Analysis of Breast Reduction Complications Derived from the BRAVO Study
Bruce L. Cunningham M.D., M.S.
Andrew J. L. Gear M.D.
Carolyn L. Kerrigan M.D.
E. Dale Collins M.D.
Minneapolis, Minn.; and Lebanon, N.H.
From the Division of Plastic Surgery, University of Minnesota, and the Section of Plastic Surgery, Dartmouth Hitchcock Medical Center.
Received for publication March 18, 2004; revised June 18, 2004.
DOI: 10.1097/01.PRS.0000160695.33457.DB
Background: Analysis of complication data derived from the Breast Reduction Assessment: Value and Outcomes (BRAVO) study, a 9-month prospective, multicenter trial, is presented.
Methods: Data derived from 179 patients were analyzed, including bivariate associations between complications and single predictor variables (Fisher’s exact test or chi-square testing) or continuous variables (two-sample t test) and, finally, logistic regression.
Results: The overall complication rate was 43 percent (77 patients). Simple, bivariate analysis linked preoperative breast volume, shoulder strap grooving, and a vertical incision with an increased incidence of complications (p < 0.05, 0.02, and 0.02, respectively). Delayed wound healing, the most common complication, correlated directly with average preoperative breast volume (p < 0.045), average resection weight/breast (p < 0.027), and smoking (p < 0.029) and inversely with age (p < 0.011). Vertical incision techniques were associated with an increased complication frequency (p < 0.05) without a link to specific complications. Logistic regression analysis associated resection weight as the sole variable for increased risk of complications (p = 0.05) and with absolute number of complications [mean resection weight of 791 g for patients without complications versus 847, 882, and 1752 g for patients with one, two, and three complications, respectively (p = 0.0022)]. Each 10-fold increase in resection weight increased the risk of complication 4.8 times and increased the risk of delayed healing 11.6 times.
Conclusions: Complication data revealed several significant features: (1) resection weight correlated with increased risk and absolute number of complications; (2) delayed healing correlated directly with resection weight and inversely with increasing age, anesthesia times, and preoperative Short Form-36 bodily pain score; (3) a vertical incision may be associated with increased incidence of complications but requires further analysis; and most importantly, (4) the presence of complications had no negative effect on improvement in Short Form-36 and Multidimensional Body-Self Relations Questionnaire scores. (Plast. Reconstr. Surg. 115: 1597, 2005.)
The delineation of variables associated with complications following breast reduction surgery remains incomplete. As early as 1964, an association among resection weight, obesity, and an increased complication rate was documented.1 Subsequent studies failed to consistently correlate obesity, typically defined as either percentage or kg above ideal body weight, with an increased risk of complications.2–8 However, resection weight of breast tissue has
consistently been correlated with an increased complication frequency.1,4,8–12 One author has noted increased seroma formation and delayed wound healing with the addition of liposuction.13 To date, no statistically significant correlation has been established between patient age, smoking status, measured preoperative breast volume, specific techniques, operative and anesthetic times, drain use and complications following breast reduction surgery. No previous study has clearly demonstrated the potential effect of complications on patient satisfaction with the operation or with potential effect on health or functional measurements. Furthermore, most of the existing complication data have been derived retrospectively, and thus the need for controlled, prospective data arises.
consistently been correlated with an increased complication frequency.1,4,8–12 One author has noted increased seroma formation and delayed wound healing with the addition of liposuction.13 To date, no statistically significant correlation has been established between patient age, smoking status, measured preoperative breast volume, specific techniques, operative and anesthetic times, drain use and complications following breast reduction surgery. No previous study has clearly demonstrated the potential effect of complications on patient satisfaction with the operation or with potential effect on health or functional measurements. Furthermore, most of the existing complication data have been derived retrospectively, and thus the need for controlled, prospective data arises.
The need to document the medical validity of breast reduction has spawned several prospective multicenter studies, including the Breast Reduction Assessment: Values and Outcomes (BRAVO) study in 1997/1998.14 BRAVO was designed to address deficiencies in previous research on the outcomes of breast reduction surgery by using a prospective, controlled design with a comprehensive set of standardized instruments for measuring a variety of outcomes, including general health, function, pain, and psychosocial status. In addition, an analytic model was developed to identify the predictors of surgical outcomes. BRAVO was conducted as a multicenter study of outcomes of women undergoing breast reduction at various geographic locations in different practice settings with two control groups (normal and hypertrophy controls who did not present for surgery) for comparison.
The BRAVO study, which was conducted in 14 separate sites over 15 months, compared patients who underwent breast reduction (n = 179) with controls with and without breast hypertrophy (n = 195) and significantly demonstrated the efficacy of breast reduction in diminishing symptoms associated with mammary hypertrophy, as measured by the Short Form-36 health questionnaire, a commonly used measure of health function levels, the Multidimensional Body-Self Relations Questionnaire survey, and the McGill Pain Questionnaire. This article represents analysis of complication data derived from the BRAVO study, with the intention of further elucidating variables implicated in morbidity following a breast reduction operation.
Patients and Methods
Data derived from the BRAVO trial study were reviewed. Fourteen sites (both academic and private) across the country participated in data collection for approximately 15 months, during which time 291 surgical candidates and 195 controls were enrolled. Study participants completed both a baseline questionnaire before the breast reduction operation and a postsurgical patient questionnaire approximately 6 to 9 months following the operation, including both the Short Form-36 health questionnaire and the Multidimensional Body-Self Relations Questionnaire. The Short Form-36 is a widely used standardized and validated measure of health status and function. Control patients completed the same questionnaire as the preoperative surgical patients. Of the 291 surgical candidates, 243 actually underwent the operation, and 179 completed the postsurgical questionnaire. The physician/study coordinators at each study site completed an initial consultation form for each patient enrolled, an operative data form, and a postoperative form for each postoperative visit. The mean number of days between the date of the breast reduction operation and completion of the postoperative patient questionnaire was 246 days, or approximately 8 months (SD, 3 days; minimum, 169 days; maximum, 628 days).
Complication events recorded and evaluated included delayed healing, infection, hematoma, seroma, spitting sutures, allergic reactions, nipple necrosis, fat necrosis, and hypertrophic scars. These complications were included as part of the postoperative data form completed at each postoperative visit and were graded as none (1), mild (2), moderate (3), or severe (4).
Statistical Analysis
Statistical analysis was performed using SSPS statistical software (SSPS, Inc., Chicago, Ill.). Descriptive statistics included mean and SD for continuous variables and percentage for categorical variables. The analysis of complications was performed in several ways, including number of complications and any complication. Any complication was defined as having at least one of the 10 complications listed on the postsurgical form, including the “other” category, at the mild or worse level. Complication rate was reported as the percentage of patients having any complication. Bivariate associations between
any complication and potential categorical predictor variables such as smoking status were assessed using the Fisher’s exact test or chi-square test. The bivariate associations between any complication and continuous predictor variables such as age were assessed using the two-sample t test.
any complication and potential categorical predictor variables such as smoking status were assessed using the Fisher’s exact test or chi-square test. The bivariate associations between any complication and continuous predictor variables such as age were assessed using the two-sample t test.
Logistic regression was then performed to evaluate potential relationships between any complication and several predictor variables. The model used any complication as the dependent variable and incorporated explanatory variables that were found to be statistically significant in the bivariate relationship analyses, in addition to variables thought to be associated with complications such as age. From this model, adjusted odds ratios and their 95 percent confidence intervals were estimated as measures of association between any complication and each of the explanatory variables and adjusted for other variables included in the model. An odds ratio greater than 1 means the explanatory variable is associated with an increased risk of having any complication. An odds ratio less than 1 means the explanatory variable is associated with a reduced risk of having any complication. Furthermore, if the 95 percent confidence interval of the odds ratio equals 1, there is no statistical association between the explanatory variable and having any complication. Similar analysis was performed for delayed healing.
Results
Comparison of the 179 operative subjects with follow-up data and the 64 women who underwent the operation but were lost to follow-up was done to rule out a systematic bias in the evaluation of complication rates. Patients lost to follow-up were not significantly different from the 179 research subjects included in this study with respect to age, body mass index, educational level, employment status, income, race, or brassiere cup size.
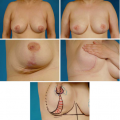
The patients included in this study were mostly white and well educated, with an average age of 39.4, a mean body mass index [Quetelet’s index (weight/height)] of 29.7, a median brassiere cup size of DD, and a median preoperative measured breast volume of 1662.6 cc per breast. Of the 179 patients, 146 patients (81.6 percent) had a keyhole incision, while 23 patients (12.8 percent) had a vertical skin incision (Table I). Approximately 78 percent of the patients’ breasts had an inferior vascular pattern; 34 patients (19.5 percent) had a superior vascular pattern (Table I). Twenty-five percent of surgical patients had liposuction in conjunction with breast reduction. The average volume of liposuction was 136.2 cc, with a range of 20 to 363 cc. The average tissue resected was 814 g per breast (SD, 596.3; range, 206.5 to 3240 g). Ninety percent of patients had general anesthesia, 30 percent had local anesthesia in conjunction with dilute epinephrine, and 46 percent had drains placed either on one or both sides of the breasts (Table I). The average operative and anesthetic times were 121 and 154.4 minutes, respectively.
TABLE I Surgical Data | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
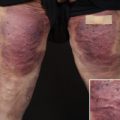
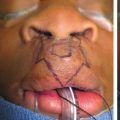
Seventy-seven patients had at least one complication, producing a complication rate of 43 percent. Fifty-nine patients (33 percent) had one complication, 14 (8 percent) had two, and four (2 percent) had three complications. The incidences of specific complications were as follows: delayed wound healing (21.6 percent), spitting sutures (9.2 percent), hematoma (3.7 percent), nipple necrosis (3.6 percent), hypertrophic scars (2.5 percent), fat necrosis (1.8 percent), seroma (1.2 percent), and infection (1.2 percent) (Table II). Using bivariate analysis, average preoperative breast volume, a vertical incision, and preoperative shoulder grooving were associated with an increased
incidence of complications (p < 0.05, 0.021, 0.02, respectively) (Table III). Age, smoking status, body mass index, weight of breast tissue resected, pedicle type, keyhole incision, free nipple grafting, operative time, use of epinephrine, drains, and liposuction were not associated with an increased incidence of complications (Table III).
incidence of complications (p < 0.05, 0.021, 0.02, respectively) (Table III). Age, smoking status, body mass index, weight of breast tissue resected, pedicle type, keyhole incision, free nipple grafting, operative time, use of epinephrine, drains, and liposuction were not associated with an increased incidence of complications (Table III).
TABLE II Complications | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Bivariate analysis comparing skin incisions demonstrated that the vertical skin incision had a higher incidence and total number of complications versus the keyhole incision (Table IV). Three patients who had both keyhole and vertical incisions were excluded. Vertical and keyhole skin incisions produced complication rates of 60 percent and 37 percent, respectively (p = 0.05; Pearson chi-square test). Comparative univariate analysis of associated variables revealed significant differences in age (p = 0.0012; t test), resection weight (p = 0.0081; t test), inferior pedicle use (p = 0.000; Fisher’s exact test), and superior pedicle use (p = 0.000; Fisher’s exact test) (Table IV). There were no significant differences in the incidence of specific complications between the two groups (Table IV). Complications included in the “other” category for the vertical group included dog-ears (two), reddened scar (one), and asymmetry (one). Complications in the “other” category for the keyhole group included dog-ears (one), epidermolysis (two),
drainage (one), dehiscence (one), ecchymosis (one), pneumothorax (one), numbness (one), hypesthesia (one), blistering (three), and nipple depigmentation (one).
drainage (one), dehiscence (one), ecchymosis (one), pneumothorax (one), numbness (one), hypesthesia (one), blistering (three), and nipple depigmentation (one).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree