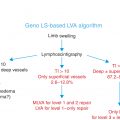
Key Points
- •
Lymphangiogenesis occurs in four distinct regular phases: lymphatic competence, lymphatic commitment, lymphatic speciation, and lymphatic maturation.
- •
Vascular endothelial growth factor C and vascular endothelial growth factor receptor-3 signaling are essential for lymphangiogenesis.
- •
Animal models of secondary lymphedema have expanded our understanding of its pathogenesis and have facilitated the advancement of its treatment.
- •
Animal models allow further investigation of the physiologic mechanism, molecular basis, and long-term outcomes of vascularized lymph node transfer and lymphovenous anastomosis in the treatment of lymphedema.
- •
Animal models of lymphedema demonstrate a promising role for growth factor–mediated therapies and nanofibrillar collagen scaffold to augment lymphedema treatment.
- •
The critical venous occlusion time for vascularized lymph node flap in a rodent model was 4 hours, which was shorter and more severe than the critical arterial ischemia time of 5 hours.
- •
Cadaver studies provide for better understanding of human anatomy of the superficial and deep lymphatic system and extend the donor vascularized lymph node basins for clinical applications.
Introduction
The lymphatic system is essential to maintaining tissue-fluid homeostasis and aids in immune surveillance by trafficking lymphocytes and antigen-presenting cells to the lymph nodes. Dysfunction in the lymphatic system leads to lymphedema, a chronic and progressive disease where regional accumulation of interstitial fluid, macromolecules, and cellular debris. This leads to regional compromise of immune function and irreversible changes to the affected tissue, skin thickening, subcutaneous fibrosis, and recurrent infections.
Lymphedema is a chronic and debilitating disease. In developed and developing countries, it is commonly a delayed sequelae of cancer treatment secondary to lymph node extirpation and/or radiation therapy. Effective pharmacologic and surgical therapies remain a challenge and currently there is no cure.
Overview of Lymphedema Research
This chapter highlights the historical and current basic research in lymphedema. The development of animal models to study the lymphatic system has resulted in better understanding of the genetic, molecular and pathophysiology of both inherited and acquired lymphedema. Animal models with chronic acquired lymphedema has allowed for refinement of surgical techniques and evidence-based assessment of treatment outcomes.
Molecular Investigation
The mechanisms of lymphangiogenesis were first described by Florence Sabin in the early 1900s based on experiments in pigs describing lymphatic development arising from primary lymph sacs that emerge from embryonic veins. Subsequent studies using genetic mouse models described early lymphatic vascular development as arising from signal-dependent differentiation of embryonic venous endothelial cells. Rickson et al. described the progression of lymphangiogenesis as four distinct stages: lymphatic competence, lymphatic commitment, lymphatic specification, and lymphatic maturation.
Lymphatic competence occurs between embryonic day 8.5 and 9.5, where all venous endothelial cells express vascular endothelial growth factor receptor-3 (VEGFR-3) and lymphatic vessel endothelial hyaluronan receptor-1 (LYVE-1). Endothelial cells can be stimulated to undergo lymphatic differentiation by vascular endothelial growth factor C (VEGF-C) and its receptor (VEGFR-3). Studies of VEGFR-3 knockout mice showed early death from cardiovascular failure and VEGF-C knockout mice showed complete cessation of lymphatic formation.
Currently, the Chy mouse model that expresses a heterogeneous inactivation of VEGFR-3 mutation is used as a model for human primary hereditary lymphedema, Milroy’s disease. In this model, lymphatic function is restored after treatment with a virus-mediated delivery of recombinant VEGF-C. Crossing a Chy mouse with a mouse strain that overexpresses the VEGFR-3–specific ligand VEGF-C156S results in lymphatic function restoration in the double-transgenic offspring. These studies indicate VEGF-C as a promising potential molecular therapy for lymphatic insufficiency.
Lymphatic commitment occurs at embryonic day 9.5–10.5 and is the stage where subpopulations of embryonic venous endothelial cells are marked for lymphatic differentiation via expression of transcription factor prospero-related homeobox (PROX-1). An unknown signal from surrounding mesenchymal cells is thought to stimulate PROX-1 transcription factor. Studies in support of PROX-1 knockout mice demonstrate the absence of lymphatic development, which indicates PROX-1 role in lymphatic-specific differentiation.
Embryonic days 10.5–11.5 mark lymphatic specification and are characterized by the expression of additional lymphatic-specific markers and downregulation of vascular endothelial-specific markers.
Rudimentary lymphatic sacs start to form on embryonic days 11.5–12.5 in a process called lymphatic maturation. By embryonic day 14.5, these lymphatic vessels’ maturation is nearly complete. The process of lymphatic vessel maturation and organization continues until the early period after birth, forming the adult lymphatic network.
Animal Models
Genetic animal models have provided insight into the embryologic development of the lymphatic system and possible mechanisms of primary lymphedema. More recent animal models of secondary lymphedema have improved our understanding and guided advancement in treatment ( Table 3.1 ). The challenge with animal models is to create reproducible and sustained lymphedema results.
| Animals | Lymphedema Model(s) | Success Rate of Lymphedema | Significances/Clinical Relevance | Advantages | Disadvantages |
|---|---|---|---|---|---|
| Canine | Preop/postop irradiation in combination with excision of a strip of skin, connective tissues, fascia, and lymphatic trunks in the hind limb | 66.6%–70.6% |
| Share similarities in lymphatic anatomy as humans |
|
| Sheep | Excision of a single popliteal lymph node and ligation of pre- and postnodal lymphatic vessels | 100% | Ability to quantify lymphatic drainage using radiolabeled albumin | Surgery is easy to perform due to straightforward anatomy and large size lymph node and lymphatic vessels |
|
| Rabbit |
|
|
|
|
|
| Rodent |
|
|
|
|
|
Canine
In 1968, Olszewski et al. described a canine lymphedema model by transecting the main lymphatics of the hind limb and excising a circumferential strip of skin, subcutaneous, fascia, and periosteum, leaving the tissue gap to heal by secondary intension. This model was modified to completely remove the lymphatic chain in the thigh in a subsequent surgery. This improved the rate of lymphedema development from one-third to two-thirds. Das et al. and Chen et al. both reported generating lower limb lymphedema in dog by circumferential excision of soft tissue down to the muscle fascia followed by irradiation of the open wound. These canine animal models were used to study lymphovenous anastomoses and efficacy of vascularized lymph node transfer in reversing lymphedema, although not completely.
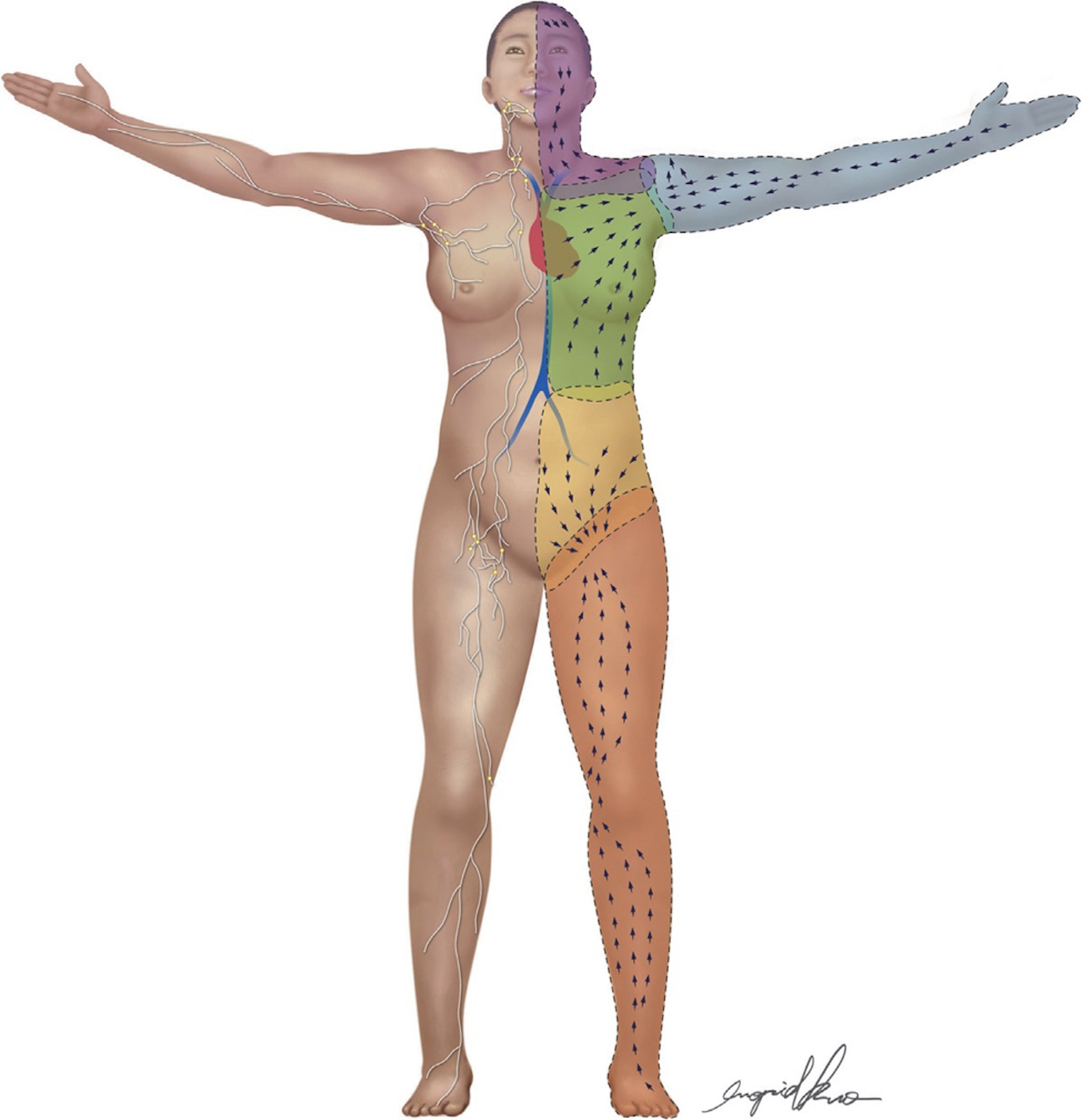
Suami et al. utilized the canine model to study lymphatic territories that showed similarities in the superficial and deep lymphatic system between canines ( Fig. 3.1 ) and humans ( Fig. 3.2 ). The canine model demonstrated lymphatic collateral formation after lymph node dissection, which may help improve understanding the lymphatic changes post lymph node dissection and radiation in human.

Sheep
Sheep were used by Tobbia et al. to create a lymphedema model after excision of a single lymph node in the popliteal region and ligation of the pre- and postnodal lymphatic vessels. They observed maximal lymphedema at 3 days postoperative with 80% of lymphatic function return by 12–16 weeks. This study demonstrates the regenerative capacity and development of collateral lymphatic pathways. Tobbia et al. also demonstrated in this sheep model that improvement in lower limb lymphedema can be achieved after vascularized lymph node transfer and that there is no improvement with non-vascularized lymph node transfer.
Rabbit
Some researchers have explored the rabbit ear as a model for lymphedema; this model is more affordable and less time consuming than other large mammal models. The rabbit ear lacks a deep lymphatic system and the main neurovascular and lymphatic systems are readily located at the base of the ear, which makes the surgical dissection easier. Through excision of a circumferential strip of tissue down to the chondrium and ligation of the lymphatic trunk, Huang et al. created stable lymphedema that persisted for more than 6 months. Critics were unable to replicate the results and reported cartilage necrosis. Fu et al. modified the technique to spare the pericondrium and observed no cartilage necrosis. Further use of this model demonstrated histologic evidence of lymphatic regeneration and improved lymphatic drainage after lymph node fragment transplantation. A subsequent study reported that this model was only effective in adult rabbits.
Local administration of human recombinant VEGF-C resulted in improved lymphatic function and decreased dermal fibrosis and showed histologic evidence of lymphangiogenesis. In vitro studies showed that bone marrow stromal cells (BMSCs) stimulated with VEGF-C can form lymphatic vessels. Zhou et al. used these findings to show that combined VEGF-C and BMSCs have a synergistic effect on lymphangiogenesis in the treatment of acquired lymphedema in the rabbit limb model.
Rodent
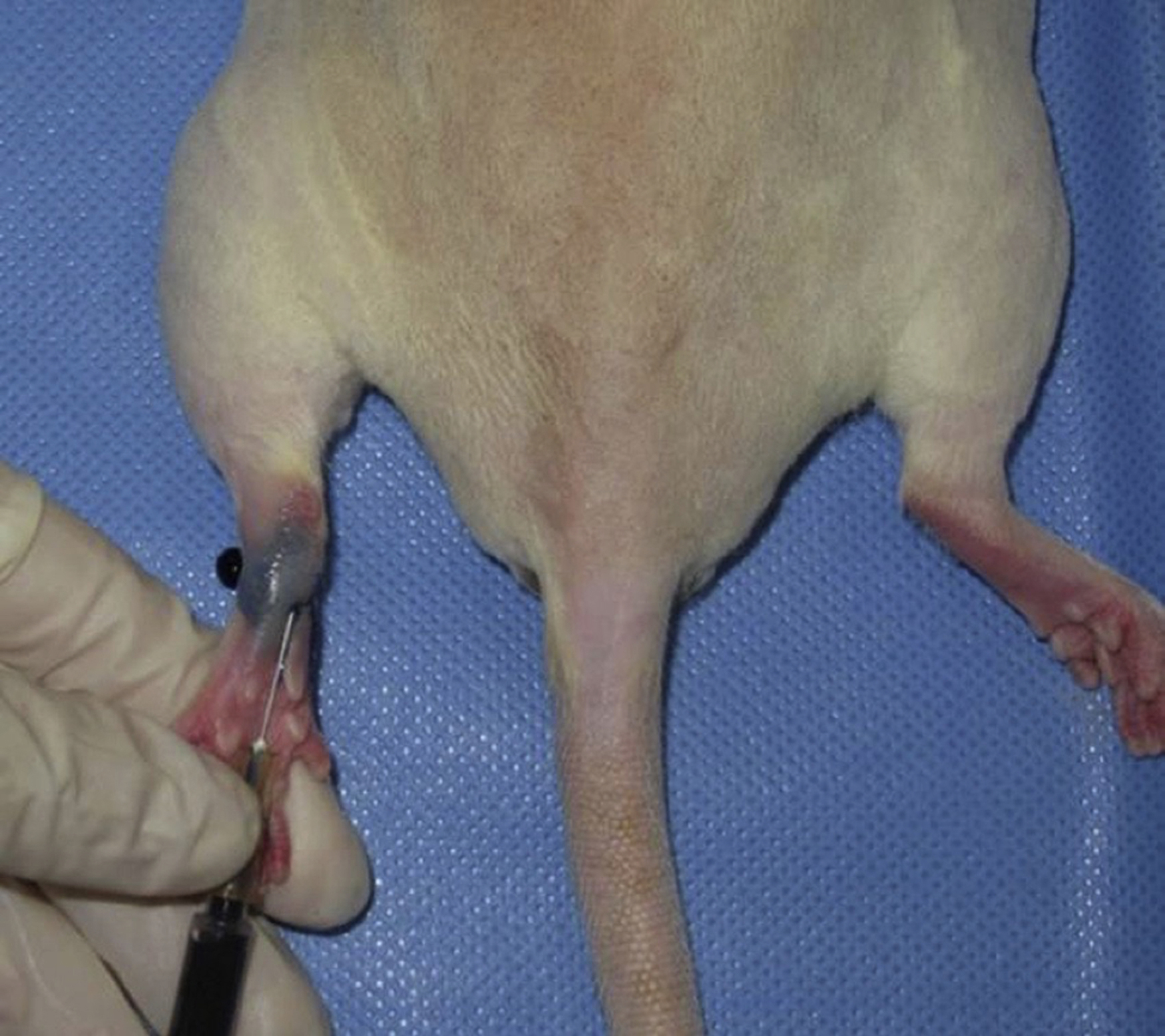
The rodent animal model is more accessible and inexpensive than large mammal models, which has led research to the development of various rodent models. The rat tail model gained popularity due to its simplicity and effectiveness in reproducing lymphedema. A circumferential incision at the base of the tail is made, the skin is retracted 3 mm, and the deep lymphatic trunk is cauterized. The rat tail model has been used to investigate the role of VEGF-C/VEGFR-3 signaling and the protective role of matrix metalloproteinase (MMP-9). Studies have shown that VEGF-C may be useful in augmenting lymphatic proliferation in the setting of absent or deficient lymphatics but not when lymphatics are present but poorly functioning. Other studies utilized the rat tail lymphedema model to show upregulation of inflammation and oxidative stress genes.
Critics of the rat tail lymphedema model suggest that it lacks correlation to human disease because the rat tail is devoid of lymph nodes and the hydrodynamics and cellular mechanism of lymphatic drainage are different from a human limb.
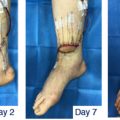
The development of a reliable and reproducible rodent hind limb model has been challenging. Initial models that removed a skin flap and resection of underlying lymphatic truck alone were not successful due to the rapid lymphatic regeneration in rats. Further research showed that combined with preoperative or postoperative radiation, chronic lymphedema was achieved. Sommer et al. reported removal of inguinal and popliteal lymph nodes and adjacent lymphatic vessels in the hind leg, followed by 15 Gy radiation with stable lymphedema at 4 weeks post treatment. Further research demonstrated regeneration of transplanted lymph node fragments across areas of radiation and lymphadenectomy with exogenous injection of VEGF-C. Yang et al. further showed chronic lymphedema in the rat model beyond 3 months by complete inguinal and popliteal lymphadenectomy, superficial and deep lymphatic ligation, followed by a single dose of 20 Gy postoperative radiation ( Figs. 3.3–3.5 ). Nguyen et al. demonstrated that vascularized para-aortic lymph nodes transfer to the acquired lymphedema limb with correction correlated with the number of lymph nodes transferred ( Figs. 3.6–3.8 ). Cheng et al. developed a rat model to quantitatively evaluate lymph drainage using indocyanine green imaging. Using this model, they demonstrated the direct role of the vascularized lymph node in draining lymph via natural lymphaticovenous connections in the vascularized lymph node flap into the recipient vein ( Figs. 3.9–3.12 ). This was not observed in a skin flap without lymph nodes ( Figs. 3.13 and 3.14 ). Their experimental data strongly corroborated with clinical findings. This study thus far proved the mechanism of vascularized lymph node flap transfer.