Abstract
All students of dermatology need a basic foundation and framework upon which to accumulate knowledge. In this chapter, the basic tenets of disease classification in dermatology are introduced. This includes division of disease processes into basic etiologic origins, most commonly inflammatory diseases versus neoplasms, with further subdivision of the former into infectious versus non-infectious. Further subcategorizations eventually result in an appropriate differential diagnosis. Descriptive terms are also introduced which represent the lexicon of dermatology and serve as the building blocks of a specialty-specific language. The principles of morphology, configuration, and distribution are stressed as is the utility of these concepts in the generation of a logical differential diagnosis. The importance of histopathologic examination of diseased skin, especially when an appropriate and representative biopsy specimen is obtained, is emphasized, as is clinicopathologic correlation. However, the latter may require both special stains and immunohistochemical stains. Advanced clinical examination techniques, in particular dermoscopy, are also outlined. In sum, this introductory chapter foreshadows a more detailed discussion of the myriad aspects of the clinical practice of dermatology and dermatopathology that follow in the remainder of the tome. In this regard, metaphorically, the chapter represents footings, placed into bedrock and designed to secure the “dermatologic skyscraper” that the remainder of the text represents.
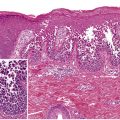
Dermatopathology combines two separate, although intimately related disciplines, clinical dermatology and general pathology. Both of these fields share the same root, i.e., morphology. The secret for learning dermatopathology is to adapt the same skill sets that enable you to recognize primary and secondary skin lesions clinically and apply them to the microscopic slide. The chapter starts with the basic principles of performing a skin biopsy, including proper selection of a clinical lesion, biopsy techniques and handling of specimens, emphasizing the prerequisites for maximizing the results of the procedure. It then describes an algorithmic approach to pattern recognition for the histopathologic diagnosis of inflammatory skin diseases. Ancillary techniques that may help in the pathologic diagnosis of skin diseases, particularly immunohistochemistry, are also discussed.
Keywords
morphology, distribution, configuration, skin color, clinicopathologic correlation, temporal course, dermatopathology, dermoscopy, dermatoscopy, skin biopsy, special stains, immunohistochemical stains, clinicopathologic correlation, dermatology lexicon, skin biopsy, pattern analysis, immunohistochemistry, special stains, inflammatory diseases, invisible dermatoses, clinicopathologic correlation
Introduction to Clinical Dermatology
The skin represents the largest organ of the human body. The average adult has 1.75 m 2 (18.5 ft 2 ) of skin that contains a variety of complex adnexal structures, including hair follicles, nails, glands and specialized sensory structures, all of which function in protection, homeostasis, and the transmission of sensation. Dermatology is the field of medicine that deals with the macroscopic study of skin, adjacent mucosa (oral and genital) and cutaneous adnexa, while dermatopathology deals with the microscopic study of the same structures. The two fields are closely allied, as they are complementary and requisite to one another.
Multiple studies have shown that a dermatologist is the most effective diagnostician with regard to skin disease . This enhanced acumen reflects experience in recognizing distribution patterns and configurations as well as subtle variations in morphology and colors, in addition to appreciating associated histopathologic findings. This chapter will not only serve as an introduction to the classification schemes, descriptive terminologies and diagnostic tools utilized in dermatology, it will also highlight additional means for studying the skin, including dermoscopy (dermatoscopy) and dermatopathology, with clinicopathologic correlation between macroscopic and microscopic findings.
Etiologic Premises
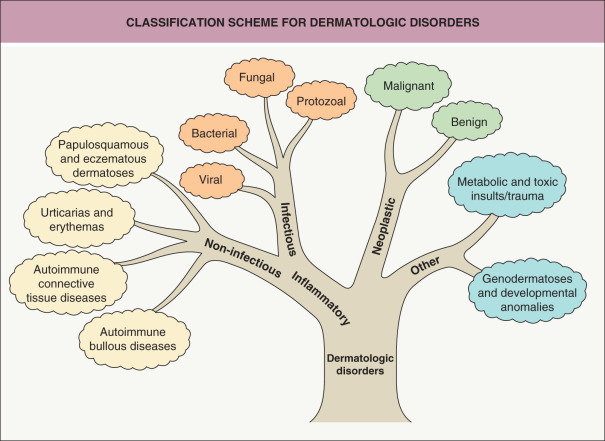
All students of dermatology, whether beginners or advanced scholars, require a basic conceptual framework upon which to organize thousands of skin diseases. A useful arrangement is one that is analogous to a tree, with a trunk, major branches, minor branches, twigs and, ultimately, leaves ( Fig. 0.1 ). Instead of memorizing thousands of leaves, a logical, progressive movement along the limbs will allow for a more complete and sophisticated differential diagnosis.
Inflammatory versus neoplastic
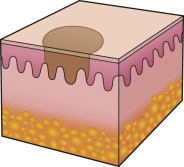
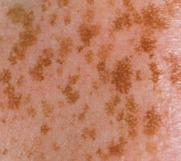
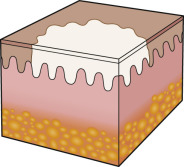
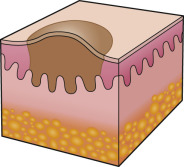
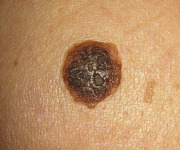
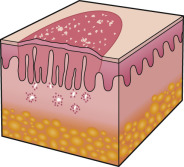
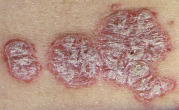
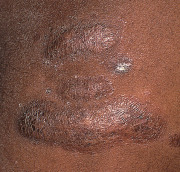
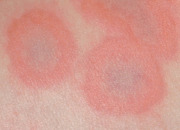
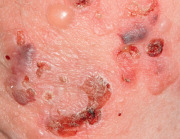
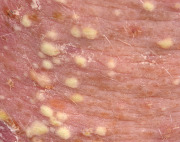
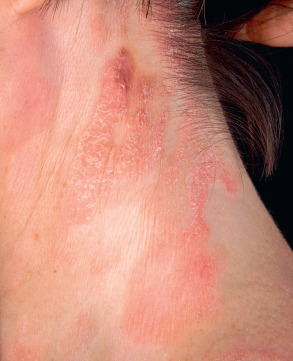
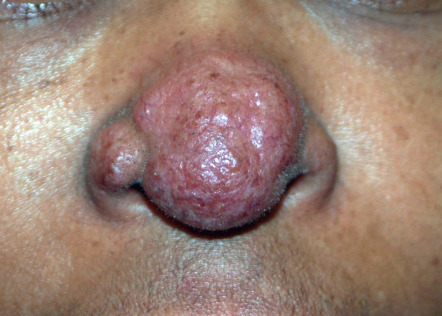
An early and major “branch point” in classifying skin diseases is deciding simply if a skin condition is “neoplastic” (either benign or malignant) or “inflammatory” (either infectious or non-infectious) (see Fig. 0.1 ). However, an experienced clinician knows that one must consider possible diagnoses along multiple limbs before narrowing the differential diagnosis, because both overlap and mimicry can occur. For example, mycosis fungoides, the most common form of cutaneous T-cell lymphoma, is a clonal lymphoproliferative disorder (a “neoplasm”), yet its clinical presentation resembles an inflammatory disorder ( Fig. 0.2 ), especially in its early stages. Conversely, sarcoidosis is an inflammatory condition, but it may present as an isolated infiltrated plaque or nodule that may mimic a neoplasm ( Fig. 0.3 ).
Morphology
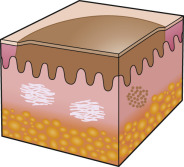
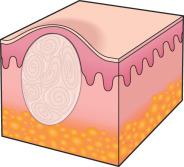
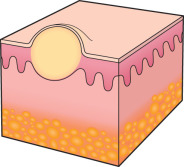
To an engineer or material scientist, the word “ morphology ” refers to the structure and appearance of a material without regard to function. In dermatology, this term is used analogously to refer to the general appearance of a skin lesion or lesions, irrespective of the etiology or underlying pathophysiology. For example, a small cutaneous blister is referred to as a “vesicle”, regardless of whether it is due to an infectious process, such as herpes zoster, or an autoimmune process, such as bullous pemphigoid ( Fig. 0.4 ). Therefore, the proper use of morphologic terms establishes a structural framework for grouping skin diseases based upon their macroscopic appearance .
In essence, morphologic terms become a “native language” by which dermatologists, and other health professionals, communicate with each other to describe skin lesions. As such, they are key elements of a lexicon. Without a basic working knowledge of morphology, it is impossible to describe cutaneous observations in a consistent manner. Therefore, one of the initial steps in studying dermatology is to learn basic morphologic definitions inherent to the specialty.
There exist both primary morphologic terms ( Table 0.1 ), which refer to the most characteristic, representative or native appearance of skin lesions (e.g. a “papule”), as well as secondary morphologic terms ( Table 0.2 ), which can augment or even supplant primary morphologic terms. Secondary morphologic terms often reflect the effects of exogenous factors or temporal changes (e.g. “scales”, “crusts”) that evolve during the course of a skin disease.
| SECONDARY FEATURES – MORPHOLOGICAL TERMS | |||
|---|---|---|---|
| Feature | Description | Disorders | |
| Crust |
| 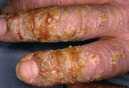 Secondarily infected hand dermatitis |
|
| Scale |
| 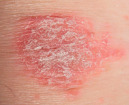 Psoriasis |
|
| Fissure |
|  Hand dermatitis |
|
| Excoriation |
|  Neurotic excoriations |
|
| Erosion |
| 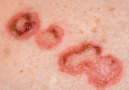 Pemphigus foliaceus |
|
| Ulcer |
| 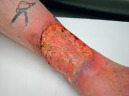 Pyoderma gangrenosum |
|
| Infarct |
|  Antiphospholipid syndrome |
|
| Atrophy |
| 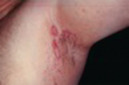 Striae secondary to potent topical corticosteroids |
|
| Lichenification |
| 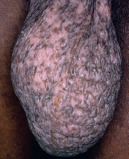 Lichen simplex chronicus |
|
Secondary changes must be considered when performing, or examining histologically, a biopsy of a skin lesion. An astute clinician will generally attempt to biopsy a well-developed but “fresh” lesion that demonstrates the expected primary pathology, free of secondary changes such as erosions, excoriations, and lichenification. This allows the dermatopathologist to evaluate the histologic features of the lesions in their native state, without potentially confounding alterations.
Lastly, the skin is a three-dimensional structure, and like the cartographers who construct maps, there are certain descriptors used by dermatologists to describe the topography of individual skin lesions. Examples include flat-topped (lichenoid), dome-shaped, verrucous, umbilicated, filiform, and pedunculated .
Palpation and appreciation of textural changes
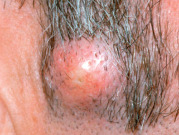
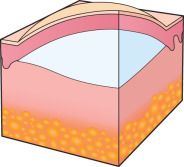
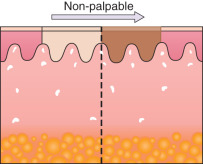
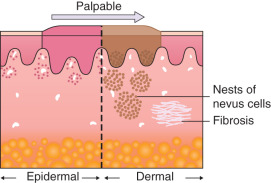
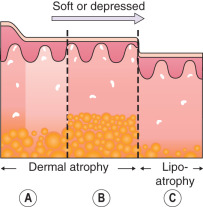
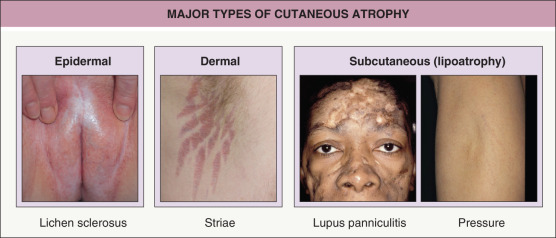
Any discussion of morphology must include textural change, and palpating a lesion often provides important diagnostic clues. In dermatology, palpation can prove useful in several ways. Firstly, it helps in making a distinction amongst primary morphologies (see Table 0.1 ). For example, the key difference between macules and papules, or patches versus plaques, is that macules and patches are flush with the surrounding skin and cannot be appreciated by palpation. On the other hand, papules and plaques, by definition, must be palpable ( Table 0.3 ). Secondly, palpation may augment the examination and appreciation of a disease process for which visual changes are absent, unimpressive, or nonspecific. For example, in morphea, an autoimmune connective tissue disease that leads to sclerotic collagen within the dermis, the skin feels indurated (very firm) while only nonspecific hyperpigmentation may be evident with visual inspection. The same is true for other fibrotic disease processes, such as nephrogenic systemic fibrosis and systemic sclerosis. Likewise, atrophy, be it epidermal, dermal or subcutaneous, also serves as a diagnostic clue ( Fig. 0.5 ). Lastly, purpura is often classified as palpable or non-palpable, and this division implies different underlying etiologies (e.g. small vessel vasculitis aligned more with palpable purpura than macular purpura). Examples of useful distinctions that can be gleaned via palpation are outlined in Table 0.4 .
| PALPATION OF CUTANEOUS LESIONS |
|
Color
The color of skin lesions can provide important clues as to the nature of the disease process. Sometimes our perception of color may be modified by palpation (see Table 0.4 ). For example, while many dermatological processes appear red–purple in color, it is important to ascertain whether this is a blanchable erythema (i.e. it disappears with pressure), which suggests the color is due to vasodilation, or whether it is due to extravasation of red blood cells into the tissue (purpura), which does not blanch. Also, it is not uncommon for exogenous sources of pigment, such as topical medicaments, oral drugs and other ingestants, to be implicated in producing discoloration of the skin. Table 0.5 lists the more frequently observed colors of skin lesions and examples of associated disorders.
| COLOR AS A CLUE TO THE CLINICAL DIAGNOSIS | |
|---|---|
| Color | Examples of diseases with this color |
Erythema (pink to red–brown, depending upon the skin phototype) 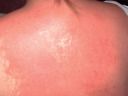 Morbilliform (exanthematous) drug eruption |
|
Black 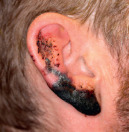 Necrosis secondary to vasculopathy from levamisole-contaminated cocaine |
|
| Blue (ceruloderma)  Dermal melanocytosis |
|
Brown 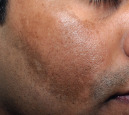 Melasma |
|
Gray  Argyria |
|
| Purple (violaceous) 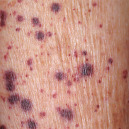 Palpable purpura of cutaneous small vessel vasculitis |
|
White 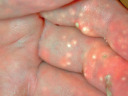 Calcinosis cutis (systemic sclerosis) |
|
Green 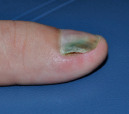 Onycholysis with secondary Pseudomonas infection |
|
Orange–red (salmon) 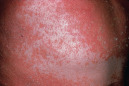 Pityriasis rubra pilaris with islands of sparing |
|
Yellow 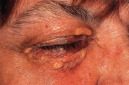 Xanthelasma |
|
Variation in skin color within the human population
Many racial and ethnic descriptors are used in common parlance, including African, African-American, Asian, Middle Easterner, Northern European, Southern European, Native American, Pacific Islander and Hispanic, to describe individuals with similar cutaneous characteristics as well as heritage. Yet even within racial and ethnic groups, gradations exist with regard to skin pigmentation. Sometimes the term “skin of color” is used to describe all skin tones darker than those of white (Caucasian) skin . However, this term encompasses more than skin color and its response to ultraviolet irradiation, as is assessed by the Fitzpatrick Scale (skin phototypes I–VI; Table 0.6 ). It also refers to other shared characteristics, such as hair color, hair texture, and a tendency toward certain reaction patterns in the skin as a response to an insult. The practice of dermatology requires a solid understanding of the differences in clinical features (e.g. hues of red) amongst individuals with different levels of skin pigmentation.
| FITZPATRICK SCALE OF SKIN PHOTOTYPES | ||
|---|---|---|
| Skin phototype | Skin color | Response to UV irradiation |
| I | White | Always burns, does not tan |
| II | White | Burns easily, tans with difficulty |
| III | Beige | Mild burns, tans gradually |
| IV | Brown | Rarely burns, tans easily |
| V | Dark brown | Very rarely burns, tans very easily |
| VI | Black | Never burns, tans very easily |
Variations in skin color are due to differences in the amount and distribution of melanin within epidermal melanocytes and keratinocytes , rather than the number of melanocytes (see Ch. 65 ). In addition, the ratio of eumelanin (brown–black) to pheomelanin (yellow–red) influences skin color, with pheomelanin the predominant pigment in those with freckles and red hair. Exposure to ultraviolet radiation also significantly impacts melanin production (tanning).
Pigmentation of the skin clearly influences the prevalence of certain cutaneous findings and disorders. For example, individuals with darkly pigmented skin are more likely to develop multiple streaks of longitudinal melanonychia (see Ch. 71 ) , pigmentation of the oral mucosa , persistent postinflammatory hyperpigmentation (see Ch. 67 ), and obvious pigmentary demarcation lines (Futcher lines or Voigt lines; see Fig. 67.12 ). Whether postinflammatory hypopigmentation is more common or just more clinically apparent is a matter of debate. In addition, discoid lupus erythematosus and keloids are seen more often in patients with darkly pigmented skin and African ancestry, but the relationship of these disorders to melanocyte function is not clear.
There can also be differences in the physiologic properties of the skin. For example, the stratum corneum of black skin often retains more layers and is more compact and cohesive than that of white skin. In addition, darker skin produces less vitamin D 3 in response to equivalent amounts of sunlight, and this is postulated to have been a driving force in the evolution of paler skin as early humans migrated away from the equator .
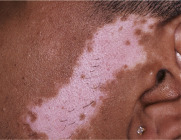
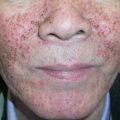
Perhaps the most important point to remember is that erythema (redness) can be difficult to appreciate in darkly pigmented skin. Erythema is caused by vasodilation and/or increased blood flow within the dermis, and if the epidermis is deeply pigmented, the red hues of oxyhemoglobin are often less obvious. For this reason, diseases that are classically described as erythematous (e.g. cellulitis) or violaceous (e.g. lichen planus) may present more subtly in darker skin types ( Fig. 0.6 ) . Diagnostic procedures that depend upon the development of erythema, such as patch testing for the evaluation of allergic contact dermatitis, can be more challenging to interpret in dark skin. Lastly, cyanosis (blue hues indicative of poor oxygenation and a critical clinical sign) is also more difficult to appreciate when the skin is darkly pigmented.
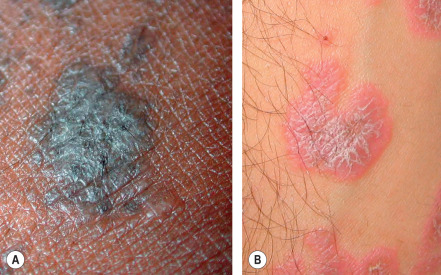
Configuration and Distribution
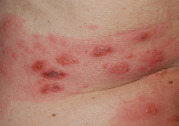
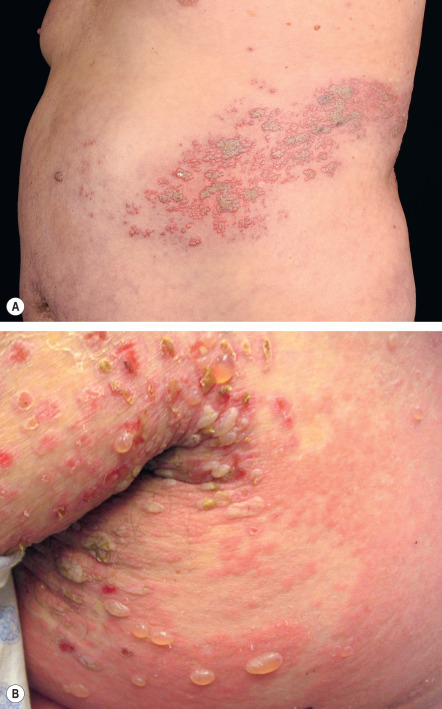
After carefully considering the morphology and color of skin lesions, the dermatologist must next analyze two closely related properties – configuration and distribution – in order to hone in on the correct diagnosis. For example, pruritic and fragile vesicles on the elbows and knees would prompt consideration of dermatitis herpetiformis, whereas grouped vesicles on an erythematous base confined to a single dermatome would mandate consideration of herpes zoster ( Fig. 0.7 ) or zosteriform herpes simplex.
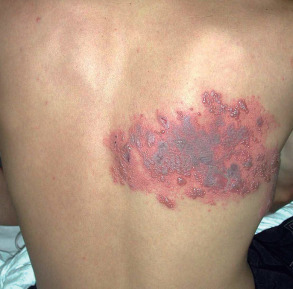
Configuration
Appreciation of the configuration or arrangement of skin lesions can provide important clues as to the diagnosis. Examples include annular (e.g. tinea corporis, granuloma annulare; see Ch. 19 ), serpiginous (e.g. cutaneous larva migrans), clustered/grouped (e.g. piloleiomyomas, herpetiform vesicles), reticulated (e.g. erythema ab igne), and retiform (e.g. purpura fulminans, purpura due to calciphylaxis [ Fig. 0.8 ]; see Ch. 22 ). The latter pattern reflects occlusion of the cutaneous vasculature .
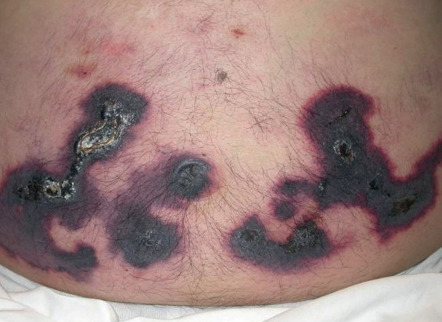
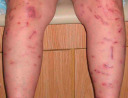
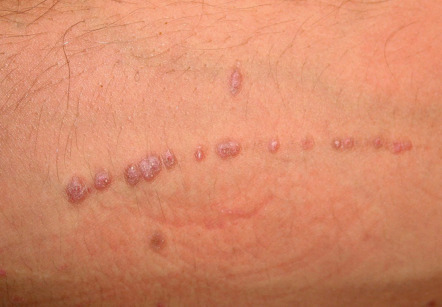
It also important to note if the cutaneous lesions are in a linear configuration ( Fig. 0.9 ). The lesions may follow the lines of Blaschko, which reflect patterns of embryonic development (see Fig. 62.1 ) , or they may be confined to a dermatome, which represents an area of skin whose innervation is from a single spinal nerve (see Fig. 80.14 ). Irrespective of whether the lesions are along the lines of Blaschko (e.g. epidermal nevi) or in a dermatomal pattern (e.g. herpes zoster [see Fig. 0.7 ]), there is often a characteristic midline demarcation. In addition to these two patterns, a linear arrangement can result from a trauma-induced Koebner phenomenon (an isomorphic response [ Table 0.7 ]), as in vitiligo, lichen planus ( Fig. 0.10 ), and psoriasis , or it may be due to trauma-induced autoinoculation, as in verrucae vulgares or verrucae planae. Linear lesions are frequently seen in acute allergic contact dermatitis due to plants (e.g. poison ivy), reflecting brushing of the branches and leaves against the skin. Lastly, papulonodules due to a range of infectious agents can align along lymphatic vessels in a sporotrichoid pattern (see Ch. 77 ).
| CLINICAL ENTITIES THAT COMMONLY DISPLAY THE KOEBNER PHENOMENON (ISOMORPHIC RESPONSE) |
|
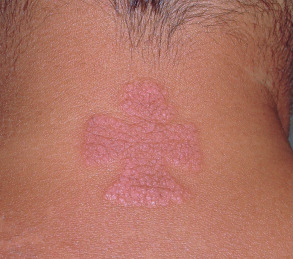
On occasion, cutaneous lesions have an unusual, even “unnatural”, shape that corresponds to an external (exogenous) insult, such as allergic or irritant contact dermatitis ( Fig. 0.11 ), an accidental or purposeful injury (see Ch. 90 ) , or even ritualistic medicinal practices (e.g. “cupping” or “coining”; see Ch. 133 ).
Distribution
Stepping back and observing the anatomic distribution pattern of skin lesions can also prove very helpful. For example, plaques of psoriasis often favor extensor surfaces (e.g. elbows and knees) while lichenified plaques of atopic dermatitis favor flexural surfaces in older children and adults (e.g. the antecubital and popliteal fossae; Table 0.8 ). However, to complicate matters a bit, there is an “inverse” form of psoriasis in which lesions are present in major body folds, i.e. in flexural areas (see Ch. 8 ). Langer cleavage lines refer to natural skin tension lines that are often used to guide the orientation of surgical excisions (see eFig. 142.3 ). The long axis of oval lesions of pityriasis rosea and erythema dyschromicum perstans follows these cleavage lines, and this pattern is most obvious on the posterior trunk.
| MAJOR DISTRIBUTION PATTERNS |
|
A seborrheic distribution pattern includes the head and neck as well as the upper trunk, and it reflects areas rich in sebaceous glands; seborrheic dermatitis, acne vulgaris, and pityriasis versicolor are dermatoses that favor these sites. The term “ photodistribution ” describes lesions that are accentuated in areas exposed to ultraviolet irradiation, and photodermatoses include polymorphic light eruption, phototoxic drug reactions (e.g. to doxycycline), and subacute cutaneous lupus erythematosus. Of note, sometimes a disorder will display a combination of distribution patterns; for example, in dermatomyositis, lesions can be both photodistributed and involve extensor surfaces (e.g. elbows, knees).
In addition to differences in the color of inflammatory lesions, individuals with darkly pigmented skin also have an increased frequency of several cutaneous disorders (see section on Color ) and certain types of reaction and distribution patterns . Examples of these reaction patterns include papular eczema and a follicular accentuation of atopic dermatitis and pityriasis versicolor, as well as an annular configuration of seborrheic dermatitis and facial secondary syphilis. An example of a favored distribution pattern is inverse pityriasis rosea in which lesions occur primarily in the axillae and groin rather than on the trunk. Although a sound explanation for these phenomena is not currently available, it is still important to be aware of their occurrence .
Sometimes the distribution is best explained by the phenomenon of locus minoris resistentiae in which certain anatomic sites are more vulnerable than others to a particular disease process . Examples would be cutaneous infections within a lymphedematous limb and asteatotic eczema within a skin graft site.
Augmented Examination – Wood’s Lamp and Dermoscopy
A Wood’s lamp emits primarily ultraviolet A radiation with a peak wavelength of 365 nm. It is most commonly used to assist in the diagnosis of pigmentary disorders and infectious diseases ( Table 0.9 ) . A Wood’s lamp examination is performed in a dark room, with the lamp 4–5 inches from the skin and illuminating the area of interest. After the target absorbs the UVA radiation, there is some loss of energy and therefore the emission is at a longer wavelength (with less energy) within the visible range. Dermoscopy is discussed in detail later in the chapter.
| WOOD’S LAMP EXAMINATION OF THE SKIN | |
|---|---|
| Disorder/infection/colonization | Fluorescent color/clinical findings |
| Pigmentary disorders | |
| Vitiligo | Chalk-white to dull bluish-white (fluorescence of dermal collagen observed due to a marked decrease or absence of melanin within the epidermis) |
| Ash leaf spots | Enhancement of hypopigmentation |
| Hyperpigmentation due to an increase in: | |
| Enhancement of brown color |
| Difference in color of lesional vs nonlesional skin becomes less obvious |
| Bacterial infections/colonizations | |
| Pseudomonas aeruginosa | Green |
| Corynebacterium minutissimum | Coral red |
| Propionibacterium acnes | Orange–red (in comedones) |
| Fungal infections | |
| Pityriasis (tinea) versicolor due to Malassezia spp. | Yellowish-white, yellow–green, golden, copper–orange |
| Tinea capitis due to Microsporum spp. | Blue–green to yellow–green |
| Favus due to Trichophyton schoenleinii | Blue–white |
Temporal Course
Central to any medical history, including that of cutaneous disorders, is the temporal course. The patient should be queried as to duration and relative change in intensity or distribution over time. For example, there are some dermatoses that have a cephalocaudal progression over time, such as measles and pityriasis rubra pilaris. Of course, the time course is more prolonged in the latter as compared to the former.
The dermatologist is at an advantage because the skin is so accessible, and information provided by the patient can be readily compared to what is seen in the physical examination. With experience, the dermatologist can usually determine by observation whether the cutaneous lesions are acute, subacute or chronic. Examples of helpful signs include scale (not to be confused with crusts), which often reflects parakeratosis that requires 2 weeks to develop, and intact tense bullae, which are rarely more than a week old. Lichenification (i.e. thickening of the skin with accentuation of normal skin markings) takes weeks to months to develop. Therefore, if lichenification is present, the lesion has not appeared acutely, despite what the patient may believe.
In an otherwise generally healthy patient, there are several diseases whose cutaneous manifestations are often acute in nature, in particular urticaria, morbilliform drug eruption, viral exanthem, acute allergic or irritant contact dermatitis, and pityriasis rosea. This is not to indicate that these diseases necessarily require immediate or emergent management, but rather that they present to the dermatologist abruptly and are distinguished, particularly from neoplasms or chronic dermatoses, by their temporal acuity. Of note, sometimes a more serious and potentially life-threatening cutaneous disease may present with skin findings that can mimic a more common and less serious disorder, especially early on.
Finally, although emergencies are unusual in dermatology, there are a few illnesses, particularly those that present with a rash and fever, which are true emergencies and must be recognized promptly and treated appropriately. Examples include Stevens–Johnson syndrome, toxic epidermal necrolysis, Kawasaki disease, meningococcemia (including purpura fulminans), Rocky Mountain spotted fever, necrotizing fasciitis, and endocarditis with cutaneous manifestations. An approach to critical dermatologic emergencies that present with a fever and rash is outlined in Fig. 0.12 .
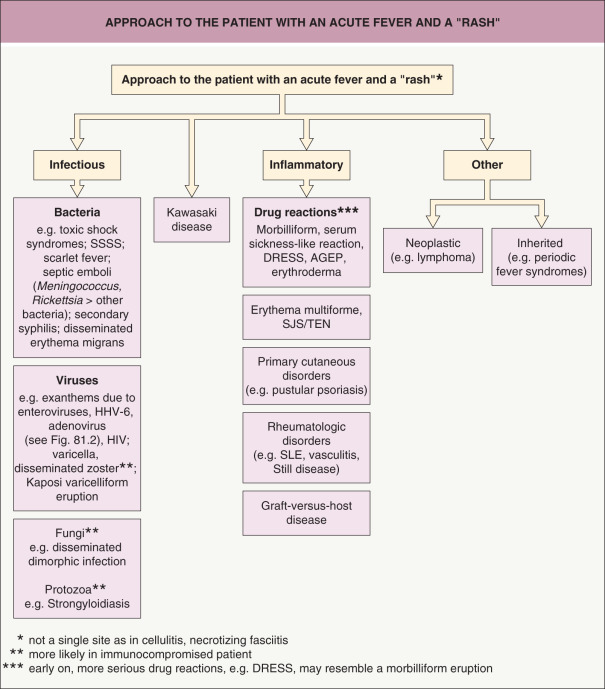
The next two sections of this introductory chapter focus on the basic principles of dermatopathology and dermoscopy, respectively, and it is important to remember that all the diagnostic techniques (unaided clinical examination, histological examination, dermatoscopic examination) discussed herein are complementary. In other words, synergistic strength and clinicopathologic correlation are achieved when the techniques are used in combination. As a corollary, using any one technique, to the exclusion of the others, may be misleading and potentially result in misdiagnosis.
| ACUTE CUTANEOUS ERUPTIONS IN OTHERWISE HEALTHY INDIVIDUALS | |
|---|---|
| Disorder | Characteristic findings |
| Urticaria (see Ch. 18 ) |
|
| Acute allergic contact dermatitis (see Ch. 14 ) |
|
| Acute irritant contact dermatitis (see Ch. 15 ) |
|
| Exanthematous (morbilliform) drug eruptions (see Ch. 21 ) |
|
| Pityriasis rosea (see Ch. 9 ) |
|
| Viral exanthems (see Ch. 81 ) |
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree