Key points
- •
Review and discuss LED physics.
- •
Describe and discuss tissue effects including ultrastructural and biochemical changes.
- •
Show indications and complications of LED therapy.
- •
Discuss current LED devices.
- •
Describe future of Leds in aesthetic medicine.
Introduction
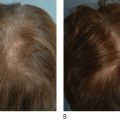
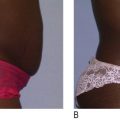
The 40-year evolution of therapeutic light-emitting diodes (LEDs), as a source for quasi-monochromatic or narrow bandwidths of light, has provided a novel, effective and multi-purpose addition to aesthetic and reconstructive practices. Despite the reported advantages for LED technology, the subject has remained controversial because the beneficial clinical effects have been either anecdotal in nature or lacking a convincing explanation of its actions at the subcellular level. This chapter will review the LED physics, LED-induced histological, ultrastructural and biochemical changes, patients’ indications, pre-operative preparation, techniques and clinical efficacy and side-effects of LED phototherapy, as used in primary (skin rejuvenation, cellulite reduction, hair stimulation) and secondary (acne treatment, rescue wound healing) aesthetic procedures.
Led theory for clinical skin treatment
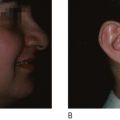
The use of LEDs and low intensity light therapy for stimulating wound healing by simultaneously up or down regulating intracellular activities in a non-thermal manner has revolutionized skin care. Photomodulation of living cellular activities by LED wavelengths has been shown to increase collagen and elastin production in fibroblasts, increase ATP production in healing cells, reduce the production of inflammatory interleukins, inhibit the production of matrix metallo-protease (collagenase), increase lymphatic drainage, and stimulate new vessel growth. Such cellular responses result in improved skin rejuvenation without the anticipated side effects of other thermally based skin care treatments. The clinical benefits of LED treatments can be observed in aesthetic skin rejuvenation and in reducing dermatitis after radiation therapy.
LED source
The most common LED-light source consists of diode heads for producing electro-magnetic radiation at specific preset wavelengths ( Table 10.1 ) that are incoherent, unpolarized, and, for the most part, non-thermal or low-thermal in origin. Most therapeutic devices precisely mount a number of LED diodes either in an articulated, multipanel array or in hand pieces that differ in their designs to account for the divergence of individual beams to maximize interference patterns, the exposed surface treatment area, and the external and internal dosimetry ( Box 10.1 ).
| Digital photography analysis | Profilometry | Skin elasticity | |
|---|---|---|---|
| Group 1 (830 nm) | 95.2% | 33.0% | 19.0% |
| Group 2 (633 nm) | 72.3% | 26.0% | 14.0% |
| Group 3 (633/830 nm) | 95.5% | 36.0% | 16.0% |
| Group 4 (sham) | 13.3% | no change | no change |
LED physics
The dosimetry of any LED treatment must be considered in two parts, the external dosimetry which involves quantum energies directly controlled by the provider (within the limits of the device) and the internal dosimetry, which is difficult to determine in vivo because the optical properties and heterogeneity of the tissue at each wavelength are not well known. Mathematical diffusion models, however, project that the light energy fluence rate (the rate at which photochemical reactions occur at the cellular level) falls to 0.7% of its effective initial value at the skin surface (external dose) at a distance of 5 mm of penetration depths into the tissue. Interpolation of data from mathematical models in fair Caucasian skin suggest that red light (630 nm), for example, penetrates on the average of 1–2 mm, while near-infrared light (800–900 nm) penetrates about twice as long. The often claimed red light penetration of 5–10 mm by commercial systems does not take into account tissue attenuation, which results in only 1% of the incident light playing a phototherapeutic role within the deeper tissue. Since facial skin thickness varies from 1 mm to as much as 4 mm (average 2 mm), the majority of photobiomodulation effects are expected to occur within the skin level.
LED photobiomodulation
Internal dosimetry is a measure of the rate that a quantity of LED-induced photons penetrates and interacts with mitochondrial non-specialized photoacceptor antennae molecules (flavoprotein NADH-dehydrogenases and cytochrome c oxidase) within targeted cells in the epidermal and dermal layers. The induced cascades of differing redox reactions produce an immediate response in seconds, such as the conversion of adenosine diphosphate (ADP) to adenosine triphosphate (ATP), which can up-regulate or down-regulate by gene expression a wide range of pathway actions.
On the other hand, LED energy can also produce many secondary biochemical reactions in cells that occur hours or days later after the LED irradiation is switched off. Of particular interest is the consistent finding that biological reactions occur in cells that are not directly irradiated by non-thermal LED energy, commonly referred to as the ‘bystander effect’. It is believed that the irradiated cells may release intercellular signaling molecules that proprogate the photobiomodulation effects between cells even within the deeper dermis, resulting in modulations in cell functions, cell proliferation and repair of compromised cells. LED-induced photobiomodulations are specifically coded by the preset wavelengths, fluences, pulsing on-times and off-times, and dedicated duty cycles that result in at least 24 immediate diverse cellular actions. More than one of the cellular actions (e.g., synthesis of ATP, mRNA, and reverse transcriptase-polymerase change reactions, collagen and elastin production, increased levels of tissue inhibitor of matrix metalloproteinase, decreased levels of matrix metalloproteinase, and induced mitosis or apoptosis) can be initiated, inhibited, or modulated by LED energy from one or more wavelengths by unknown regulatory mechanisms.
Controlled biochemical, histological–ultrastructural and clinical and in vitro studies
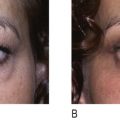
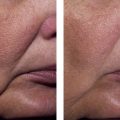
Recent peer-reviewed publications (e.g. ) have reported encouraging results after LED exposures in clinical trials that utilize a randomized, double blinded, placebo-controlled hemi-faced methodology (treated and covered sides of face in the same patient and at a sham site). In one such clinical study of 76 patients, all subjects were randomly divided into four groups to treat only the right side of their facial skin by LED phototherapy. Hemi-faces received either 830 nm near-infrared alone (Group 1), 633 nm red alone (Group 2), a combination of 633 nm and 830 nm (Group 3), or a sham treatment light (Group 4), twice a week for 4 weeks. Clinical assessment of the periorbital rhytids was evaluated after 3 months by blinded digital photographic analyses (5 point scale), profilometric silicone imprints with a Viometer SV600, and skin elasticity with a Cutometer®, as listed in Table 10.1 . The cumulative data indicated not only that LED energy results in objective improvements in skin over sham controls, but also that a greater clinical efficacy was observed with longer (more penetration) wavelengths and with combined 800 nm and 633 nm wavelength exposures.
Results from tissue studies at 2 weeks only in all post-treatment Groups 1, 2 and 3 demonstrated not only increased collagen throughout the entire dermis, especially in the papillary and upper reticular dermis with a clearly defined grenz zone by Masson trichome stains, but also significant increase of normal, non-elastotically degenerated elastin fibers down to the mid-reticular dermis by Alcian blue and Verhoeff–van Gieson stains. These histologic findings were further confirmed by transmission electromicroscopy only in Groups 1, 2 and 3 with the presence of increased size and number of collagen and elastin fibers along with numerous activated fibroblasts containing enlarged endoplasmic reticula.
Although the immunohistochemistry results demonstrated no significant decrease in matrix metalloproteinase (collagenase) staining in MMP-1 or MMP-2 in Groups 1, 2 and 3 between baseline and at 2 weeks later, noticeable increases in tissue inhibitor of matrix metalloproteinase (TIMP) staining were observed in the treatment groups that may keep the newly produced collagen from MMP degradation. It is very possible that MMPs may be synthesized earlier than 2 weeks because mRNA levels of pro-inflammatory cytokines (IL-1β, TNF-α and Cx43) were observed to be elevated 20 minutes after treatment in Groups 1, 2 and 3 ( Table 10.2 ) and are known to induce the subsequent production of MMPs. From such controlled data arises the postulation that LED treatment induces immediately the release of pro-inflammatory cytokines that both increase collagen/elastin formation and limits their degradation by reduction of MMPs and simultaneous stimulation of TIMPs.
| IL-1β (interleukin-6) | TNF-α (tumor necrosis factor-α) | Cx43 (connexin-43) | |
|---|---|---|---|
| Group 1 (830 nm) | 13.3 | 1.6 | 1.31 |
| Group 2 (633 nm) | 6.165 | 1.36 | 2.53 |
| Group 3 (633/830 nm) | 13.7 | 2.5 | 2.42 |
| Group 4 (sham) | 1.59 | 0.38 | 0.92 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree