients with acute or chronic renal failure, in those on diuretics, in those who are intoxicated with ethanol, or in those with glucosuria. In such patients, additional variables must be monitored as described later.
Heart rate may be a reasonable indicator of fluid status in nonburn patients; however, because of the marked adrenergic response that accompanies a significant burn, adult burn patients will likely have tachycardia with heart rates between 100 and 120 bpm despite adequate resuscitation. A higher heart rate during burn shock should prompt a reexamination of the patient’s volume status or other causes of tachycardia. Conversely, failure to mount a tachycardic response to an extensive injury is a poor prognostic indicator, may indicate underlying heart disease, and should prompt evaluation thereof.
High circulating levels of catecholamines also tend to maintain the blood pressure despite progressive hypovolemia during burn shock, particularly in young and otherwise healthy individuals. Thus a normal blood pressure should not be taken as an indicator of adequate resuscitation. On the other hand, the occurrence of hypotension during burn shock is worrisome and should be addressed promptly with (1) reassessment of volume status and cardiovascular function, (2) consideration of an increase in the fluid infusion rate, (3) consideration of a vasoactive infusion (such as norepinephrine) as a temporizing measure, and (4) reevaluation of the patient’s narcotic and sedative regimen. In particular, the use of sedative-hypnotics (e.g., propofol), which adversely impact sympathetic nerve activity and the ability to compensate for hypovolemia, should be minimized during burn shock. A mean arterial pressure target of 60 mmHg is a reasonable goal for most patients during resuscitation.
The results of laboratory tests, such as complete blood count, electrolytes, glucose, and renal and hepatic function studies, are routinely obtained but are not particularly useful as indicators of resuscitation adequacy. However, hemoconcentration (i.e., an elevated hemoglobin and hematocrit), hypernatremia, and an elevated blood urea nitrogen to creatinine ratio may be seen early in burn shock. Normalization of these values with resuscitation is a reasonable goal of therapy; in fact, several early resuscitation formulas were based on hematocrit monitoring.
Serum (or arterial) lactate and arterial base deficit tend to correlate with burn size, the presence of inhalation injury, shock severity, and ultimate mortality; these should be routinely monitored during resuscitation. Cartotto et al. demonstrated a correlation between a mean base deficit during the first 24 hours postburn less than −6 mmol/L, and more severe systemic inflammatory response syndrome on day 1, increased prevalence of acute respiratory distress syndrome (ARDS), and increased multiorgan dysfunction syndrome. Other studies have demonstrated that levels of lactate and base deficit are useful markers of shock and resuscitation status. Decreasing values with ongoing resuscitation indicate effective resuscitation, with normalization being the ultimate goal. Decreasing time to normalization of lactate and base deficit has also been shown to be correlated with survival. How best to use these data is, however, uncertain. In trauma patients, base deficit and lactate correlate modestly. Furthermore, factors other than shock severity affect these values. Lactate, for example, is increased by drugs such as epinephrine, which increase its production, and by diseases such as cirrhosis, which impair its clearance. In the presence of a normal anion gap, a base deficit likely reflects processes other than lactic acidosis, such as renal tubular acidosis, large-volume crystalloid resuscitation, or ethanol intoxication. ,
A number of more complex techniques have been used to monitor resuscitation; these may be useful when there are concerns about the reliability of UO data. Pulmonary artery (Swan-Ganz) catheters were once used frequently to assist in resuscitation, particularly in so-called goal-directed therapy. The use of these devices is now largely reserved for patients in whom significant questions arise concerning cardiac function that cannot be resolved by means of echocardiography. Transthoracic echocardiography is a convenient and less invasive way to evaluate cardiac function and volume status. It can be performed at the bedside, provides immediate information, and can be used to guide fluid administration and ionotropic/pressor therapy. Disadvantages include lack of continuous data and the need for trained operators.
Arterial waveform analysis has been used to provide an estimate of beat-to-beat stroke volume, which in turn can be used to calculate stroke volume variability—a measure of volume responsiveness. Another method of predicting volume responsiveness is measurement of pulse pressure variability. These variability-based metrics have not been adequately assessed during burn shock, and the risks of treating volume responsiveness in a patient who is otherwise responding appropriately to burn shock resuscitation must be considered cautiously. Near-infrared spectroscopy, gastric tonometry, and sublingual capnometry have each been shown to be effective in various patient populations but are not widely available.
How does one put all these data together during a difficult resuscitation? Internal and external inconsistencies should be identified and analyzed. Internal inconsistencies among the data (discordant findings) should prompt further investigation. Examples abound: The patient with an adequate UO and a rising lactate may have a missed injury or other explanation for lactic acidosis. The patient with falling UO despite increasing fluid infusion may have acute renal failure or abdominal compartment syndrome. External inconsistencies between the patient and the expected patient trajectory should be evaluated. The team must interpret the data in context. Burn resuscitation is a dynamically changing process. A patient’s status while moving through this process is expected to change from hour to hour. This means that the expected cardiovascular performance of a patient who is on a successful resuscitation trajectory (“on target”) looks different at hour 4 than at hour 24. At hour 4, cardiac output is low, SVR is high, and the hourly infusion rate is often being increased. At hour 24, cardiac output is increasing, SVR is decreasing, and the hourly infusion rate should be coming down. Divergence from these patterns requires analysis.
Adjunctive therapies for burn resuscitation
Typically, adjunctive therapies come into play in the face of complicated or failing resuscitation. These therapies include early surgical intervention, infusion of inotropes/vasopressors, continuous renal replacement therapy (CRRT), plasma exchange, and administration of IV vitamin C.
When a patient begins to require more fluid than predicted by the formula in use, providers should remap the patient and ensure that the extent of burn is not greater than originally estimated. Additionally, the burn should be examined to see if there is a large full-thickness component to the injury, which could explain increased fluid needs. Likewise, consideration should be given to an undiagnosed concomitant traumatic injury. Paracentesis, or decompressive laparotomy, may be required to relieve abdominal compartment syndrome. Early burn excision (typically a fascial excision) has been suggested as an approach to the patient who is failing resuscitation, although evidence to support this high-risk practice is lacking.
The infusion of cardiovascular drugs such as pressors or inotropes should theoretically not be needed during burn resuscitation primarily because the burn leads to maximal endogenous secretion of catecholamines. Nevertheless, in some patients, even after appropriate fluid administration has been initiated, therapy with drugs such as norepinephrine or epinephrine may be required to support blood pressure and to help maintain tissue perfusion. Vasopressin is routinely added to the catecholamine. Drugs such as dobutamine, which are peripheral vasodilators, should be used with prudence in hypovolemic burn patients because of the possibility that they may induce hypotension. They may be useful in patients whose preload is adequate but whose cardiac output remains diminished. Monitoring with echocardiography or a pulmonary artery catheter may be useful to guide therapy.
In some burn centers, CRRT is routinely used to assist with electrolyte and acid-base management during difficult burn shock resuscitations and to permit more rapid and controlled deresuscitation (fluid removal) after the burn shock phase. This practice is based in part on the work of Chung et al., who demonstrated that septic burn patients with acute kidney injury treated with CRRT had improved survival and less need for pressors than historic controls. It was speculated that the mechanism of CRRT in this context was nonspecific removal of inflammatory cytokines. More research on the mechanism and efficacy of CRRT during burn shock is warranted.
In total plasma exchange (TPE), the patient’s plasma volume is replaced with FFP. The theory proposed is that TPE removes circulating inflammatory mediators, which contribute to burn shock, and reduces the need for fluid administration. Two recent studies have analyzed TPE in patients with complicated or failing resuscitation. , Some clinical improvements, such as increased blood pressure and UO, decreased IV fluid volume, and correction of lactate levels, were seen, but there were no differences in mortality compared to an untreated group.
Extracorporeal blood purification aims to remove inflammatory mediators from the circulation. A recent animal study using the Cytosorb hemoadsorptive column demonstrated removal of proinflammatory mediators without a reduction in systemic levels of the same molecules, indicating the limitations posed by CRRT-like extracorporeal blood flow rates and by ongoing cytokine production in the body. By contrast, SeaStar Medical’s Selective Cytopheretic Device (SCD) is a cell-directed extracorporeal therapy that selectively targets the most highly activated neutrophils and monocytes. A clinical trial (NEUTRALIZE-AKI) is examining the SCD in a general acute kidney injury population (see clinicalTrials.gov: NCT05758077).
High-dose vitamin C (ascorbic acid) has been studied as an adjunct to burn shock resuscitation. The proposed mechanism of action is that it serves as a free-radical scavenger, reducing microvascular fluid extravasation. A study by Tanaka et al., using a dose of 66 mg/kg/hour for 24 hours, demonstrated a reduction in 24-hour fluid requirements, less weight gain, decreased edema, fewer ventilator days, lower rates of acute lung injury, and comparable hemodynamic profiles and UO compared to a control group. Other studies have demonstrated similar results. A possible disadvantage of vitamin C is that it can act as an osmotic diuretic, thus compromising the use of UO as an end point. Because of its mechanism of action, vitamin C is probably most beneficial when used early in resuscitation rather than as a rescue intervention for a complicated or failed resuscitation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree