Definition
Blepharoptosis is defined as a lower than normal position of the upper eyelid in relation to the globe and pupil. The term is often abbreviated to ptosis.
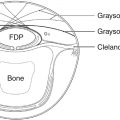
The upper eyelid normally covers the upper 1–2 mm of the cornea, but this tends to increase slightly in old age. As the cornea has a diameter of 11 mm, the distance from the upper eyelid margin to the inferior corneoscleral limbus is therefore normally 9–10 mm. This distance can be used as a means of measuring the amount of ptosis but, conventionally, the distance from the upper eyelid margin to a reflection of a point source of light shone directly at the patient is used. This distance is called the Margin Reflex Distance 1 or MRD1. This is normally 3–4 mm. (The MRD2 is the distance from the light reflex to the lower lid margin.) The disadvantage of using the MRD1 over a simple measure from the upper lid margin to the inferior limbus is that when the upper eyelid margin is lower than the position of the light reflex on the cornea, the lid must be lifted up to then get an indirect measure of how far below the light reflex the lid margin sits. If it is lower than the light reflex it is expressed as a negative value in millimeters.
Anatomy
Knowledge of the anatomy relevant to ptosis is vital in understanding the etiology and mechanisms of ptosis, and the clinical signs and surgery of ptosis.
The main surface anatomy marking of relevance is the upper eyelid crease or eyelid fold. In Caucasians this lies about 6–10 mm above the eyelash line. In Asian eyelids there is much more variability, with the crease being absent or often much lower than in Caucasian eyelids, with an associated fullness of the upper eyelid. This is because the orbital septum attaches much lower in most Asian eyelids with the preaponeurotic fat pad extending more inferiorly giving the eyelid a fuller appearance.
The upper eyelid retractor, the levator palpebrae superioris (LPS), is innervated by the third cranial nerve (oculomotor nerve). The muscle belly arises from the apex of the orbit on the lesser wing of the sphenoid, just above the optic canal. It travels anteriorly in the orbit below the orbital roof, partly overlapping the superior rectus muscle, which lies immediately below it. As it reaches the anterior orbit, it turns more inferiorly towards the tarsal plate of the eyelid. About 8–10 mm above the superior border of the tarsus, it broadens out into a tendinous aponeurosis. This aponeurosis attaches onto the anterior surface of the tarsal plate, and also sends fibers to attach between bundles of the orbicularis oculi muscle just under the eyelid skin to form the upper eyelid skin crease ( Fig. 69.1 ).
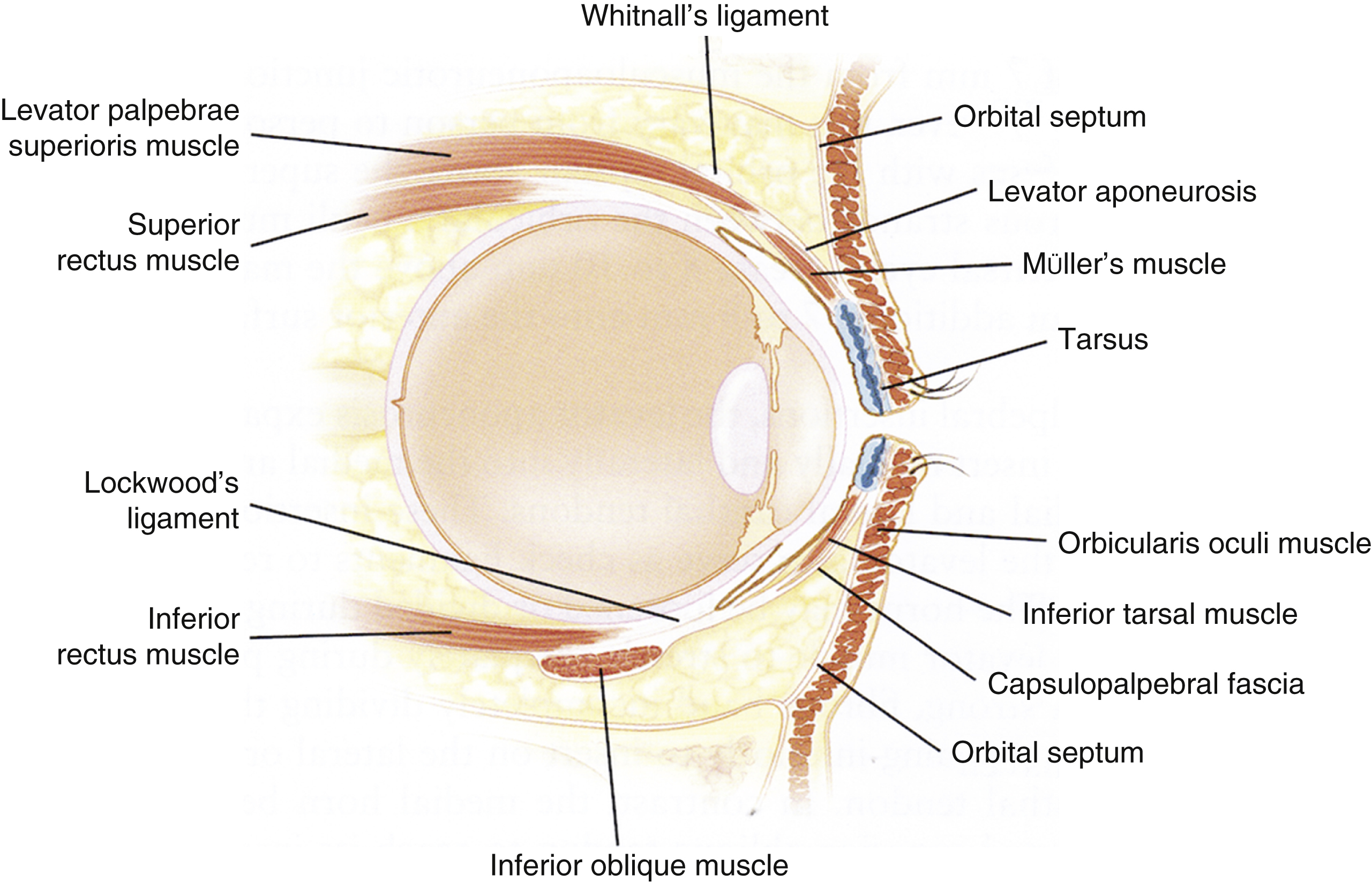
The levator aponeurosis expands medially and laterally as its medial and lateral horns. The lateral horn is much stronger and attaches on to Whitnall’s tubercle just inside the lateral orbital rim. The lateral horn also partially divides the lacrimal gland into orbital and palpebral lobes, with the two lobes continuous behind the posterior edge of the lateral horn. The medial horn is less well defined and becomes weaker with age. It attaches to the superior border of the medial canthal tendon.
There is a smooth muscle component of the levator apparatus, called Müller’s muscle, which is innervated by sympathetic nerve fibers. This is a sheet of smooth muscle fibers with a rich blood supply that arises from the undersurface of the levator muscle and extends downwards under the levator aponeurosis to attach to the superior border of the tarsal plate. It is about 10 mm in length.
There is a strong transverse band of connective tissue running from the region of the trochlea to the lacrimal gland, which runs over the superior surface of the levator muscle near the point where it turns more inferiorly towards the tarsal plate. This is called the superior transverse ligament or Whitnall’s ligament. There is a less well-defined portion of this structure which passes under the levator muscle, called the transverse superior fascial expansion.
The antagonist muscle of the LPS is the orbicularis oculi muscle, innervated by branches of the facial nerve. It runs circumferentially around the eyelids, divided into parts called pretarsal, preseptal and orbital. The orbicularis oculi muscle lies immediately below the skin of the upper eyelid, which is extremely thin.
The orbital septum in the upper eyelid extends from the superior orbital rim downwards to attach to the anterior surface of the levator aponeurosis. Behind the orbital septum and lying over the levator aponeuroris and anterior portion of the levator muscle belly is the preaponeurotic fat pad, a vital landmark in levator aponeurosis and muscle surgery. Laterally, in the same retroseptal space, the orbital lobe of the lacrimal gland is located. Medially, there is a medial upper fat pad. The tendon of the superior oblique muscle partially separates the central preaponeurotic and medial fat pads as it extends down to attach to the globe medial to the medial edge of the levator muscle belly.
The tarsal plate of the upper eyelid is a semilunar-shaped semi-rigid structure made up of meibomian glands set in a fibrous stroma with the conjunctiva firmly adherent to its undersurface. It attaches via the upper limbs of the medial and lateral canthal tendons to Whitnall’s tubercle laterally and the frontal process of the maxilla just anterior to the anterior lacrimal crest (anterior limb of the medial canthal tendon) and to the posterior lacrimal crest (posterior limb). The upper tarsal plate is about 9–10 mm in vertical height centrally and about 1 mm thick.
There is a vascular arcade overlying Müller’s muscle at its lower edge, and under the levator aponeurosis, just above the superior border of the tarsal plate. A smaller vascular arcade runs just above the lash bulbs near the margin of the eyelid, within fibers of orbicularis oculi.
Etiological Factors
When considering the causes of blepharoptosis, it is conventional to divide them into congenital and acquired. However, there is some overlap in these two groups.
The following classification is useful:
- A.
Congenital Ptosis
- 1.
Congenital neurogenic ptosis
- 1a.
Congenital third nerve palsy
- 1b.
Congenital Horner’s syndrome
- 1a.
- 2.
Congenital myogenic ptosis (sometimes called congenital dystrophic ptosis)
- 2a.
Isolated congenital dystrophic ptosis
- 2b.
Congenital dystrophic ptosis with associated weak globe elevator(s)
- 2c.
Congenital dystrophic ptosis as part of a syndrome
- i.
Blepharophimosis syndrome
- ii.
Other syndromes (e.g., Noonan’s, Saethre Chotzen)
- i.
- 2a.
- 3.
Congenital ptosis with synkinesis (jaw-winking ptosis, others)
- 4.
Congenital aponeurotic ptosis (rare)
- 1.
- B.
Acquired Ptosis
- 1.
Neurogenic ptosis
- 1a.
Third nerve palsy
- 1b.
Horner’s syndrome
- 1a.
- 2.
Myogenic ptosis
- 2a.
Myopathic ptosis
- i.
Mitochondrial cytopathies (chronic progressive external ophthalmoplegia (CPEO) and its variants)
- ii.
Myotonic dystrophy
- iii.
Oculopharyngeal muscular dystrophy
- iv.
Other myopathies
- i.
- 2b.
Myasthenia
- i.
Ocular myasthenia
- ii.
Myasthenia gravis
- i.
- 2a.
- 3.
Involutional ptosis
- 3a.
Involutional aponeurotic ptosis
- 3b.
Involutional myogenic ptosis
- 3a.
- 4.
Mechanical ptosis (lumps and swellings of the eyelid)
- 5.
Traumatic ptosis (may be multiple factors)
- 1.
- C.
Pseudoptosis
- 1.
Globe hypotropia
- 2.
Enophthalmos, or a small eye
- 3.
Blepharospasm
- 4.
Dermatochalasis.
- 1.
Congenital and acquired neurogenic ptosis can be considered together in terms of investigation and management. Third nerve palsies may be partial or complete, and there may be aberrant regeneration complicating the clinical picture. Congenital aponeurotic ptosis is rare, but can also be managed in the same way as acquired aponeurotic ptosis.
Congenital myogenic or dystrophic ptosis may be unilateral or bilateral (symmetric or asymmetric) and is due to a maldevelopment of the levator muscle, which is variably replaced by fatty and fibrous tissue. The muscle contracts and relaxes less than normally. This type of ptosis often occurs with a positive family history and there are a number of genetic defects that continue to be identified. Whilst it is usually an isolated abnormality, there are well-recognized associations, including poor functioning of the superior rectus muscle and double elevator palsy. Children with these additional muscle problems will usually have a vertical strabismus with the globe pointing downwards (hypotropia).
Congenital myogenic ptosis may be part of a more extensive syndrome. The best known of these is blepharophimosis or BPES (blepharophimosis, ptosis, epicanthus inversus syndrome). Numerous other less common syndromes may have ptosis as one manifestation of the condition and Noonan’s and Saethre Chotzen syndrome are two examples.
Congenital ptosis with synkinesis is a special subcategory of congenital ptosis. The best recognized form is Marcus Gunn jaw-winking ptosis. The child will have unilateral ptosis and with contraction of the ipsilateral lateral pterygoid muscle, the levator muscle is stimulated and the lid rises, sometimes higher than its normal position. Synkinesis with other muscles is less common but well described. These include lid elevation with contraction of the medial pterygoid, digastric, the lateral rectus muscle, and with swallowing.
Acquired myogenic ptosis is an important diagnosis to make because of the significant general health aspects of these conditions.
Involutional or age-related ptosis is by far the commonest form of ptosis seen in clinical practice. A mild degree of ptosis is common in the elderly, but more advanced degrees may occur and impact on visual function. Traditionally, the majority of involutional ptosis has been attributed to dehiscence and/or stretching of the levator aponeurosis, but in a significant proportion of older patients with acquired ptosis, the levator muscle itself appears to be abnormal with infiltration by fatty tissue into the muscle belly.
Some lumps on the upper eyelid may induce ptosis by their weight alone, and this is the classic cause of so-called mechanical ptosis. Other mechanical factors may be at play in some cases of ptosis, including scarring or adhesion between parts of the muscle and other structures such as the orbital roof after fractures, or adhesions between the eyelid and the globe.
Traumatic ptosis is often multifactorial. Traumatic nerve injuries may lead to ptosis, as may injuries to the muscle belly or aponeurosis. Some patients will develop ptosis after episodes of eyelid bruising or swelling and it is thought that such swelling may lead to dehiscence of the levator aponeurosis. Other forms of ocular surgery may sometimes lead to ptosis. The mechanism is not always clear.
Repeated minor trauma may also lead to ptosis. This is thought to be the cause in patients who develop ptosis after many years of wearing hard or rigid gas permeable contact lenses, who often remove the lenses by applying traction at the lateral canthus to remove the lens.
Pseudoptosis has a number of causes. An important one to recognize is hypotropia. With the globe looking down compared to its fellow eye, the lid will also be lower than normal because the lid follows the eye downwards. If the normal eye is covered and the patient takes up fixation with the hypotropic eye, the lid will usually rise at least partially as well. This uncovers the element of the ptosis which is due to “pseudoptosis.” Ideally, the hypotropia should be corrected first to eliminate any element of pseudoptosis.
A small or enophthalmic eye will often have a narrower than normal palpebral fissure, due to inadequate posterior support of the eyelid, but this is not due to any abnormality of the levator apparatus or eyelid and is classified as a form of pseudoptosis.
Patients with increased tone in the orbicularis oculi muscle will often have a narrowed palpebral fissure also. This may be due to increased tone with or without an element of aberrant reinnervation after a facial palsy. The lid fissure may narrow further when muscles in the lower face contract. Similarly patients with hemifacial spasm will display episodic narrowing of the palpebral fissure associated with spasm of other facial muscles on the same side.
Finally, marked dermatochalasis will often look like true ptosis but if the hood of abnormal skin is lifted out of the way, the lid position can then be properly assessed. Many cases of true involutional ptosis will of course also have a degree of dermatochalasis.
Preoperative Assessment
As in any medical condition, a proper history and examination is essential. The history will usually easily separate congenital from acquired ptosis. Questions should be asked about time and rapidity of onset and variability of the ptosis, associated diplopia, other neuromuscular problems, any history of trauma, lid swelling or contact lens wear. A family history of ptosis or muscle problems should be sought. Older patients should be asked about a history of dry eye problems as elevating the eyelid in a patient with dry eyes may exacerbate the dry eye problem.
A standard sequence of examination steps will usually give an answer, in addition to the details of the history, as to what has caused the ptosis, and make surgical planning safer. The examination steps may seem arduous, but with experience, a targeted ptosis examination can be performed in a few minutes. The steps should include the following:
- 1.
Measure and record the patient’s vision. This is especially important in children in the amblyogenic age group (less than 10 years of age). There is a strong association between ptosis and amblyopia.
- 2.
Measure the degree of ptosis. This can be done by measuring the MRD1 (upper lid to corneal light reflex), or the distance from upper lid to lower limbus (normal is 9–10 mm). The degree of ptosis should be measured with the brow held in a neutral position to eliminate any elevation of the lid by the brow.
- 3.
Measure the levator excursion or “levator function.” This is the distance the lid moves between extreme up- and downgaze. Normal should be about 13–15 mm.
- 4.
Look at the position of the ptotic lid on downgaze. In congenital myogenic ptosis, because the levator relaxes less well, the lid does not drop as much as normal on downgaze. With involutional aponeurotic ptosis, the lid will drop more than normal on downgaze.
- 5.
Measure the position of the eyelid crease. This helps in diagnosis and surgical planning. The crease is usually elevated compared to the normal side in aponeurotic ptosis. The crease may be absent in more severe congenital ptosis.
- 6.
Examine the patient’s eye movements. This is a vital part of the examination. Patients with myogenic ptosis will usually have signs of abnormal eye movements, which may be subtle at the first presentation. The range of eye movements, rapidity of eye movements (tested by examining saccadic eye movement), and presence or absence of strabismus in any position of gaze by performing a cover test, should all be performed.
- 7.
Examine the pupils. The pupil will be enlarged in many third nerve palsies, and in Horner’s syndrome the pupil will be smaller on the affected side. This difference is more obvious in dim light where the normal side dilates more.
- 8.
Examine eyelid closure. Gentle eyelid closure should be tested first, looking for lagophthalmos, indicating orbicularis weakness. Forced closure should be tested also, looking for weakness.
- 9.
Examine for Bell’s phenomenon. This is where the eye rolls upwards on forced lid closure. If absent, the risk of corneal exposure problems is increased with ptosis surgery.
- 10.
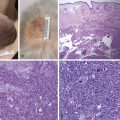
Evert the upper eyelid and examine the conjunctival fornices. The tarsal surface of the lid may be abnormal after previous ptosis surgery, and the conjunctival fornices may be hiding rare disorders such as subconjunctival lymphoma (seen as a salmon patch) or conjunctival amyloidosis.
- 11.
Look for associated clinical signs. These include brow position (the frontalis muscle on the side of a unilateral ptosis will be usually overacting, elevating the brow), the presence of synkinetic movements (ask the patient to open the mouth and move the jaw side-to-side), and evidence of other features of myopathies or myasthenia (frontal balding in myotonic dystrophy, muscle weakness elsewhere, fatigability with myasthenia on sustained upgaze, a twitch of the upper eyelid on upward saccades of the eyes, known as Cogan’s lid twitch, and occurring with myasthenia).
- 12.
Check that corneal sensation is normal. This can be done with a strand of cotton teased from the end of a cotton tip. Reduced corneal sensation is rarely seen in ptosis patients, but if present, it markedly increases the risk of corneal complications after ptosis surgery.
- 13.
Examine the ocular surface. This is best done with a slit-lamp after placing a drop of fluorescein in the eye. Alternatively, a direct ophthalmoscope with a cobalt blue filter can be used. The adequacy of the tear film should be assessed as well as the presence of corneal drying. A Schirmer’s test can be helpful. If the tear film is deficient or the cornea stains with fluorescein, there is a significant risk of worsening corneal drying or even ulceration with ptosis surgery.
At the end of the history and examination, a confident diagnosis of the cause of the ptosis can be made in most patients. If there is doubt, or certain diagnoses are suspected, then further investigations or referral to other specialists may be appropriate. The following scenarios deserve further investigation or referral:
- 1.
Acquired 3rd nerve palsy. Should be referred for neurological assessment and imaging.
- 2.
Acquired Horner’s syndrome. Neurological referral.
- 3.
Suspected myasthenia. An ice-test is a simple investigation that can help establish a diagnosis of myasthenia. An ice pack is placed over the eye with ptosis for 3–5 minutes. In myasthenia, the lid should elevate by at least 2 mm, and in patients with diplopia, this may temporarily resolve or reduce. All suspected myasthenics should be referred for neurological assessment and investigation.
- 4.
Suspected myopathic ptosis. Neurological referral. Most myasthenics are managed medically. Occasionally, myasthenic ptosis may resist medical therapy and in some cases, surgery can be cautiously offered.
- 5.
Reduced vision in a child under 10. Should be referred to an ophthalmologist.
- 6.
Presence of strabismus. Referral to an ophthalmologist.
There may be other scenarios requiring further investigation or referral and if in doubt about the cause of the ptosis, then assessment by an ocular plastics specialist ophthalmologist or neuro-ophthalmologist is appropriate.
Treatment Principles
The principles of surgical and other forms of management of ptosis can be divided into the following groups:
- 1.
Congenital ptosis
- 2.
Blepharophimosis syndrome
- 3.
Jaw-winking ptosis
- 4.
Myopathic ptosis
- 5.
Involutional ptosis
Congenital Ptosis
The management of congenital ptosis can be divided into several steps.
- 1.
Refer all congenital ptosis for ophthalmological assessment at first diagnosis. Ideally this should be as soon as the diagnosis is made, shortly after birth. The ophthalmologist will assess the infant for refractive error and amblyopia and initiate appropriate treatment. If the ptosis is severe and the vision is not developing normally despite amblyopia therapy (glasses, patching or atropine penalization with drops), then early surgery may be indicated. Otherwise, the surgery can be delayed until accurate measurements of the degree of ptosis and levator function can be made, allowing proper surgical planning.
- 2.
If there is a squint present, then the patient should also be referred as early as possible to an ophthalmologist. The commonest squint present with unilateral congenital ptosis is a hypotropia. If present, the hypotropia should be corrected before any ptosis surgery, because there will often be an element of pseudoptosis present which will be reduced by correcting the hypotropia. Additionally, correcting the hypotropia will reduce the risk of corneal exposure problems by improving globe elevation and the Bell’s phenomenon.
- 3.
If the vision is developing normally and there is no strabismus, then ptosis surgery can be planned around the age of 3–5 years, when good measurements can be obtained that help in the planning of ptosis surgery.
Surgery for Congenital Ptosis
If early intervention is required because of the presence of amblyopia, then normally a brow suspension is performed using non-autogenous material such as silicone. This allows for later removal and more definitive ptosis surgery at a later age if required. The technique for brow suspension is discussed later.
If the ptosis surgery is performed electively at or after the age of about 4 years, when good and reproducible measurements of the degree of ptosis and levator function can be obtained, then the choice of procedure will be determined principally by the degree of ptosis and levator function. If the levator function is poor (5–6 mm or less), then performing a levator resection or shortening procedure will lead inevitably to undercorrection. In this setting a brow suspension is preferred.
For congenital ptosis with 7 mm or more of levator function, a levator shortening or resection is preferred. The amount of resection is determined by the degree of ptosis and levator function. Complex tables are published, and the best known of these is that of Beard. In general, levator function can be divided into poor, moderate, and good. “Good” levator function in congenital ptosis is 8 mm or more. Moderate is 6–8 mm. The range of shortening or resection for congenital ptosis is of the order of 10–25 mm. For genuine congenital ptosis with a myogenic cause, a resection of less than 10 mm is likely to lead to undercorrection. Within the group of patients with moderate levator function, the amount of resection will depend on the degree of ptosis ( Table 69.1 ). An additional useful guide for the amount to resect the levator is Berke’s rule of 7s. Under general anesthesia, if the levator function is 7 mm then the lid should be set to the desired level (usually 2 mm below the superior limbus). If more than 7 mm, it is set lower, and of less than 7 mm, it is set higher.