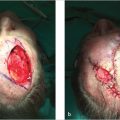
7Auricular Reconstruction
Abstract
This chapter focuses on preservation of the symmetrical projection of the ear (which is the top aesthetic concern in auricular reconstruction, followed by maintaining the nautilus shape), and preservation of the helical rim and special considerations for the same. This chapter also discusses several reconstructive options, and cartilage flaps and grafts that may be needed to prevent auricular distortion. The authors also consider here that not all wounds necessitate reconstruction; many wounds on the anterior and posterior surfaces, not involving the helical rim, heal well by second intention.
Keywords: Auricular reconstruction, cartilage flap, cartilage graft, helical rim advancement, external auditory canal stenosis, retroauricular interpolation
Capsule Summary and Pearls
•Preserving the symmetrical projection of the ear is the top aesthetic concern in auricular reconstruction followed by maintaining the nautilus shape.
•Preservation of the helical rim is important and special considerations should be taken to preserve it.
•When there is significant structural loss, cartilage flaps and grafts may be needed to prevent auricular distortion.
•When there are significant defects involving the external auditory canal (EAC), caution must be taken for stenosis.
•Lastly, it is important to consider that not all wounds necessitate reconstruction. Many wounds on the anterior and posterior surfaces, not involving the helical rim, heal well by second intention.
7.1 Introduction
The structure and form of the ear itself has many complex intricacies, while its functions are simple and basic. The ear has many variations in the quality of the cartilage, skin, and soft tissue that make each auricle unique to reconstruct. The two primary goals of any reconstruction are to maintain the function, and secondarily, preserve the aesthetic form. When reconstructing the auricle, preserving auditory function and maintaining the normal projection of the ear will help attain these primary goals. The cartilage of the ear provides the foundation for its structure and identifiable shape. The anterior and lateral profiles comprise each ear’s unique appearance such that their preservation is critical for a successful reconstruction. When contemplating a reconstruction of the ear, it is often simplest to approach each anatomical zone separately. Table 7.1 lists each of the anatomical zones of the ear. It is best to approach the aesthetics of reconstruction based on the hierarchy listed in Table 7.2. Each auricular defect is unique and calls for careful consideration for each reconstructive option.
Table 7.1 Anatomical zones
Superior helix |
Mid helix |
Lobule |
Antihelix |
Concha |
Tragus |
Remainder of anterior ear |
Remainder of posterior ear |
Table 7.2 Hierarchy of aesthetic concerns
1. Symmetrical projection |
2. Smooth, nautilus-like shape of the helix |
3. Color/texture of skin |
4. Anterior/posterior dimension |
5. Vertical height |
6. Presence and symmetry of tragus |
7. Contour of antihelix and conchal bowl |
The topographical anatomy of the ear is unique (see Fig. 7.1a). An interesting feature of the ear is the variation in its components. Cartilage ranges from soft and delicate to rigid. Skin ranges from elastic on the earlobe to rigid anteriorly. Therefore, it is important to assess and appreciate each ear’s unique features.
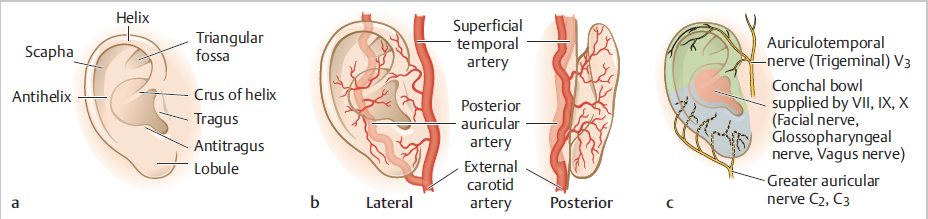
Fig. 7.1 (a) Anatomic subunits of the ear. (b) Vascular supply of the lateral and posterior ear. (c) Cutaneous innervation of the ear.
7.2 Relevant Anatomy
The topographical anatomy of the ear is noteworthy and unlike any other anatomical site on the body. These topographical landmarks are illustrated in Fig. 7.1a. Not only are there multiple anatomical subunits of the ear, but there are also many anatomical variations within each subunit, such as rigid and soft cartilage. The skin on the earlobe is relatively elastic while the skin on the anterior surface has no elasticity. With the multiple convexities and concavities involving the auricle, as well as varying characteristics of the skin and subcutaneous tissue, it is important to assess and appreciate each ear’s unique structure.
While the ear is relatively inconspicuous in its lateral location with limited visibility from the frontal view, change of its projection from the head can lead to noticeable deformities. There is a well-established hierarchy of aesthetic concerns of the ear that are listed in Table 7.2. We recommend prioritizing each concern as listed to obtain the best cosmetic outcome.
The ear has many complexities while its framework is relatively simple. The cartilage serves as the foundation that is then draped by the perichondrium, subcutaneous tissue, then the skin. On the posterior ear, the skin and subcutaneous tissue remains relatively loose and nonadherent as compared with the anterior ear. As a result, the posterior ear has more mobility and, to some degree, can serve as a skin reservoir for flaps and grafts. When performing these procedures, the undermining plane on the ear remains superficial to the perichondrium. If undermining is necessary on the lobule, it is best to do so in the mid fat.
Relaxed skin tension lines are not important for ear reconstructive strategy. Due to the complex topography of the ear, the scars of reconstruction remain relatively camouflaged and inconspicuous and are more dependent on reconstruction design and suturing technique.
The arterial supply of the auricle is from the external carotid artery with two main branches: the superficial temporal artery and the posterior auricular artery (see Fig. 7.1b.) These two networks have extensive anastomoses providing a very rich blood supply. This makes random patterned flaps dependable and relatively low risk with appropriate execution.
Cutaneous innervation of the ear is supplied by both cranial and spinal nerves (see Fig. 7.1c). Cranial nerves VII, IX, and X innervate the conchal bowl. The greater auricular nerve, arising from C2 and C3, innervates the lower half of the ear. The superior ear anteriorly is innervated by the auriculotemporal nerve, a branch of CN V3. The remainder of the superior ear posteriorly is innervated by the lesser occipital nerve (C2, C3).
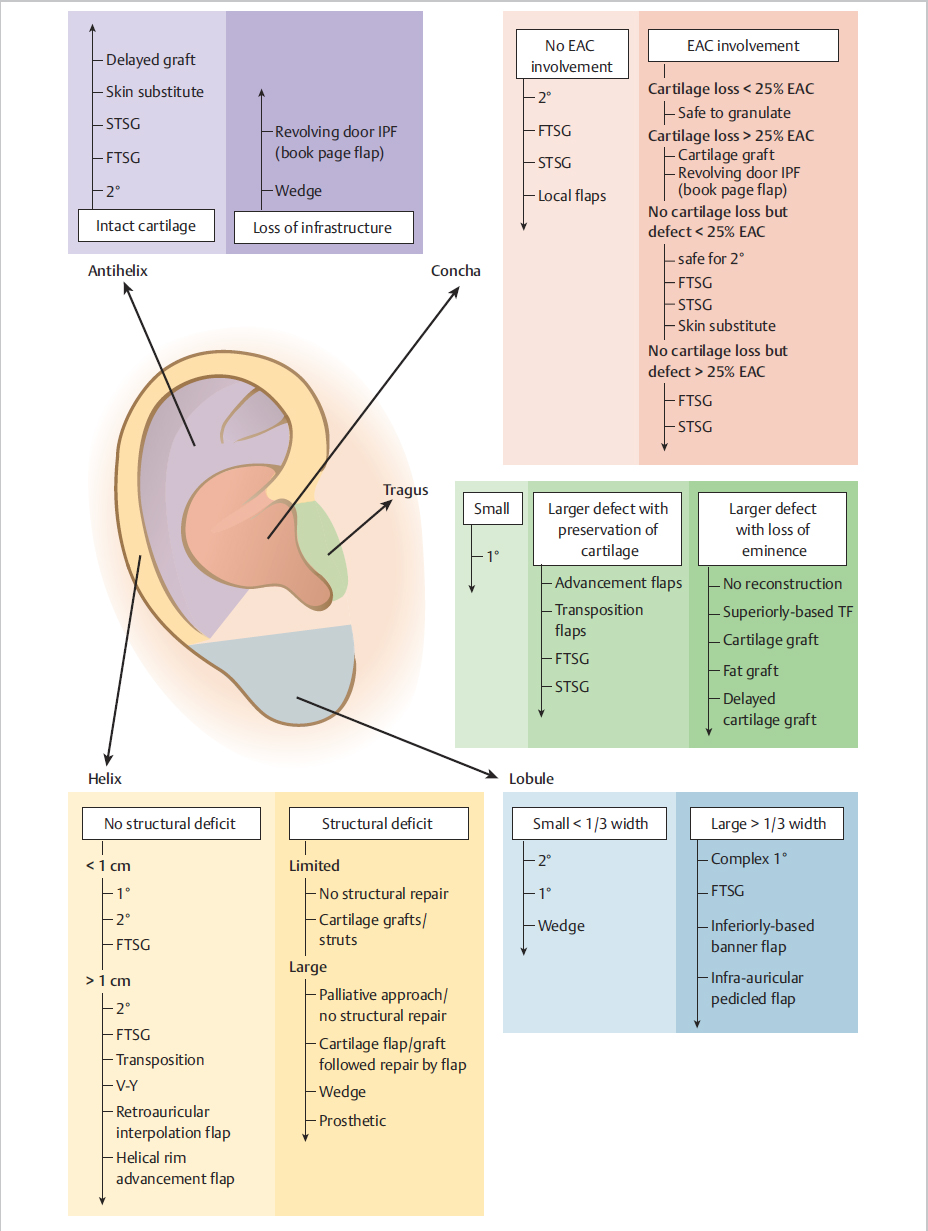
7.3 Reconstructive Options (Fig. 7.2)
7.3.1 Superior Helix
As the most iconic component of the auricle, the helix is responsible for the normal shape and position of the ear. It is comprised of delicate cartilage that can be easily deformed during reconstructions. In addition, it is the most common location of skin cancers of the ear. For these reasons, the proper reconstruction of the helix is paramount to maintaining its normal contour.
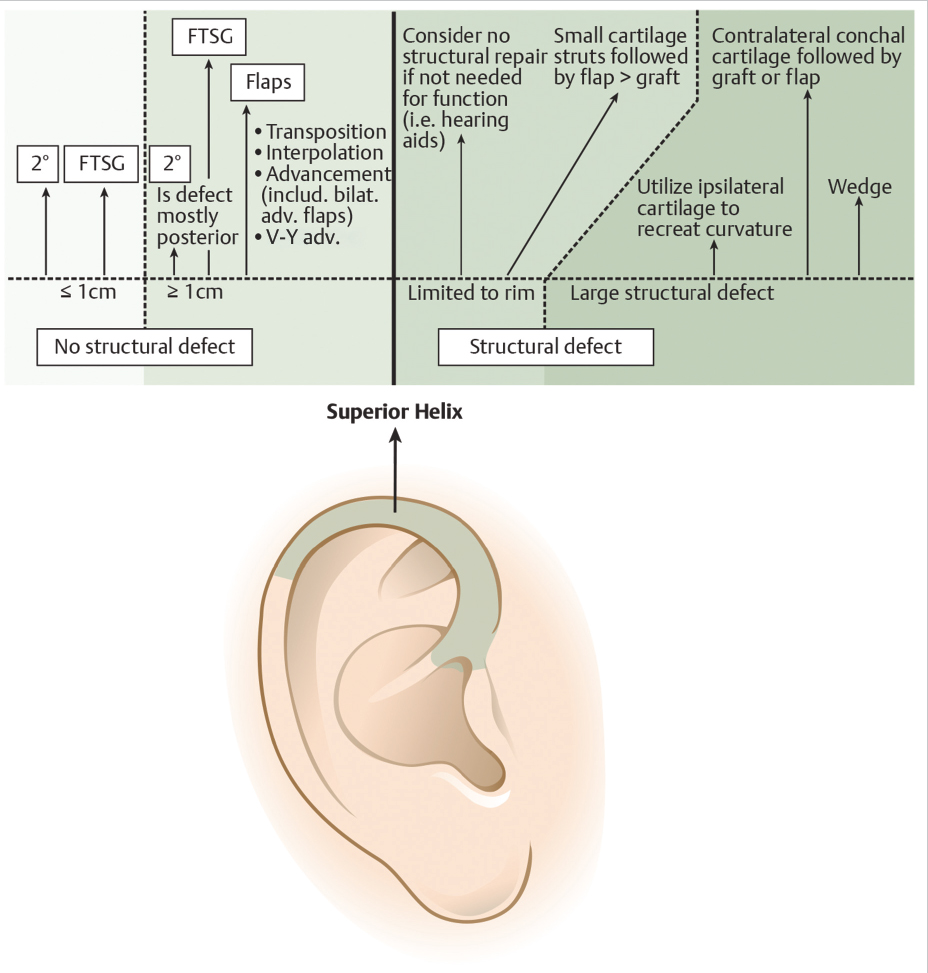
We propose the algorithm in Fig. 7.3 to approach defects on the superior helix. First, it is necessary to determine if there is infrastructural compromise. If there is none, we recommend a simplified closure. If the defect remains small (<1 cm), does not involve the helical groove, and adequate soft tissue remains, a simple primary closure may be performed with excellent results. After undermining above the perichondrium, an elongated linear or curvilinear closure oriented diagonally to the helical rim is a simple method to prevent standing vertical cones and flattening of the helical curvature. This elongated closure often has a larger length-to-width ratio than the typical 3:1 observed at other anatomic locations.
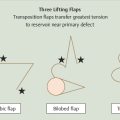
Additional options for the superior helix without structural deficits are second intention, full-thickness skin grafts (FTSGs), and flaps. Second intention is best reserved for wounds that are shallower and have intact perichondrium. They have a smaller risk of auricular distortion from wound contraction as compared with deeper and larger wounds. If the wound is larger and adjacent to the less rigid cartilage of the helical rim, distortion may be more likely. If the wound is more posterior where tissue is looser and supported by underlying rigid structural cartilage, distortion is less likely. FTSGs from the postauricular area, preauricular area, or the conchal bowl are excellent options for repair of superior helical defects without extensive infrastructural compromise. Assessing the wound bed’s vascular base is also important. FTSGs are better utilized with intact perichondrium and structurally insignificant loss of cartilage. Flaps (as listed in the superior helix reconstructive algorithm) should be considered when defects are larger, the wound bed has a poor vascular base, or when larger cartilage grafts have been used for structural support.
Repairs of superior helical defects with loss of structural integrity require more careful planning and discussion with the patient. Patient discussion should detail the types of reconstructions and review the goals and expectations of each option. For example, if the patient depends on the integrity of the auricle for hearing aids, reading glasses, etc., then discussing the need to preserve this function may be necessary. In the case of a larger structural defect, cartilage grafts or struts may be used to optimize auricular restoration and cosmesis.
Donor sites for cartilage grafts are demonstrated in Fig. 7.4. For larger structural defects, there are several reconstructive options. If the nautilus shape of the auricle is compromised, harvesting the entire ipsilateral or contralateral conchal bowl cartilage can be used to recreate this shape nicely. In addition to cartilage grafts, cartilage flaps can be performed. It is possible to transpose or advance cartilage with the aid of a muscular or subcutaneous vascular base. In Fig. 7.5 and Fig. 7.6, a transposition of cartilage is utilized to provide structural support for the helical rim. For defects of the superior helical rim that are deep but remain less than 1.0 to 1.5 cm in width, a wedge closure is an alternate option for repair. If a defect is too large for an aesthetic wedge closure, reestablishing structural support with a cartilage graft and performing a multistaged flap, such as a retroauricular interpolation flap or a tube flap, may yield the most optimal cosmetic results.
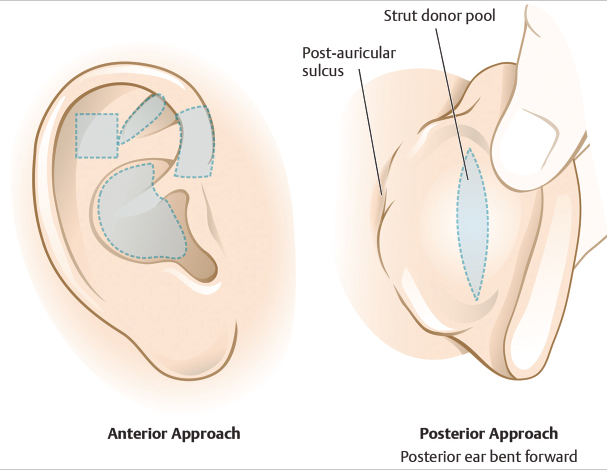
Fig. 7.4 Illustration of cartilage donor sites of the ear.
Superior helix: most visible subunit/iconic feature
I.No structural deficit.
1.≤1 cm:
a)Primary closure.
b)Second intention.
c)FTSG.
2.>1 cm:
a)Second intention.
b)FTSG.
c)Flaps.
•Transposition.
•Interpolation.
•V-Y advancement.
•Helical rim advancement.
II.Structural deficit.
1.Limited to the cartilaginous rim.
a)No structural repair.
b)Small cartilage grafts/struts.
2.Large structural deficit.
a)Palliative approach to expedite wound healing without structural repair.
b)Cartilage graft.
c)Cartilage flap with muscular/subcutaneous base.
d)Wedge closure (defect deep and width <1.5 cm).
e)Cartilage graft/flap with multistage repair (defect >2.5 cm).
Reconstructive Steps for Repairs Suitable for the Superior Helix
Cartilage Grafts
•Select donor site.
–Ipsilateral vs. contralateral.
○Prefer ipsilateral to simplify wound care and minimize number of wounds when practical.
○If cartilage adjacent to wound is accessible (due to exposure) and its use as a cartilage graft would not compromise auricular structure, consider as a potential donor site.
○Contralateral if the donor pool is absent from tumor removal or if ipsilateral harvest would cause structural or aesthetic compromise.
–Conchal bowl.
○Anterior approach.
○Posterior approach.
–Anterior helix.
–Scaphae.
–Crus of antihelix.
Step-by-step:
•Cleanse entire ear including posterior ear and the most external portion of the EAC (not chlorhexidine due to possible ototoxicity).
•Select donor site.
•Create single incision.
–Linear for long, narrow strut-like grafts.
–U-shaped for larger, broader grafts.
•Dissect skin off the underlying cartilage in the perichondrial plane.
•Incise precisely measured cartilage needed.
•Using forceps, carefully stabilize cartilage and dissect cartilage off the undersurface of the skin from the other side of the ear within the perichondrial plane.
–Remember the cartilage is extremely fragile and can fracture easily. This dissection must be performed with caution.
•After cartilage is harvested, place in saline, obtain hemostasis of the donor site.
•We recommend closing the skin on the ear with simple interrupted sutures.
–Interrupted 5–0 chromic or 5–0 fast-absorbing gut suture may be used; may also use nonabsorbable suture (5–0 polypropylene).
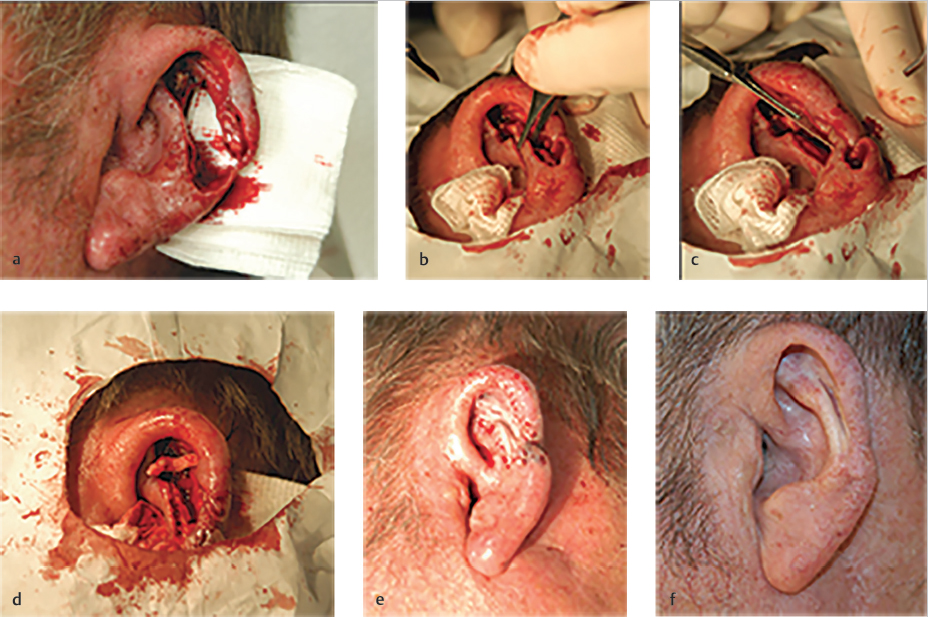
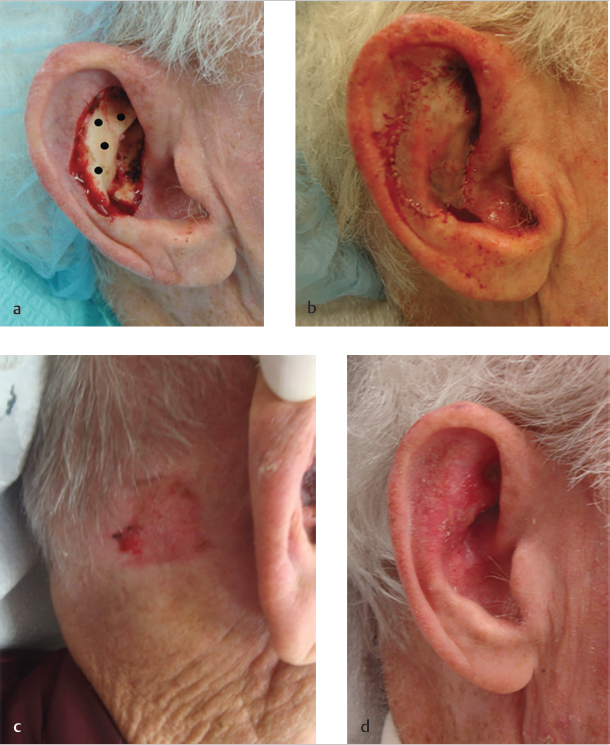
Spoke and Wheel Cartilage Struts (Fig. 7.5a–f)
Goals:
• The wheel: Reestablish structure of outer ear margin (helical rim).
• The spoke: Provide structure radiating from the EAC/conchal bowl (the hub).
Step-by-step:
•Evaluate the defect and determine where the strut would provide the most benefit for infrastructural support.
•Factors to consider:
–Size of the defect.
–Likely direction of maximal wound contraction.
–Size of cartilage strut available.
•Harvest cartilage as previously outlined.
•Harvest strip of cartilage with appropriate curvature to simulate the helix and straight cartilaginous struts to serve as radial structure (the spokes).
•Helical cartilage will need to be enveloped within residual helical skin or well-vascularized flap.
•The radial spokes are typically 3 to 4 mm in width and need to be securely seated in soft tissue pockets on the central end created by sharp and/or blunt dissection. The peripheral end and, if possible, the midportion of the strut must be fixated to the underlying tissue. This end should also be affixed to the helical cartilage by lashing-type suturing or if possible, direct suturing to the cartilage.
•With a vascularized base of either remnant ear tissue or flap tissue, the exposed side of the spoke and wheel structure may be covered with a split-thickness skin graft (STSG). An STSG is preferred because its lower metabolic requirement is preferred as graft coverage.
•A tightly compressed bolster dressing is key to the secure immobilization of the new structure to avoid dislodgement and to assure survival of the graft. Extreme care for ideally one month postoperatively is critical to avoid dislocation of the cartilage components and should be emphasized to the patients.
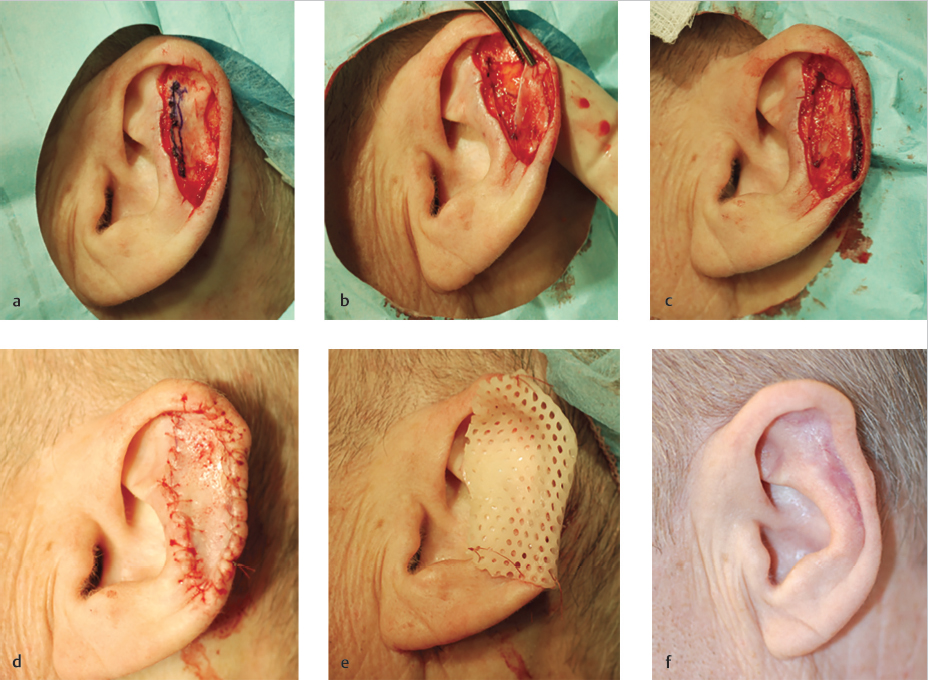
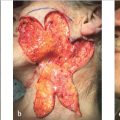
Cartilage Flap (Fig. 7.6a–f)
Step-by-step:
•Determine where the infrastructural support is needed as described in the spoke and wheel instructions.
•Identify donor source.
–Ideally cartilage can be transposed or slightly rotated on a perichondrial or subcutaneous base.
•Ideal to recreate the natural architecture of the helix.
–Important to be mindful of the contractile forces of healing and provide structural support to maintain the natural shape of the ear.
•Once donor site is identified, incise the cartilage, again leaving the subcutaneous base intact.
–There are effectively two methods to executing a cartilage flap depending on the flexibility/rigidity of the cartilage.
○If cartilage is more rigid, it may require complete separation from the auricle, remaining embedded within the skin flap with the posterior surface as a vascular base (serving as an island pedicle flap).
•Very gently move cartilage to desired location.
–Important to acknowledge the fragility of the cartilage and ease with which this may fracture.
•Suture into place with 5–0 or 4–0 absorbable suture or clear nylon.
•May place graft or flap over the cartilage flap.
–Important for cartilage to have a vascular base to optimize survival.
•We recommend the use of a bolster or even a moldable thermoplastic splint to further immobilize and shape the flap +/− graft for 1 week.
–After removal of the bolster, may preserve the thermoplastic ic splint and continue to use as a formal brace for one or more additional weeks thereafter.
–Recommend bandaging the area for protection from accidental distortional injury for up to one month postoperatively.
*In this case, adequate movement of flap could be obtained without completely incising and separating the cartilage flap from the inferior antihelix (Fig. 7.6b, c).
Full-Thickness Skin Grafts (FTSGs)
Donor sites:
•<1.5 cm:
–Consider postauricular sulcus (relatively easy to close primarily).
–Conchal bowl (donor site heals by second intention).
•>1.5 cm:
–Postauricular.
–Preauricular.
–Conchal bowl.
–Supraclavicular.
–Upper inner arm.
–Burow’s triangle from closure of another defect.
Step-by-step:
•Create template of defect.
•Place template at the donor site over taut skin.
•Trace template/score skin with scalpel.
•Pearl: keep template on sterile tray to use in “right sizing” nonstick gauze to be placed over the graft at the conclusion of the procedure.
•Raise the graft in the subdermal plane with scissors or scalpel.
•Defat the graft to visualize glistening, white dermis; thin as needed, tailored to the wound depth.
•Place graft in saline.
•Obtain hemostasis and suture donor site.
–4–0 or 5–0 dermal +/− epidermal sutures.
•Suture graft in place.
–Recommend 6–0 or 5–0 fast-absorbing gut with interrupted sutures placed in key locations (4 corners on a <2-cm graft) to secure the orientation of the graft in the defect.
–Running 6–0 or 5–0 fast-absorbing gut around the perimeter of the graft focusing on perfect edge approximation and contact of the graft with the wound bed.
○May need basting suture(s) to maximize wound bed contact and/or recreate natural topography of the ear (i.e., helical groove, etc.).
•Apply a thin layer of Vaseline to the graft.
•Create bolster dressing utilizing the template for sizing the nonadherent gauze (Telfa) on the graft and folded gauze overtop. Vaseline gauze is also an effective bolster material.
•Tie bolster with remainder of dermal suture or other nonabsorbable suture.
•Consider thermoplastic ic splint in lieu of gauze bolster dressing.
Split-Thickness Skin Graft (STSGs) (Fig. 7.7a–d)