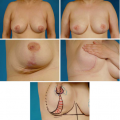
An Evidence-Based Approach to Reduction Mammaplasty
R. Barrett Noone M.D.
Bryn Mawr Pa.
From the Division of Plastic Surgery, Department of Surgery, University of Pennsylvania School of Medicine.
Received for publication January 26, 2010; accepted April 30, 2010.
Copyright ©2010 by the American Society of Plastic Surgeons
DOI: 10.1097/PRS.0b013e3181f830d7
Disclosure: The author has no financial interest in any of the products, drugs, or devices mentioned in this article.
The Maintenance of Certification module series is designed to help the clinician structure his or her study in specific areas appropriate to his or her clinical practice. This article is prepared to accompany practice-based assessment of preoperative assessment, anesthesia, surgical treatment plan, perioperative management, and outcomes. In this format, the clinician is invited to compare his or her methods of patient assessment and treatment, outcomes, and complications, with authoritative, information-based references.
This information base is then used for self-assessment and benchmarking in parts II and IV of the Maintenance of Certification process of the American Board of Plastic Surgery. This article is not intended to be an exhaustive treatise on the subject. Rather, it is designed to serve as a reference point for further in-depth study by review of the reference articles presented. (Plast. Reconstr. Surg. 126: 2171, 2010.)
Clinical Scenario
A 27-year-old woman desires symptomatic relief of neck, back, and shoulder pain by reduction mammaplasty. She has never been pregnant, desires to breast-feed in the future, and wants to retain as much nipple/areola sensibility as possible. She is concerned that surgery on her breasts may increase her chance of getting breast cancer. What is the best evidence to guide her care?
The literature is replete with articles describing the various surgical techniques available for success in breast reduction. The purpose of this report is not to elaborate on technique but to present the most reliable recent evidence about outcomes from reduction mammaplasty.
Methods for Identifying Evidence
A literature search of PubMed, Cumulative Index to Nursing and Allied Health Literature, and the Cochrane Library was performed to obtain the best available evidence on reduction mammaplasty, with emphasis on preoperative assessment, anesthesia/analgesia, antibiotic and deep vein thrombosis prophylaxis, treatment, and outcomes. The following search terms were combined as appropriate, and PubMed MeSH terms were used when available: breast reduction, reduction mammaplasty, mammaplasty, diagnosis, preoperative assessment, risk factors, diabetes mellitus, breast cancer, hormone replacement therapy, oral contraceptives, smoking, body mass index, venous thrombosis, DVT, deep vein thrombosis, prevention, control, prophylaxis, antibiotics, methods, surgical treatment plan, treatment, anesthetics, premedication outcome, complications, postoperative complications, hematoma, necrosis, seroma, surgical wound dehiscence, scars, pain management, and analgesia. The initial search was limited to human studies that were published from 1999 to 2009 and indexed as meta-analyses, randomized controlled trials, clinical trials, or comparative studies; however, additional references were included if deemed necessary for discussion. Studies were excluded if the full text was inaccessible or of non-English language, as the study quality could not be evaluated. Relevant studies were appraised for quality and validity according to criteria published by the Critical Appraisal Skills Program1 and assigned a level of evidence with the American Society of Plastic Surgeons Evidence Rating Scales (Tables 1 and 2). Studies included in this review are identified below by the type of clinical
question (therapy or risk) and level of evidence. Evidence ratings were not assigned to studies with inadequately described methods and/or worrisome biases or to references included for discussion purposes only (e.g., narrative reviews).
question (therapy or risk) and level of evidence. Evidence ratings were not assigned to studies with inadequately described methods and/or worrisome biases or to references included for discussion purposes only (e.g., narrative reviews).
Table 1. American Society of Plastic Surgeons Evidence Rating Scale for Therapy | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Table 2. American Society of Plastic Surgeons Evidence Rating Scale for Risk | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Evidence on Preoperative Assessment
Breast hypertrophy has a significant impact on the health status and quality of life of women. Pain is a significant symptom, and both pain and overall health status are markedly improved by breast reduction. Collins et al., in a multicentered cohort study, confirmed that half of operative subjects had breast related pain in the back, neck, or shoulders all or most of the time before surgery, compared with less than 10 percent postoperatively (Therapy: Level II Evidence).2 Conservative treatments, including weight loss, did not provide permanent relief in operative subjects or hypertrophy controls. Medical necessity for breast reduction surgery is better defined by symptoms reported by the patient than by existing criteria, as documented by Kerrigan et al., who also quoted literature to support overwhelming evidence that reduction mammaplasty successfully relieves symptoms of breast hypertrophy (Therapy: Level II Evidence).3
Preoperative discussion about risk of breast cancer in patients desiring reduction mammaplasty should include results from cohort studies in both Canada and Sweden.4,5 The incidence of breast cancer in both retrospective, population-based cohort studies, with adjustments for both age and calendar years, and with follow-up intervals of 6.5 years in Canada and 7.5 years in Sweden, was decreased in patients with reduction mammaplasty compared with nonsurgical cohorts (Risk: Level II Evidence). In the report by Boice et al., the decrease in risk was most pronounced if the procedure was performed after age 50, and this Swedish study had a statistically significant 28 percent decreased risk of breast cancer, consistent with the protective effect of partial breast tissue removal.5 Using the data from Sweden, Brinton et al. demonstrated that the amount of breast tissue removed was a significant predictor of risk with the largest quantity of breast tissue removal correlating with lowest risk (Risk: Level III Evidence).6 In this group, risk improved significantly when compared with the risk for those with less tissue removed. This relationship persisted after adjusting for other breast carcinoma risk factors and persisted in every subgroup examined.
Evidence on Analgesia and Anesthesia
A prospective, single-blind, randomized controlled trial by Culliford et al. showed that in 40 patients, 20 in each group, those treated with a single dose of Sensorcaine (Astra Zeneca, London, England) irrigation through Jackson-Pratt drains were discharged home significantly faster than those treated with saline irrigation (Therapy: Level II Evidence).7 They also had shortened stays in the postanesthesia care unit and significantly decreased use of narcotic medication. DiMarco et al. also showed that patients treated with ropivacaine incision infiltration took significantly less postoperative narcotic in the 24-hour period than those in a control group infiltrated with saline (Therapy: Level II Evidence).8 Also in a randomized controlled trial, Fayman et al., studying both breast augmentation and breast reduction, demonstrated no significant difference in pain relief when local infiltration of bupivacaine was compared with ropivacaine, although it is important to note that a higher dose of ropivacaine was used,
which may have affected the outcome (Therapy: Level II Evidence).9 Pain relief, however, was statistically significantly better with either agent in reduction mammaplasty than augmentation.
which may have affected the outcome (Therapy: Level II Evidence).9 Pain relief, however, was statistically significantly better with either agent in reduction mammaplasty than augmentation.
An added benefit of using local anesthetics containing epinephrine is control of blood loss. Mataxotos et al. showed a significant reduction in perioperative blood loss when breasts were infiltrated with bupivacaine containing epinephrine in comparison with controls and also a significant difference in postoperative pain in the 3 hours following surgery (Therapy: Level II Evidence).10 In a prospective comparison study, Thomas et al. showed that epinephrine infiltration decreased intraoperative blood loss and facilitated the operation without the need for blood transfusion and with very few side effects (Therapy: Level II Evidence).11 Improvement in control of blood loss can also be achieved by hypotensive anesthetic techniques. Kop et al. showed that blood loss was significantly lower in the hypotensive group, although the study was not controlled for length of operation (Therapy: Level II Evidence).12
Evidence on Antibiotic Prophylaxis
The cost of antibiotics, potential allergies, concern about suppression of normal flora, or the production of resistant organisms during reduction mammaplasty have stimulated reviews of antibiotic use. Ahmadi et al. randomized 50 patients into three groups: (1) no antibiotics, (2) preoperative antibiotics only, and (3) preoperative and postoperative antibiotics (Therapy: Level II Evidence).13 Cefazolin was the antibiotic of choice. There were no differences in the groups regarding patient population or intraoperative data, such as specimen weight, operative time, blood loss, or positive bacterial cultures. Cultures from tissue biopsies during surgery were positive in 55 percent, with 90 percent showing staphlococcal epidermidis. There was no difference in the rate of infection among the three groups, but delayed wound healing was decreased in the group receiving preoperative antibiotics only, resulting in the conclusion that the timing of prophylactic antibiotics did not have an effect on infection but a single preoperative dose improved wound healing.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree