Breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) is an emerging cancer of the immune system that is exclusively associated with textured-surface breast implants. This clinical review provides an update on the diagnosis and management of BIA-ALCL with an emphasis on major advances. The epidemiology and pathophysiology of the disease are also reviewed, focusing on current paradigm shifts and highlighting current controversies related to disease classification and risk mitigation. Finally, the authors conclude by discussing medicolegal and ethical issues surrounding BIA-ALCL while establishing a future basic science and clinical research agenda that is central to improving patient safety.
Key points
- •
Breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) incidence and risk varies widely by surface texturization techniques.
- •
Factors contributing to disease initiation and progression remain under intense study. Recent research has focused on the role of particulate debris and the process of perpetual exhaustive phagocytosis in response to the surface of a textured breast implant.
- •
Complete surgical resection is essential for BIA-ALCL treatment and is now the standard of care for most of cases.
- •
Advanced BIA-ALCL disease is the end of the spectrum of cancer stages and substantiates the World Health Organization classification of BIA-ALCL as a lymphoma.
- •
BIA-ALCL remains an indolent disease with an undercharacterized risk profile, but the consequences of failed detection or surveillance can lead to advanced disease and patient demise.
Background
Breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) is a non-Hodgkin lymphoma subtype that arises around textured-surface breast implants. Since first being described in the mid-to-late 90s, more than 800 cases have been pathologically confirmed worldwide. Cases of BIA-ALCL vary widely by both geography and textured device characteristics, suggesting a complex individual risk profile. , Allergan’s (Dublin, Ireland) Biocell textured implants, which are responsible for greater than 90% of reported cases worldwide when the device history was known, are now subject to a worldwide recall. Despite the removal of these high-risk devices from the global market, other textured devices remain commercially available. Given the emerging nature of the disease and the millions of patients still at risk for developing the disease, heightened awareness and a thorough knowledge of BIA-ALCL is required.
Over the past several years, plastic surgeons, together with oncologists and pathologists, have standardized the current guidelines on diagnosis and treatment—as failure to appropriately identify or manage BIA-ALCL cases can lead to advanced disease and death in rare cases. This evidence-based review aims to provide clinical updates on the epidemiology and pathophysiology of BIA-ALCL while placing a particular emphasis on major advances in diagnosis and treatment. The purpose of this article is to highlight evidence-based surgical and therapeutic strategies for achieving complete remission. Finally, the authors discuss regulatory oversight issues surrounding textured devices and breast implants in general and conclude with establishing future research priorities.
Epidemiology
Since first described, the epidemiology and awareness of BIA-ALCL has evolved considerably. The calculated risk of developing the disease has increased with improved awareness and reporting of cases. Recently, Cordeiro and colleagues estimated a 26-year cumulative incidence of 1:355 patients with an Allergan Biocell implant and a patient-specific incidence rate of 0.311 cases per 1000 person-years (95% confidence interval [CI]: 0.018–0503). They demonstrated an implant-specific risk of BIA-ALCL at 1:602 devices. It remains unclear what the risk of BIA-ALCL is for other commercially available textured devices in the United States. Data from Doren and colleagues provide a risk estimate of Mentor (Mentor Worldwide LLC, Irvine, Calif.) Siltex implants at 1:51,4 28. Calobrace and colleagues estimate a global combined risk of BIA-ALCL for Sientra (Santa Barbara, Calif.) and Silimed (Rio de Janeiro, Brazil.) implants at 1:200,000. In the United States, Sientra has been commercially available for approximately 8 years since 2012, not yet achieving the average time interval for the development of BIA-ALCL. To date, 5 cases of BIA-ALCL with Sientra implants have been reported in the United States. US sales data have not been made available by the company despite request. Based on reported annualized sales revenue compared with total market, Sientra represents less than 5% of the US market. Magnusson and colleagues challenged the Silimed international calculation and found Silimed polyurethane implants to have the highest risk of BIA-ALCL at 1:2832 compared with Allergan Biocell 1:3345 and Mentor Siltex at 1:86,029. From both a methodologic and epidemiologic standpoint, the Calobrace study was not designed to determine the risk of BIA-ALCL. Many epidemiologic studies of BIA-ALCL, including the Doren study, have been limited by inadequate postmarket surveillance. Without the knowledge of global sales data standardized across different populations (which may carry different nonequivalent, unmodifiable risks), actual comparisons cannot be evaluated.
Importantly, the only currently modifiable risk factor identified to date remains surface texturization of the implanted device. Although previously confined to the breast implant pocket, gluteal implant–associated anaplastic large cell lymphoma associated with textured devices have now been reported, providing strong evidence that anatomic location does not play a significant role in tumor development. Moreover, recently described cases in the transgender breast implant population indicate that the disease does not show a predilection for gender. The US Food and Drug Administration (FDA) currently acknowledges a risk of developing ALCL from a textured device at 1:3817 to 1:30,000. However, heterogeneity in the worldwide literature, along with differences in regulatory agency estimates, underscores the significant geographic variation in the reported risk. Although the risk of BIA-ALCL remains relatively low, the increasing number of global cases emphasizes the emerging nature of the disease.
Pathogenesis
This section provides a cursory overview of the pathogenesis of BIA-ALCL relevant to practicing clinicians. An in-depth analysis of the cellular and molecular mechanisms is outside of the scope of this article and has been reviewed in detail elsewhere.
Bacterial Wall Lipopolysaccharide Hypothesis
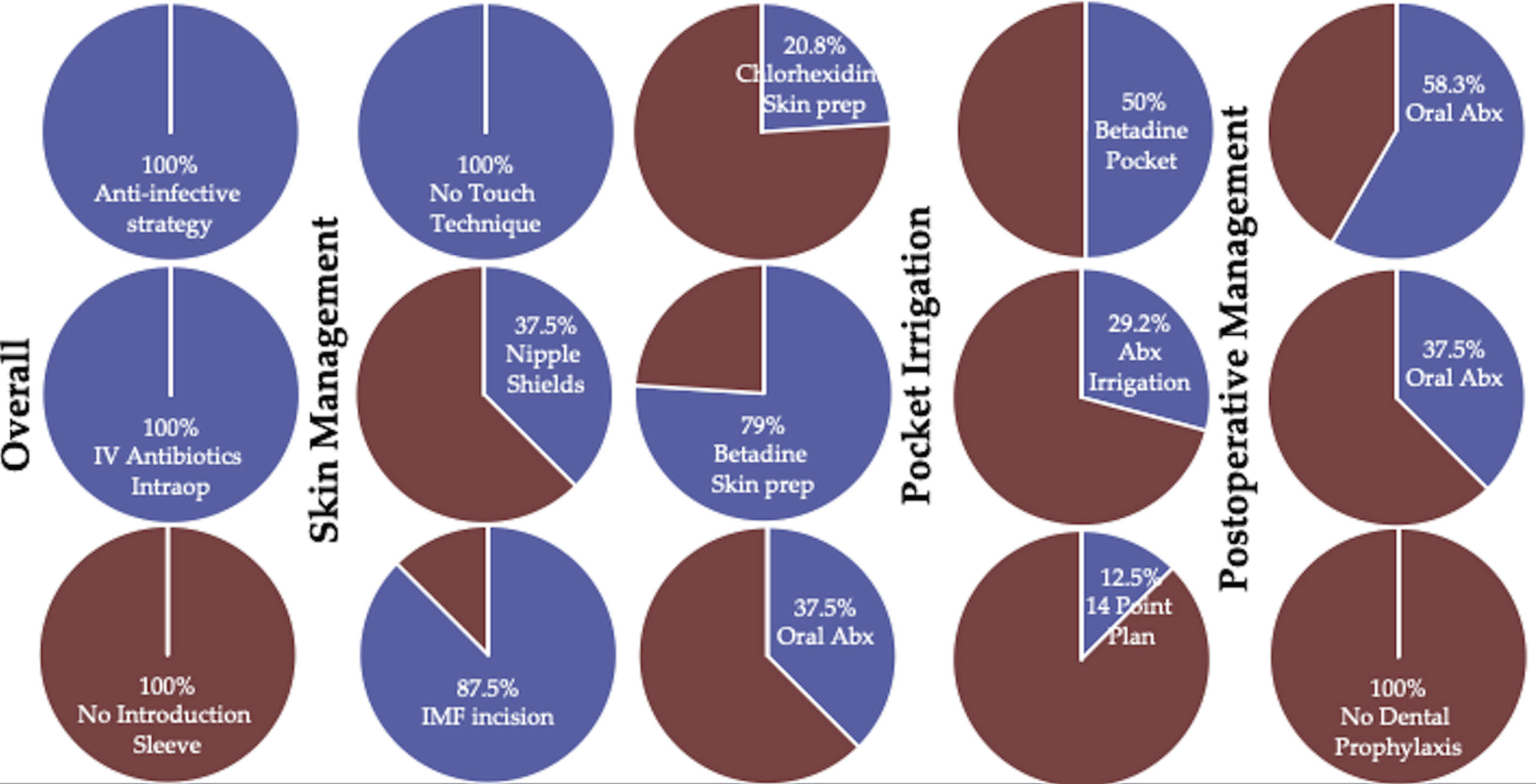
Early investigations into the pathogenesis of BIA-ALCL suggested that subclinical, periprosthetic biofilms drive BIA-ALCL tumorigenesis. Bacteria-induced cancer has medical precedent, as a causal link between some gastric-associated cancers and bacterial invasion ( Helicobacter pylori ) is well established. Loch-Wilkinson and colleagues demonstrated that the risk of BIA-ALCL increases as the surface area increases, a major predictor of bacterial load on implants, with the highest risk of BIA-ALCL residing in implants with the highest surface area. These researchers implicated the gram-negative bacillus Ralstonia pickettii in altering the implant pocket microbiome and causing oncogenic transformation of BIA-ALCL. However, Ralstonia is a commonly identified pathogen in water sources, and the initial studies linking Ralstonia have not been replicated, suggesting this once-promising hypothesis may be inaccurate. In a subsequent study using 16S RNA sequencing, Walker and colleagues demonstrated that the microbiome has no apparent role in BIA-ALCL formation. To that end, the microbiome in the nondiseased breast has yet to be established and is currently the focus of federally funded research. The biofilm hypothesis has since been adapted into a lipopolysaccharide-driven carcinogenesis, which has been shown in oral squamous cell carcinoma and colon cancers. However, a mechanism by which lipopolysaccharide facilitates malignant transformation in even well-described cancers remains incompletely understood. In a prospective study of BIA-ALCL at the senior author’s institution, 24 patients included intraoperative technique details at the time of original breast implant placement. Patients with BIA-ALCL had received betadine irrigation (12 patients: six 50% strength, four 25% strength, 2 “tea-colored”) and 7 patients had received antibiotic irrigation: (5 bacitracin/cefazolin/gentamicin and 2 polymyxin/bacitracin) and still went on to develop disease ( Fig. 1 ). These results demonstrated that BIA-ALCL can still develop in the setting of strict antimicrobial measures. To date, no operative strategy has been shown to decrease the future risk of BIA-ALCL. Around the world, there are approximately 18 BIA-ALCL clusters of 2 or more cases being reported by the same surgeon. However, when the practices are examined, in only 5 of those “clusters” involved the surgeon that originally implanted the device rather than simply having the case secondarily referred to them. Of those clusters, 4 of the surgeons are frequent speakers on proper best implant safety and all have published peer-reviewed articles on BIA-ALCL. These cases represent surgeons from well-established implant practices with strict follow-up, low threshold for CD30 screening of seromas, and surveillance regimens annually for the life time of the patient where possible. Misattributing clusters to “poor breast implant technique” without any supportive data may lead to falsely shaming surgeons and potentially discouraging the reporting of cases.
Cancer Genetics
Recent molecular investigations have provided novel insights into the biological mechanisms responsible for BIA-ALCL lymphomagenesis, but much work remains. Over the last 5 years, high-throughput genetic sequencing technologies have enabled the identification of somatic mutations in DNMT3A and the JAK-STAT3 pathway, as well as germline mutations in TP53. Data from Di Napoli and colleagues have identified the JAK-STAT3 pathway as a key component of disease progression. Unpublished studies from our laboratory mechanistically corroborate these data and suggest JAK-STAT3 as a potential actionable therapeutic target with candidate drugs (eg, JAK inhibitors), which may prove to be beneficial in patients with advanced or surgically unresectable disease. The prevailing hypothesis behind aberrant JAK-STAT3 pathway activation considers an overactivated immune system driving the malignant transformation of capsular lymphocytes. Interestingly, the JAK-STAT3 pathway mechanistically links chronic inflammation and other cancers, including lymphomas. Comprehensive in vitro and ex vivo studies using BIA-ALCL tissues are required to identify the inciting event responsible for driving JAK-STAT activation and is the current focus of our research group.
The Role of Chronic Inflammation and Implant Surface Characteristics
The link between chronic inflammation and cancer has been well established. Our group has provided evidence that a chronically overactivated immune system predisposes to errors in DNA replication and subsequent driver gene mutations (eg, STAT3). , Alternatively, some have postulated that chronic trauma to the breast pocket induces a chronic inflammatory state that cultivates a microenvironment that favors tumorigenesis. However, a low incidence of BIA-ALCL despite millions of patients with textured implants indicates a host-specific immune susceptibility for developing the disease. The foreign body reaction has been an attractive area of research in establishing a chronic inflammatory state, suggesting investigation in host-implant interactions may elucidate pathogenic signaling. Work by Turner has investigated the role of aryl hydrocarbons, a conserved chemical structure found on the textured implant surfaces, to drive cellular proliferation of capsular lymphocytes through their associated receptor. An alternative hypothesis by Kadin and colleagues suggests an allergen-driven cause from a chronic allergic response to the implant itself. However, a unifying hypothesis linking immune responses and carcinogenesis remains elusive and underscores the critical need for comprehensive genetic studies to identify patient-specific risk profiles.
Breast Implant–Associated Anaplastic Large Cell Lymphoma: Lymphoproliferative Disorder Versus Lymphoid Neoplasm
The debate over the classification of BIA-ALCL as a “benign condition,” as opposed to a lymphoid malignancy, has served to limit the initiation of surveillance and definitive treatment. Some investigators have argued that BIA-ALCL is a lymphoproliferative disorder that encompasses a broad spectrum of CD30+ benign seromas, malignant seromas, and distant metastasis. Recently, experts from the World Health Organization (WHO) have provisionally classified BIA-ALCL as a unique lymphoid neoplasm and specifically not a lymphoproliferative disorder. Advanced disease is the end of the spectrum of cancer stages and substantiates the WHO classification of BIA-ALCL as a lymphoma rather than benign or lymphoproliferative. Untreated BIA-ALCL leads to invasive, metastatic disease—and misclassification or overt failure to diagnose this disorder can lead to patient death.
Diagnosis and treatment
Clinical Presentation
BIA-ALCL typically presents as an acute-onset periprosthetic fluid collection greater than 1 year following implantation in approximately 80% of cases ( Fig. 2 ). Patients may also present with lymphadenopathy (4%–12%) or a palpable mass (8%–24%). Less often (<5%), patients may present with capsular contracture or cutaneous involvement, such as a rash. The median time to presentation is 7 to 10 years (range, 1–28 years) following device implantation. BIA-ALCL is equally distributed between cosmetic and reconstructive patients, suggesting that history of a previous malignancy such as breast cancer does not predispose to the subsequent development of the disease.
Diagnostic Workup
The presentation of any delayed seroma should raise immediate clinical suspicion for BIA-ALCL. Importantly, all implants contain a trace amount (5–10 mL) of fluid in the periprosthetic space, which is normal and does not warrant further screening. The National Comprehensive Cancer Network (NCCN) has released guidelines for the diagnosis of BIA-ALCL. First, clinicians should start by obtaining a detailed clinical history and perform a thorough (not just breast) physical examination. Next, the clinician should obtain conclusive imaging (ultrasound or MRI) to characterize the seroma. After ruling out other causes of late seromas (eg, infection), a diagnostic workup should commence with fine-needle aspiration (>50 mL) of the seroma under ultrasound guidance or in consultation with interventional radiology. The aspirate should be sent for cytopathology with the request to rule-out BIA-ALCL. BIA-ALCL rule out order set includes cytology, CD30 immunohistochemistry (IHC), and flow cytometry to determine the presence of a clonal T-cell gene rearrangement.
Specific protocols to guide the pathologic diagnosis of BIA-ALCL have recently been established. , In order to establish a diagnosis of BIA-ALCL, 3 criteria must be met: CD30+ cells by IHC, T-cell clonality, as evidenced by flow cytometry, and the presence of large, anaplastic lymphoma cells on histology. Importantly, CD30+ cells evidenced by IHC alone do not constitute a diagnosis of BIA-ALCL, as benign seromas have been found to harbor CD30+ lymphocytes. The other pathogenic features must also be present for a definitive diagnosis. In order to ensure identification of 95% of randomly distributed lesions in specimens without grossly identifiable lesions, 12 capsular biopsies should be taken, 2 from each side of the face of a cube.
Several attempts have been made to streamline diagnosis to decrease time to definitive management. Hanson and colleagues recently developed a novel, low-cost screening test that can be deployed in the clinical setting. Using a commercially available (R&D Systems, Minneapolis, MN), CD30-specific enzyme-linked immunosorbent assay, they standardized and validated the ability of the assay to reliably detect BIA-ALCL in seroma fluid. The investigators demonstrated that the assay could effectively be used to evaluate suspicious seromas, presenting a reliable and more rapid alternative to standard CD30 immunohistochemistry. Nevertheless, it is important to reinforce that this should only be used as an office-based screening tool. As outlined earlier, a definitive diagnosis still requires further pathologic evaluation.
Oncologic Resection and Adjuvant Therapy
One of the most significant advances in BIA-ALCL treatment has been the extensive examination of the role of surgical intervention by Clemens and colleagues. , When diagnosed and treated in accordance with NCCN guidelines, BIA-ALCL carries an excellent prognosis, with 5-year overall and event-free survival rates approaching 91% and 46%, respectively. With complete surgical excision, event rates are reduced to 0% (stages T1 and T2) and 14.3% for stage T3. A TNM staging system has since been proposed and validated, which replaces the previously used Ann Arbor Lugano Classification for BIA-ALCL. The MD Anderson TNM staging system of BIA-ALCL is summarized in Table 1 . Collectively, these data demonstrate the clinical superiority of complete surgical resection over adjuvant therapy. This is reflected in NCCN guidelines, which highlight complete surgical resection as the standard of care, whereas adjuvant therapy is reserved for MD Anderson stages IIB to IV.